Arsenic is a chemical element; it has symbol As and atomic number 33. It is a metalloid and one of the pnictogens, and therefore shares many properties with its group 15 neighbors phosphorus and antimony. Arsenic is a notoriously toxic heavy metal. It occurs naturally in many minerals, usually in combination with sulfur and metals, but also as a pure elemental crystal. It has various allotropes, but only the grey form, which has a metallic appearance, is important to industry.

Hypothetical types of biochemistry are forms of biochemistry agreed to be scientifically viable but not proven to exist at this time. The kinds of living organisms currently known on Earth all use carbon compounds for basic structural and metabolic functions, water as a solvent, and DNA or RNA to define and control their form. If life exists on other planets or moons it may be chemically similar, though it is also possible that there are organisms with quite different chemistries – for instance, involving other classes of carbon compounds, compounds of another element, or another solvent in place of water.

An extremophile is an organism that is able to live in extreme environments, i.e., environments with conditions approaching or stretching the limits of what known life can adapt to, such as extreme temperature, pressure, radiation, salinity, or pH level.

Thiomargarita namibiensis is a harmless, gram-negative, facultative anaerobic, coccoid bacterium found in the ocean sediments of the continental shelf of Namibia. The genus name Thiomargarita means "sulfur pearl." This refers to the appearance of the cells as they contain microscopic sulfur granules that scatter incident light, lending the cell a pearly luster. This causes the cells to form chains, resembling strings of pearls. The species name namibiensis means "of Namibia", which is an ode to their country of discovery and existence. Together, Thiomargarita namibiensis means “Sulfur pearl of Namibia".

Paul Charles William Davies is an English physicist, writer and broadcaster, a professor in Arizona State University and director of BEYOND: Center for Fundamental Concepts in Science. He is affiliated with the Institute for Quantum Studies in Chapman University in California. He previously held academic appointments in the University of Cambridge, University College London, University of Newcastle upon Tyne, University of Adelaide and Macquarie University. His research interests are in the fields of cosmology, quantum field theory, and astrobiology.

Prochlorococcus is a genus of very small (0.6 μm) marine cyanobacteria with an unusual pigmentation. These bacteria belong to the photosynthetic picoplankton and are probably the most abundant photosynthetic organism on Earth. Prochlorococcus microbes are among the major primary producers in the ocean, responsible for a large percentage of the photosynthetic production of oxygen. Prochlorococcus strains, called ecotypes, have physiological differences enabling them to exploit different ecological niches. Analysis of the genome sequences of Prochlorococcus strains show that 1,273 genes are common to all strains, and the average genome size is about 2,000 genes. In contrast, eukaryotic algae have over 10,000 genes.

Halomonadaceae is a family of halophilic Pseudomonadota.
The arsenate is an ion with the chemical formula AsO3−4. Bonding in arsenate consists of a central arsenic atom, with oxidation state +5, double bonded to one oxygen atom and single bonded to a further three oxygen atoms. The four oxygen atoms orient around the arsenic atom in a tetrahedral geometry. Resonance disperses the ion's −3 charge across all four oxygen atoms.
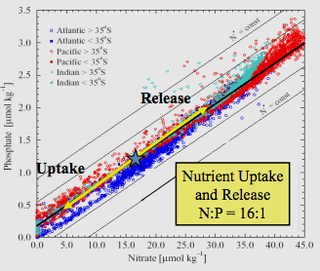
The Redfield ratio or Redfield stoichiometry is the consistent atomic ratio of carbon, nitrogen and phosphorus found in marine phytoplankton and throughout the deep oceans.

Phosphate solubilizing bacteria (PSB) are beneficial bacteria capable of solubilizing inorganic phosphorus from insoluble compounds. P-solubilization ability of rhizosphere microorganisms is considered to be one of the most important traits associated with plant phosphate nutrition. It is generally accepted that the mechanism of mineral phosphate solubilization by PSB strains is associated with the release of low molecular weight organic acids, through which their hydroxyl and carboxyl groups chelate the cations [an ion that have positive charge on it.] bound to phosphate, thereby converting it into soluble forms. PSB have been introduced to the Agricultural community as phosphate Biofertilizer. Phosphorus (P) is one of the major essential macronutrients for plants and is applied to soil in the form of phosphate fertilizers. However, a large portion of soluble inorganic phosphate which is applied to the soil as chemical fertilizer is immobilized rapidly and becomes unavailable to plants. Currently, the main purpose in managing soil phosphorus is to optimize crop production and minimize P loss from soils. PSB have attracted the attention of agriculturists as soil inoculums to improve the plant growth and yield. When PSB is used with rock phosphate, it can save about 50% of the crop requirement of phosphatic fertilizer. The use of PSB as inoculants increases P uptake by plants. Simple inoculation of seeds with PSB gives crop yield responses equivalent to 30 kg P2O5 /ha or 50 percent of the need for phosphatic fertilizers. Alternatively, PSB can be applied through fertigation or in hydroponic operations. Many different strains of these bacteria have been identified as PSB, including Pantoea agglomerans (P5), Microbacterium laevaniformans (P7) and Pseudomonas putida (P13) strains are highly efficient insoluble phosphate solubilizers. Recently, researchers at Colorado State University demonstrated that a consortium of four bacteria, synergistically solubilize phosphorus at a much faster rate than any single strain alone. Mahamuni and Patil (2012) isolated four strains of phosphate solubilizing bacteria from sugarcane (VIMP01 and VIMP02) and sugar beet rhizosphere (VIMP03 and VIMP 04). Isolates were strains of Burkholderia named as VIMP01, VIMP02, VIMP03 and VIMP04. VIMP (Vasantdada Sugar Institute Isolate by Mahamuni and Patil) cultures were identified as Burkholderia cenocepacia strain VIMP01 (JQ867371), Burkholderia gladioli strain VIMP02 (JQ811557), Burkholderia gladioli strain VIMP03 (JQ867372) and Burkholderia species strain VIMP04 (JQ867373).

Felisa Wolfe-Simon is an American microbial geobiologist and biogeochemist. In 2010, Wolfe-Simon led a team that discovered GFAJ-1, an extremophile bacterium that they claimed was capable of substituting arsenic for a small percentage of its phosphorus to sustain its growth, thus advancing the remarkable possibility of non-RNA/DNA-based genetics. However, these conclusions were immediately debated and criticized in correspondence to the original journal of publication, and have since come to be widely disbelieved, though they have never been disputed by any legitimate scientific studies. In 2012, two reports refuting the most significant aspects of the original results were published in the same journal in which the original findings had been previously published.
Arsenic biochemistry refers to biochemical processes that can use arsenic or its compounds, such as arsenate. Arsenic is a moderately abundant element in Earth's crust, and although many arsenic compounds are often considered highly toxic to most life, a wide variety of organoarsenic compounds are produced biologically and various organic and inorganic arsenic compounds are metabolized by numerous organisms. This pattern is general for other related elements, including selenium, which can exhibit both beneficial and deleterious effects. Arsenic biochemistry has become topical since many toxic arsenic compounds are found in some aquifers, potentially affecting many millions of people via biochemical processes.

Halomonas titanicae is a gram-negative, halophilic species of bacteria which was isolated in 2010 from rusticles recovered from the wreck of the RMS Titanic. It has been estimated by Henrietta Mann, one of the researchers that first isolated it, that the action of microbes like Halomonas titanicae may bring about the total deterioration of the Titanic by 2030. While the bacteria have been identified as a potential danger to oil rigs and other man-made objects in the deep sea, they also have the potential to be used in bioremediation to accelerate the decomposition of shipwrecks littering the ocean floor.

Dr. Mary A. Voytek is the director of the National Aeronautics and Space Administration (NASA) Astrobiology Program at NASA Headquarters in Washington, D.C. In 2015, Voytek formed Nexus for Exoplanet System Science (NExSS), a systems science initiative by NASA, to search for life on exoplanets. Voytek came to NASA from the U.S. Geological Survey in Reston, VA, where she headed the USGS Microbiology and Molecular Ecology Laboratory from 1998 to 2009.
Arsenate-reducing bacteria are bacteria which reduce arsenates. Arsenate-reducing bacteria are ubiquitous in arsenic-contaminated groundwater (aqueous environment). Arsenates are salts or esters of arsenic acid (H3AsO4), consisting of the ion AsO43−. They are moderate oxidizers that can be reduced to arsenites and to arsine. Arsenate can serve as a respiratory electron acceptor for oxidation of organic substrates and H2S or H2. Arsenates occur naturally in minerals such as adamite, alarsite, legrandite, and erythrite, and as hydrated or anhydrous arsenates. Arsenates are similar to phosphates since arsenic (As) and phosphorus (P) occur in group 15 (or VA) of the periodic table. Unlike phosphates, arsenates are not readily lost from minerals due to weathering. They are the predominant form of inorganic arsenic in aqueous aerobic environments. On the other hand, arsenite is more common in anaerobic environments, more mobile, and more toxic than arsenate. Arsenite is 25–60 times more toxic and more mobile than arsenate under most environmental conditions. Arsenate can lead to poisoning, since it can replace inorganic phosphate in the glyceraldehyde-3-phosphate --> 1,3-biphosphoglycerate step of glycolysis, producing 1-arseno-3-phosphoglycerate instead. Although glycolysis continues, 1 ATP molecule is lost. Thus, arsenate is toxic due to its ability to uncouple glycolysis. Arsenate can also inhibit pyruvate conversion into acetyl-CoA, thereby blocking the TCA cycle, resulting in additional loss of ATP.

Julia A. Vorholt is a full professor of microbiology at ETH Zurich and an elected member of the German Academy of Sciences Leopoldina.

Rosemary Jeanne Redfield is a microbiologist associated with the University of British Columbia where she worked as a faculty member in the Department of Zoology from 1993 until retiring in 2021.

Crocosphaera watsonii is an isolate of a species of unicellular diazotrophic marine cyanobacteria which represent less than 0.1% of the marine microbial population. They thrive in offshore, open-ocean oligotrophic regions where the waters are warmer than 24 degrees Celsius. Crocosphaera watsonii cell density can exceed 1,000 cells per milliliter within the euphotic zone; however, their growth may be limited by the concentration of phosphorus. Crocosphaera watsonii are able to contribute to the oceanic carbon and nitrogen budgets in tropical oceans due to their size, abundance, and rapid growth rate. Crocosphaera watsonii are unicellular nitrogen fixers that fix atmospheric nitrogen to ammonia during the night and contribute to new nitrogen in the oceans. They are a major source of nitrogen to open-ocean systems. Nitrogen fixation is important in the oceans as it not only allows phytoplankton to continue growing when nitrogen and ammonium are in very low supply but it also replenishes other forms of nitrogen, thus fertilizing the ocean and allowing more phytoplankton growth.

Ronald Oremland was an American microbiologist, astrobiologist, and emeritus senior scientist at the United States Geological Survey. He authored over 200 papers on the microbiology of extreme habitats.
Lulu Qian is a Chinese-American biochemist who is a professor at the California Institute of Technology. Her research uses DNA-like molecules to build artificial machines.