Benign prostatic hyperplasia (BPH), also called prostate enlargement, is a noncancerous increase in size of the prostate gland. Symptoms may include frequent urination, trouble starting to urinate, weak stream, inability to urinate, or loss of bladder control. Complications can include urinary tract infections, bladder stones, and chronic kidney problems.
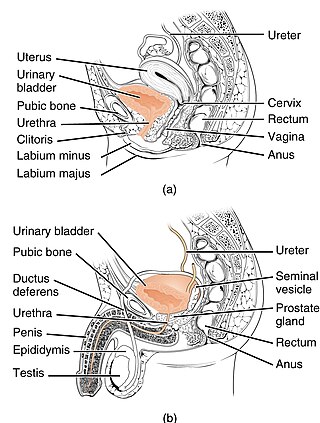
Urinary incontinence (UI), also known as involuntary urination, is any uncontrolled leakage of urine. It is a common and distressing problem, which may have a large impact on quality of life. It has been identified as an important issue in geriatric health care. The term enuresis is often used to refer to urinary incontinence primarily in children, such as nocturnal enuresis. UI is an example of a stigmatized medical condition, which creates barriers to successful management and makes the problem worse. People may be too embarrassed to seek medical help, and attempt to self-manage the symptom in secrecy from others.
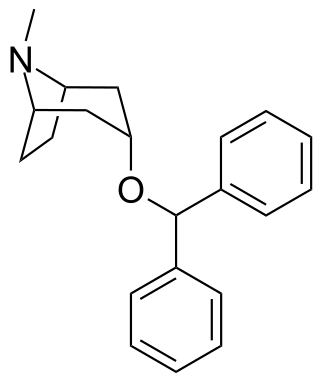
Benzatropine (INN), known as benztropine in the United States and Japan, is a medication used to treat movement disorders like parkinsonism and dystonia, as well as extrapyramidal side effects of antipsychotics, including akathisia. It is not useful for tardive dyskinesia. It is taken by mouth or by injection into a vein or muscle. Benefits are seen within two hours and last for up to ten hours.
Anticholinergics are substances that block the action of the neurotransmitter called acetylcholine (ACh) at synapses in the central and peripheral nervous system.
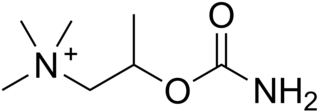
Bethanechol is a parasympathomimetic choline carbamate that selectively stimulates muscarinic receptors without any effect on nicotinic receptors. Unlike acetylcholine, bethanechol is not hydrolyzed by cholinesterase and will therefore have a long duration of action. Bethanechol is sold under the brand names Duvoid (Roberts), Myotonachol (Glenwood), Urecholine and Urocarb (Hamilton). The name bethanechol refers to its structure as the urethane of beta-methylcholine.

Tolterodine, sold under the brand name Detrol among others, is a medication used to treat frequent urination, urinary incontinence, or urinary urgency. Effects are seen within an hour. It is taken by mouth.
Percutaneous tibial nerve stimulation (PTNS), also referred to as posterior tibial nerve stimulation, is the least invasive form of neuromodulation used to treat overactive bladder (OAB) and the associated symptoms of urinary urgency, urinary frequency and urge incontinence. These urinary symptoms may also occur with interstitial cystitis and following a radical prostatectomy. Outside the United States, PTNS is also used to treat fecal incontinence.

Oxybutynin, sold as under the brand names Ditropan among others, is a medication used to treat overactive bladder. It works similar to tolterodine, Darifenacin, and Solifenacin. While used for bed wetting in children, evidence to support this use is poor. It is taken by mouth or applied to the skin.

Solifenacin, sold as the brand name Vesicare among others, is a medicine used to treat overactive bladder and neurogenic detrusor overactivity (NDO). It may help with incontinence, urinary frequency, and urinary urgency.

Trospium chloride is used to treat overactive bladder.

Overactive bladder (OAB) is a common condition where there is a frequent feeling of needing to urinate to a degree that it negatively affects a person's life. The frequent need to urinate may occur during the day, at night, or both. Loss of bladder control may occur with this condition. Overactive bladder affects approximately 11% of the population and more than 40% of people with overactive bladder have incontinence. Conversely, about 40% to 70% of urinary incontinence is due to overactive bladder. Overactive bladder is not life-threatening, but most people with the condition have problems for years.

Propiverine is an anticholinergic drug used for the treatment of urinary urgency, frequency and urge incontinence, all symptoms of overactive bladder syndrome. It is a muscarinic antagonist. A modified release preparation is also available, taken once daily.

Fesoterodine is an antimuscarinic drug developed by Schwarz Pharma AG to treat overactive bladder syndrome (OAB). It was approved by the European Medicines Agency in April 2007, the US Food and Drug Administration on October 31, 2008 and Health Canada on February 9, 2012.
Lower urinary tract symptoms (LUTS) refer to a group of clinical symptoms involving the bladder, urinary sphincter, urethra and, in men, the prostate. The term is more commonly applied to men—over 40% of older men are affected—but lower urinary tract symptoms also affect women. The condition is also termed prostatism in men, but LUTS is preferred.

Overflow incontinence is a concept of urinary incontinence, characterized by the involuntary release of urine from an overfull urinary bladder, often in the absence of any urge to urinate. This condition occurs in people who have a blockage of the bladder outlet, or when the muscle that expels urine from the bladder is too weak to empty the bladder normally. Overflow incontinence may also be a side effect of certain medications.
Treatments for overactive bladder are therapies used to treat overactive bladder or related conditions, such as urinary incontinence and frequent urination. Behavioral modification and medications are commonly used to treat this condition.

Methoctramine is a polymethylene tetraamine that acts as a muscarinic antagonist. It preferentially binds to the pre-synaptic receptor M2, a muscarinic acetylcholine ganglionic protein complex present basically in heart cells. In normal conditions -absence of methoctramine-, the activation of M2 receptors diminishes the speed of conduction of the sinoatrial and atrioventricular nodes thus reducing the heart rate. Thanks to its apparently high cardioselectivity, it has been studied as a potential parasymphatolitic drug, particularly against bradycardia. However, currently it is only addressed for research purposes, since the administration to humans is still unavailable.
Jerry G. Blaivas is an American urologist and senior faculty at the Icahn School of Medicine at Mount Sinai in New York City and adjunct professor of Urology at SUNY Downstate Medical School in Brooklyn, as well as professor of clinical urology at College of Physicians and Surgeons, Columbia University and clinical professor of Urology at Weill Medical College of Cornell University. He has four patents pending, has received four research grants for which he served as the principal investigator, and served as a major in the United States Army assigned to the Walson Army Hospital. He additionally served as president for the Urodynamic Society.
The β3 adrenergic receptor agonist or β3-adrenoceptor agonist, also known as β3-AR agonist, are a class of medicine that bind selectively to β3-adrenergic receptors.

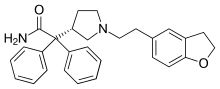
Vibegron, sold under the brand name Gemtesa, is a medication for the treatment of overactive bladder. Vibegron is a selective beta-3 adrenergic receptor agonist.