
A placebo is a substance or treatment which is designed to have no therapeutic value. Common placebos include inert tablets, inert injections, sham surgery, and other procedures.

PLOS is a nonprofit publisher of open-access journals in science, technology, and medicine and other scientific literature, under an open-content license. It was founded in 2000 and launched its first journal, PLOS Biology, in October 2003.
Clinical trials are prospective biomedical or behavioral research studies on human participants designed to answer specific questions about biomedical or behavioral interventions, including new treatments and known interventions that warrant further study and comparison. Clinical trials generate data on dosage, safety and efficacy. They are conducted only after they have received health authority/ethics committee approval in the country where approval of the therapy is sought. These authorities are responsible for vetting the risk/benefit ratio of the trial—their approval does not mean the therapy is 'safe' or effective, only that the trial may be conducted.

PLOS Medicine is a peer-reviewed weekly medical journal covering the full spectrum of the medical sciences. It began operation on October 19, 2004, as the second journal of the Public Library of Science (PLOS), a non-profit open access publisher. All content in PLOS Medicine is published under the Creative Commons "by-attribution" license. To fund the journal, the publication's business model requires in most cases that authors pay publication fees. The journal was published online and in a printed format until 2005 and is now only published online. The journal's acting chief editor is Clare Stone, who replaced the previous chief editor, Larry Peiperl, in 2018.
A pilot experiment, pilot study, pilot test or pilot project is a small-scale preliminary study conducted to evaluate feasibility, duration, cost, adverse events, and improve upon the study design prior to performance of a full-scale research project.
An electronic data capture (EDC) system is a computerized system designed for the collection of clinical data in electronic format for use mainly in human clinical trials. EDC replaces the traditional paper-based data collection methodology to streamline data collection and expedite the time to market for drugs and medical devices. EDC solutions are widely adopted by pharmaceutical companies and contract research organizations (CRO).
In medicine an intention-to-treat (ITT) analysis of the results of a randomized controlled trial is based on the initial treatment assignment and not on the treatment eventually received. ITT analysis is intended to avoid various misleading artifacts that can arise in intervention research such as non-random attrition of participants from the study or crossover. ITT is also simpler than other forms of study design and analysis, because it does not require observation of compliance status for units assigned to different treatments or incorporation of compliance into the analysis. Although ITT analysis is widely employed in published clinical trials, it can be incorrectly described and there are some issues with its application. Furthermore, there is no consensus on how to carry out an ITT analysis in the presence of missing outcome data.
Clinical trials are medical research studies conducted on human subjects. The human subjects are assigned to one or more interventions, and the investigators evaluate the effects of those interventions. The progress and results of clinical trials are analyzed statistically.
ClinicalTrials.gov is a registry of clinical trials. It is run by the United States National Library of Medicine (NLM) at the National Institutes of Health, and holds registrations from over 444,000 trials from 221 countries.
A Cytomegalovirus vaccine is a vaccine to prevent cytomegalovirus (CMV) infection or curb virus re-activation in persons already infected. Challenges in developing a vaccine include adeptness of CMV in evading the immune system and limited animal models. As of 2018 no such vaccine exists, although a number of vaccine candidates are under investigation. They include recombinant protein, live attenuated, DNA and other vaccines.

Hookworm vaccine is a vaccine against hookworm. No effective vaccine for the disease in humans has yet been developed. Hookworms, parasitic nematodes transmitted in soil, infect approximately 700 million humans, particularly in tropical regions of the world where endemic hookworms include Ancylostoma duodenale and Necator americanus. Hookworms feed on blood and those infected with hookworms may develop chronic anaemia and malnutrition. Helminth infection can be effectively treated with benzimidazole drugs, and efforts led by the World Health Organization have focused on one to three yearly de-worming doses in schools because hookworm infections with the heaviest intensities are most common in school-age children. However, these drugs only eliminate existing adult parasites and re-infection can occur soon after treatment. School-based de-worming efforts do not treat adults or pre-school children and concerns exist about drug resistance developing in hookworms against the commonly used treatments, thus a vaccine against hookworm disease is sought to provide more permanent resistance to infection.
A pivotal trial is typically a Phase III clinical trial in the multi-year process of clinical research intended to demonstrate and confirm the safety and efficacy of a treatment – such as a drug candidate, medical device or clinical diagnostic procedure – and to estimate the incidence of common adverse effects. A successful pivotal trial is required as evidence for drug marketing approval by the relevant approval authorities, such as the European Medicines Agency, Health Canada or United States Food and Drug Administration (FDA).

AllTrials is a project advocating that clinical research adopt the principles of open research. The project summarizes itself as "All trials registered, all results reported": that is, all clinical trials should be listed in a clinical trials registry, and their results should always be shared as open data.

Sonidegib (INN), sold under the brand name Odomzo, is a medication used to treat cancer.

MIN-117 is an investigational antidepressant which is under development by Minerva Neurosciences for the clinical treatment of major depressive disorder (MDD). It is described as a 5-HT1A and 5-HT2A receptor antagonist and inhibitor of serotonin and dopamine reuptake, and is also reported to possess affinity for the α1A- and α1B-adrenergic receptors. As of May 2015, MIN-117 is in phase II clinical trials for MDD. In December 2019, Minerva announced that MIN-117 was no longer in clinical development for MDD after disappointing results in a phase IIb trial.

Toludesvenlafaxine, also formerly known as ansofaxine and sold under the brand name Ruoxinlin, is an antidepressant which is approved for the treatment of major depressive disorder in China. It is also under development for use in other countries like the United States. It is a serotonin–norepinephrine–dopamine reuptake inhibitor (SNDRI) and was developed by Luye Pharma Group.

Endoxifen, also known as 4-hydroxy-N-desmethyltamoxifen, is a nonsteroidal selective estrogen receptor modulator (SERM) of the triphenylethylene group as well as a protein kinase C (PKC) inhibitor. It is under development for the treatment of estrogen receptor-positive breast cancer and for the treatment of mania in bipolar disorder. It is taken by mouth.

Mericitabine (RG-7128) is an antiviral drug, a deoxycytidine analog. It was developed as a treatment for hepatitis C, acting as a NS5B RNA polymerase inhibitor, but while it showed a good safety profile in clinical trials, it was not sufficiently effective to be used as a stand-alone agent. However mericitabine has been shown to boost the efficacy of other antiviral drugs when used alongside them, and as most modern treatment regimens for hepatitis C use a combination therapy of several antiviral drugs, clinical trials have continued to see if it can form a part of a clinically useful drug treatment program.
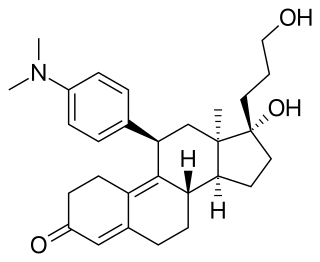
Onapristone (INN) is a synthetic and steroidal antiprogestogen with additional antiglucocorticoid activity which was developed by Schering and described in 1984 but was never marketed. It is a silent antagonist of the progesterone receptor (PR), in contrast to the related antiprogestogen mifepristone. Moreover, compared to mifepristone, onapristone has reduced antiglucocorticoid activity, shows little antiandrogenic activity, and has 10- to 30-fold greater potency as an antiprogestogen. The medication was under development for clinical use, for instance in the treatment of breast cancer and as an endometrial contraceptive, but was discontinued during phase III clinical trials in 1995 due to findings that liver function abnormalities developed in a majority patients.
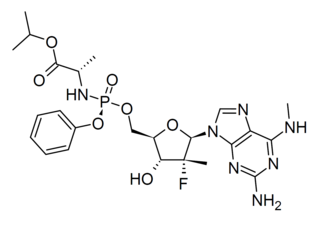
Bemnifosbuvir is an antiviral drug invented by Atea Pharmaceuticals and licensed to Roche for clinical development, a novel nucleotide analog prodrug originally developed for the treatment of hepatitis C. AT-527 is the orally bioavailable hemisulfate salt of AT-511, which is metabolised in several steps to the active nucleotide triphosphate AT-9010, acting as an RNA polymerase inhibitor and thereby interfering with viral replication. AT-527 has been researched for the treatment of coronavirus diseases such as that produced by SARS-CoV-2. It showed good results in early clinical trials but had inconsistent results at later stages, so the planned Phase 3 trials are being redesigned and results are not expected until late 2022.












