
A nuclear weapon is an explosive device that derives its destructive force from nuclear reactions, either fission or a combination of fission and fusion reactions, producing a nuclear explosion. Both bomb types release large quantities of energy from relatively small amounts of matter.
A neutron bomb, officially defined as a type of enhanced radiation weapon (ERW), is a low-yield thermonuclear weapon designed to maximize lethal neutron radiation in the immediate vicinity of the blast while minimizing the physical power of the blast itself. The neutron release generated by a nuclear fusion reaction is intentionally allowed to escape the weapon, rather than being absorbed by its other components. The neutron burst, which is used as the primary destructive action of the warhead, is able to penetrate enemy armor more effectively than a conventional warhead, thus making it more lethal as a tactical weapon.

Nuclear fallout is the residual radioactive material propelled into the upper atmosphere following a nuclear blast, so called because it "falls out" of the sky after the explosion and the shock wave has passed. It commonly refers to the radioactive dust and ash created when a nuclear weapon explodes. The amount and spread of fallout is a product of the size of the weapon and the altitude at which it is detonated. Fallout may get entrained with the products of a pyrocumulus cloud and fall as black rain. This radioactive dust, usually consisting of fission products mixed with bystanding atoms that are neutron-activated by exposure, is a form of radioactive contamination.

Nuclear technology is technology that involves the nuclear reactions of atomic nuclei. Among the notable nuclear technologies are nuclear reactors, nuclear medicine and nuclear weapons. It is also used, among other things, in smoke detectors and gun sights.

Nuclear weapon designs are physical, chemical, and engineering arrangements that cause the physics package of a nuclear weapon to detonate. There are three existing basic design types:

A mushroom cloud is a distinctive mushroom-shaped flammagenitus cloud of debris, smoke, and usually condensed water vapor resulting from a large explosion. The effect is most commonly associated with a nuclear explosion, but any sufficiently energetic detonation or deflagration will produce the same effect. They can be caused by powerful conventional weapons, like thermobaric weapons such as the ATBIP and GBU-43/B MOAB. Some volcanic eruptions and impact events can produce natural mushroom clouds.

Radiological warfare is any form of warfare involving deliberate radiation poisoning or contamination of an area with radiological sources.

Nuclear fission products are the atomic fragments left after a large atomic nucleus undergoes nuclear fission. Typically, a large nucleus like that of uranium fissions by splitting into two smaller nuclei, along with a few neutrons, the release of heat energy, and gamma rays. The two smaller nuclei are the fission products..

Neutron activation is the process in which neutron radiation induces radioactivity in materials, and occurs when atomic nuclei capture free neutrons, becoming heavier and entering excited states. The excited nucleus decays immediately by emitting gamma rays, or particles such as beta particles, alpha particles, fission products, and neutrons. Thus, the process of neutron capture, even after any intermediate decay, often results in the formation of an unstable activation product. Such radioactive nuclei can exhibit half-lives ranging from small fractions of a second to many years.

Iodine-131 is an important radioisotope of iodine discovered by Glenn Seaborg and John Livingood in 1938 at the University of California, Berkeley. It has a radioactive decay half-life of about eight days. It is associated with nuclear energy, medical diagnostic and treatment procedures, and natural gas production. It also plays a major role as a radioactive isotope present in nuclear fission products, and was a significant contributor to the health hazards from open-air atomic bomb testing in the 1950s, and from the Chernobyl disaster, as well as being a large fraction of the contamination hazard in the first weeks in the Fukushima nuclear crisis. This is because 131I is a major fission product of uranium and plutonium, comprising nearly 3% of the total products of fission. See fission product yield for a comparison with other radioactive fission products. 131I is also a major fission product of uranium-233, produced from thorium.

There are 37 known isotopes of iodine (53I) from 108I to 144I; all undergo radioactive decay except 127I, which is stable. Iodine is thus a monoisotopic element.
A pure fusion weapon is a hypothetical hydrogen bomb design that does not need a fission "primary" explosive to ignite the fusion of deuterium and tritium, two heavy isotopes of hydrogen used in fission-fusion thermonuclear weapons. Such a weapon would require no fissile material and would therefore be much easier to develop in secret than existing weapons. Separating weapons-grade uranium (U-235) or breeding plutonium (Pu-239) requires a substantial and difficult-to-conceal industrial investment, and blocking the sale and transfer of the needed machinery has been the primary mechanism to control nuclear proliferation to date.

Cobalt-60 (60Co) is a synthetic radioactive isotope of cobalt with a half-life of 5.2714 years. It is produced artificially in nuclear reactors. Deliberate industrial production depends on neutron activation of bulk samples of the monoisotopic and mononuclidic cobalt isotope 59
Co
. Measurable quantities are also produced as a by-product of typical nuclear power plant operation and may be detected externally when leaks occur. In the latter case the incidentally produced 60
Co
is largely the result of multiple stages of neutron activation of iron isotopes in the reactor's steel structures via the creation of its 59
Co
precursor. The simplest case of the latter would result from the activation of 58
Fe
. 60
Co
undergoes beta decay to the stable isotope nickel-60. The activated cobalt nucleus emits two gamma rays with energies of 1.17 and 1.33 MeV, hence the overall equation of the nuclear reaction is: 59
27Co
+ n → 60
27Co
→ 60
28Ni
+ e− + 2 γ

Caesium-137, cesium-137 (US), or radiocaesium, is a radioactive isotope of caesium that is formed as one of the more common fission products by the nuclear fission of uranium-235 and other fissionable isotopes in nuclear reactors and nuclear weapons. Trace quantities also originate from spontaneous fission of uranium-238. It is among the most problematic of the short-to-medium-lifetime fission products. Caesium-137 has a relatively low boiling point of 671 °C (1,240 °F) and easily becomes volatile when released suddenly at high temperature, as in the case of the Chernobyl nuclear accident and with atomic explosions, and can travel very long distances in the air. After being deposited onto the soil as radioactive fallout, it moves and spreads easily in the environment because of the high water solubility of caesium's most common chemical compounds, which are salts. Caesium-137 was discovered by Glenn T. Seaborg and Margaret Melhase.

Environmental radioactivity is produced by radioactive materials in the human environment. While some radioisotopes, such as strontium-90 (90Sr) and technetium-99 (99Tc), are only found on Earth as a result of human activity, and some, like potassium-40 (40K), are only present due to natural processes, a few isotopes, e.g. tritium (3H), result from both natural processes and human activities. The concentration and location of some natural isotopes, particularly uranium-238 (238U), can be affected by human activity.

Strontium-90 is a radioactive isotope of strontium produced by nuclear fission, with a half-life of 28.8 years. It undergoes β− decay into yttrium-90, with a decay energy of 0.546 MeV. Strontium-90 has applications in medicine and industry and is an isotope of concern in fallout from nuclear weapons, nuclear weapons testing, and nuclear accidents.
This article compares the radioactivity release and decay from the Chernobyl disaster with various other events which involved a release of uncontrolled radioactivity.
The Pechora–Kama Canal, or sometimes the Kama–Pechora Canal, was a proposed canal intended to link the basin of the Pechora River in the north of European Russia with the basin of the Kama, a tributary of the Volga. Completion of this project would integrate the Pechora into the system of waterways of European Russia, centered on the Volga – something that was of importance before the advent of railways, or before the first railway reached the Pechora in the 1940s. Later the project was proposed mostly to transfer Pechora's water to the Volga and further on to the Caspian Sea.
A salted bomb is a nuclear weapon designed to function as a radiological weapon by producing larger quantities of radioactive fallout than unsalted nuclear arms. This fallout can render a large area uninhabitable. The term is derived both from the means of their manufacture, which involves the incorporation of additional elements to a standard atomic weapon, and from the expression "to salt the earth", meaning to render an area uninhabitable for generations. The idea originated with Hungarian-American physicist Leo Szilard, in February 1950. His intent was not to propose that such a weapon be built, but to show that nuclear weapon technology would soon reach the point where it could end human life on Earth.
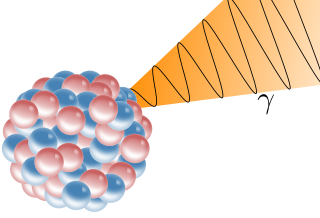
A gamma ray, also known as gamma radiation (symbol
γ
), is a penetrating form of electromagnetic radiation arising from the radioactive decay of atomic nuclei. It consists of the shortest wavelength electromagnetic waves, typically shorter than those of X-rays. With frequencies above 30 exahertz (3×1019 Hz), each gamma ray imparts the highest photon energy of any form of electromagnetic radiation. Paul Villard, a French chemist and physicist, discovered gamma radiation in 1900 while studying radiation emitted by radium. In 1903, Ernest Rutherford named this radiation gamma rays based on their relatively strong penetration of matter; in 1900 he had already named two less penetrating types of decay radiation (discovered by Henri Becquerel) alpha rays and beta rays in ascending order of penetrating power.














