Related Research Articles

Chemotherapy is a type of cancer treatment that uses one or more anti-cancer drugs as part of a standardized chemotherapy regimen. Chemotherapy may be given with a curative intent or it may aim to prolong life or to reduce symptoms. Chemotherapy is one of the major categories of the medical discipline specifically devoted to pharmacotherapy for cancer, which is called medical oncology.

Small-cell carcinoma is a type of highly malignant cancer that most commonly arises within the lung, although it can occasionally arise in other body sites, such as the cervix, prostate, and gastrointestinal tract. Compared to non-small cell carcinoma, small cell carcinoma is more aggressive, with a shorter doubling time, higher growth fraction, and earlier development of metastases.

Anaplastic large-cell lymphoma (ALCL) refers to a group of non-Hodgkin lymphomas in which aberrant T cells proliferate uncontrollably. Considered as a single entity, ALCL is the most common type of peripheral lymphoma and represents ~10% of all peripheral lymphomas in children. The incidence of ALCL is estimated to be 0.25 cases per 100,000 people in the United States of America. There are four distinct types of anaplastic large-cell lymphomas that on microscopic examination share certain key histopathological features and tumor marker proteins. However, the four types have very different clinical presentations, gene abnormalities, prognoses, and/or treatments.
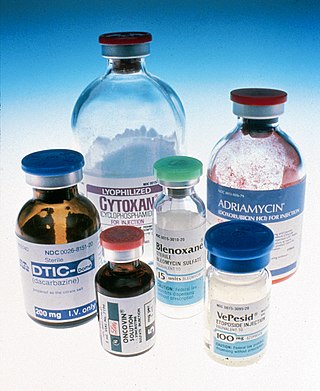
The era of cancer chemotherapy began in the 1940s with the first use of nitrogen mustards and folic acid antagonist drugs. The targeted therapy revolution has arrived, but many of the principles and limitations of chemotherapy discovered by the early researchers still apply.

Procarbazine is a chemotherapy medication used for the treatment of Hodgkin's lymphoma and brain cancers. For Hodgkin's it is often used together with chlormethine, vincristine, and prednisone while for brain cancers such as glioblastoma multiforme it is used with lomustine and vincristine. It is typically taken by mouth.
CHOP is the acronym for a chemotherapy regimen used in the treatment of non-Hodgkin lymphoma. CHOP consists of:
Stanford V, is a chemotherapy regimen intended as a first-line treatment for Hodgkin lymphoma. The regimen was developed in 1988, with the objective of maintaining a high remission rate while reducing the incidence of acute and long term toxicity, pulmonary damage, and sterility observed in alternative treatment regimens such as ABVD. The chemical agents used are:
ABVD is a chemotherapy regimen used in the first-line treatment of Hodgkin lymphoma, replacing the older MOPP protocol. It consists of concurrent treatment with the chemotherapy drugs:
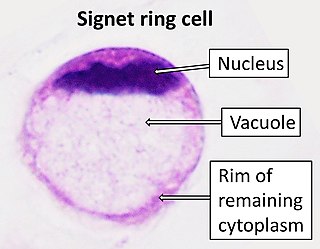
Signet ring cell carcinoma (SRCC) is a rare form of highly malignant adenocarcinoma that produces mucin. It is an epithelial malignancy characterized by the histologic appearance of signet ring cells.
Esthesioneuroblastoma is a rare cancer of the nasal cavity. Arising from the upper nasal tract, esthesioneuroblastoma is believed to originate from sensory neuroepithelial cells, also known as neuroectodermal olfactory cells.
ICE in the context of chemotherapy is an acronym for one of the chemotherapy regimens, used in salvage treatment of relapsed or refractory non-Hodgkin's lymphoma and Hodgkin lymphoma.

Langerhans cell sarcoma (LCS) is a rare form of malignant histiocytosis. It should not be confused with Langerhans cell histiocytosis, which is cytologically benign. It can present most commonly in the skin and lymphatic tissue, but may also present in the lung, liver, and bone marrow. Treatment is most commonly with surgery or chemotherapy.

Follicular dendritic cell sarcoma (FDCS) is an extremely rare neoplasm. While the existence of FDC tumors was predicted by Lennert in 1978, the tumor wasn't fully recognized as its own cancer until 1986 after characterization by Monda et al. It accounts for only 0.4% of soft tissue sarcomas, but has significant recurrent and metastatic potential and is considered an intermediate grade malignancy. The major hurdle in treating FDCS has been misdiagnosis. It is a newly characterized cancer, and because of its similarities in presentation and markers to lymphoma, both Hodgkin and Non-Hodgkin subtypes, diagnosis of FDCS can be difficult. With recent advancements in cancer biology better diagnostic assays and chemotherapeutic agents have been made to more accurately diagnose and treat FDCS.
Treatment of lung cancer refers to the use of medical therapies, such as surgery, radiation, chemotherapy, immunotherapy, percutaneous ablation, and palliative care, alone or in combination, in an attempt to cure or lessen the adverse impact of malignant neoplasms originating in lung tissue.
Targeted therapy of lung cancer refers to using agents specifically designed to selectively target molecular pathways responsible for, or that substantially drive, the malignant phenotype of lung cancer cells, and as a consequence of this (relative) selectivity, cause fewer toxic effects on normal cells.
Chemotherapy-induced nausea and vomiting (CINV) is a common side-effect of many cancer treatments. Nausea and vomiting are two of the most feared cancer treatment-related side effects for cancer patients and their families. In 1983, Coates et al. found that patients receiving chemotherapy ranked nausea and vomiting as the first and second most severe side effects, respectively. Up to 20% of patients receiving highly emetogenic agents in this era postponed, or even refused, potentially curative treatments. Since the 1990s, several novel classes of antiemetics have been developed and commercialized, becoming a nearly universal standard in chemotherapy regimens, and helping to better manage these symptoms in a large portion of patients. Efficient mediation of these unpleasant and sometimes debilitating symptoms results in increased quality of life for the patient, and better overall health of the patient, and, due to better patient tolerance, more effective treatment cycles.
High-dose chemotherapy and bone marrow transplant (HDC/BMT), also high-dose chemotherapy with autologous bone marrow transplant, was an ineffective treatment regimen for metastatic breast cancer, and later high-risk breast cancer, that was considered promising during the 1980s and 1990s. With an overall idea that more is better, this process involved taking cells from the person's bone marrow to store in a lab, then to give such high doses of chemotherapy drugs that the remaining bone marrow was destroyed, and then to inject the cells taken earlier back into the body as replacement. It was ultimately determined to be no more effective than normal treatment, and to have significantly higher side effects, including treatment-related death.

Durvalumab, sold under the brand name Imfinzi, is an FDA-approved immunotherapy for cancer, developed by Medimmune/AstraZeneca. It is a human immunoglobulin G1 kappa (IgG1κ) monoclonal antibody that blocks the interaction of programmed cell death ligand 1 (PD-L1) with the PD-1 (CD279).
This is a historical timeline of the development and progress of cancer treatments, which includes time of discovery, progress, and approval of the treatments.
Metronomic therapy is a new type of chemotherapy in which anti-cancer drugs are administered in a lower dose than the maximum tolerated dose repetitively over a long period to treat cancers with fewer side effects. Metronomic therapy is shown to affect both tumor microenvironment and tumor cells to achieve its therapeutic effects. Metronomic therapy is also cost-effective as a lower dose is used compared to conventional chemotherapy. The use of metronomic therapy has been extensively investigated and can be advantageous in selected group of patients. Yet, more clinical trials are necessary to generalize the method.
References
- ↑ Mayer, RJ, Targeted therapy for advanced colorectal cancer -- more is not always better, N Engl J Med. 2009;360:623 which is a [possibly critical and not peer reviewed] comment to Chemotherapy, bevacizumab, and cetuximab in metastatic colorectal cancer.
- ↑ Cancer.net - Explaining Maintenance Therapy
- ↑ BEACOPP chemotherapy regimen
- ↑ "MVAC Still the 'Best Treatment' for Advanced Bladder Cancer Patients. 1999". Archived from the original on 2012-01-09. Retrieved 2010-11-18.
- ↑ "Ovarian Cancer Chemotherapy: Know Your Treatment Options".
- ↑ Rodriguez Victorio (1973). "POMP combination chemotherapy of adult acute leukemia". Cancer. 32 (1): 69–75. doi: 10.1002/1097-0142(197307)32:1<69::AID-CNCR2820320109>3.0.CO;2-0 . PMID 4515259.
- ↑ Kiesewetter B, Mayerhoefer ME, Lukas J, Zielinski CC, Müllauer L, Raderer M (2014). "Rituximab plus bendamustine is active in pretreated patients with extragastric marginal zone B cell lymphoma of the mucosa-associated lymphoid tissue (MALT lymphoma)". Ann. Hematol. 93 (2): 249–53. doi:10.1007/s00277-013-1865-3. PMID 23925930. S2CID 12851937.
- 1 2 3 4 5 Treatment of Wilms Tumor at National Cancer Institute. Last Modified: 03/29/2012
- ↑ El Weshi, A; Memon, M; Raja, M; Bazarbashi, S; Rahal, M; El Foudeh, M; Pai, C; Allam, A; El Hassan, I; Ezzat, A (October 2004). "VIP (etoposide, ifosfamide, cisplatin) in adult patients with recurrent or refractory Ewing sarcoma family of tumors". American Journal of Clinical Oncology. 27 (5): 529–34. doi:10.1097/01.coc.0000135815.94162.83. PMID 15596925. S2CID 6362786.
- ↑ Kosmidis, P; Mylonakis, N; Fountzilas, G; Pavlidis, N; Samantas, E; Karabelis, A; Kattis, K; Skarlos, D (July 1996). "A prospective randomized phase III study in non-small-cell lung cancer comparing cisplatin, ifosfamide, vinblastine (VIP) versus cisplatin, ifosfamide and etoposide (VIP-16). Hellenic Co-Operative Oncology Group". Annals of Oncology. 7 (5): 517–20. doi: 10.1093/oxfordjournals.annonc.a010642 . PMID 8839908.