Related Research Articles
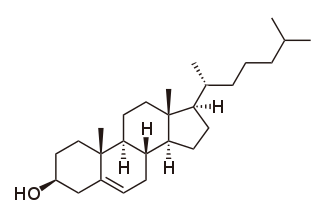
Cholesterol is the principal sterol of all higher animals, distributed in body tissues, especially the brain and spinal cord, and in animal fats and oils.

Abetalipoproteinemia is a disorder that interferes with the normal absorption of fat and fat-soluble vitamins from food. It is caused by a mutation in microsomal triglyceride transfer protein resulting in deficiencies in the apolipoproteins B-48 and B-100, which are used in the synthesis and exportation of chylomicrons and VLDL respectively. It is not to be confused with familial dysbetalipoproteinemia.

A lipoprotein is a biochemical assembly whose primary function is to transport hydrophobic lipid molecules in water, as in blood plasma or other extracellular fluids. They consist of a triglyceride and cholesterol center, surrounded by a phospholipid outer shell, with the hydrophilic portions oriented outward toward the surrounding water and lipophilic portions oriented inward toward the lipid center. A special kind of protein, called apolipoprotein, is embedded in the outer shell, both stabilising the complex and giving it a functional identity that determines its role.
Juvenile hormones (JHs) are a group of acyclic sesquiterpenoids that regulate many aspects of insect physiology. The first discovery of a JH was by Vincent Wigglesworth. JHs regulate development, reproduction, diapause, and polyphenisms. The chemical formula for juvenile hormone is .
Very-low-density lipoprotein (VLDL), density relative to extracellular water, is a type of lipoprotein made by the liver. VLDL is one of the five major groups of lipoproteins that enable fats and cholesterol to move within the water-based solution of the bloodstream. VLDL is assembled in the liver from triglycerides, cholesterol, and apolipoproteins. VLDL is converted in the bloodstream to low-density lipoprotein (LDL) and intermediate-density lipoprotein (IDL). VLDL particles have a diameter of 30–80 nm. VLDL transports endogenous products, whereas chylomicrons transport exogenous (dietary) products. In the early 2010s both the lipid composition and protein composition of this lipoprotein were characterised in great detail.

Lipoprotein lipase (LPL) (EC 3.1.1.34, systematic name triacylglycerol acylhydrolase (lipoprotein-dependent)) is a member of the lipase gene family, which includes pancreatic lipase, hepatic lipase, and endothelial lipase. It is a water-soluble enzyme that hydrolyzes triglycerides in lipoproteins, such as those found in chylomicrons and very low-density lipoproteins (VLDL), into two free fatty acids and one monoacylglycerol molecule:

Apolipoproteins are proteins that bind lipids to form lipoproteins. They transport lipids in blood, cerebrospinal fluid and lymph.
Hyperlipidemia is abnormally elevated levels of any or all lipids or lipoproteins in the blood. The term hyperlipidemia refers to the laboratory finding itself and is also used as an umbrella term covering any of various acquired or genetic disorders that result in that finding. Hyperlipidemia represents a subset of dyslipidemia and a superset of hypercholesterolemia. Hyperlipidemia is usually chronic and requires ongoing medication to control blood lipid levels.

Apolipoprotein B (ApoB) is a protein that in humans is encoded by the APOB gene. It is commonly used to detect risk of atherosclerotic cardiovascular disease.
Lipid metabolism is the synthesis and degradation of lipids in cells, involving the breakdown and storage of fats for energy and the synthesis of structural and functional lipids, such as those involved in the construction of cell membranes. In animals, these fats are obtained from food and are synthesized by the liver. Lipogenesis is the process of synthesizing these fats. The majority of lipids found in the human body from ingesting food are triglycerides and cholesterol. Other types of lipids found in the body are fatty acids and membrane lipids. Lipid metabolism is often considered as the digestion and absorption process of dietary fat; however, there are two sources of fats that organisms can use to obtain energy: from consumed dietary fats and from stored fat. Vertebrates use both sources of fat to produce energy for organs such as the heart to function. Since lipids are hydrophobic molecules, they need to be solubilized before their metabolism can begin. Lipid metabolism often begins with hydrolysis, which occurs with the help of various enzymes in the digestive system. Lipid metabolism also occurs in plants, though the processes differ in some ways when compared to animals. The second step after the hydrolysis is the absorption of the fatty acids into the epithelial cells of the intestinal wall. In the epithelial cells, fatty acids are packaged and transported to the rest of the body.

Vitellogenesis is the process of yolk protein formation in the oocytes of non mammalian vertebrates during sexual maturation. The term vitellogenesis comes from the Latin vitellus. Yolk proteins, such as Lipovitellin and Phosvitin, provides maturing oocytes with the metabolic energy required for development. Vitellogenins are the precursor proteins that lead to yolk protein accumulation in the oocyte. Estrogen and vitellogenin production have a positive correlation. When estrogen production in the ovary is increased via the activation of the hypothalmo-pituitary axis it leads to heightened vitellogenin production in the liver. Vitellogenin production in the liver is the first step of vitellogenesis. Once Vitellogenins are released into the blood stream where they are then transported to the growing oocyte where they lead to yolk protein production. The transport of vitellogenins into the maturing oocyte is done via receptor mediated endocytosis which is a low-density lipoprotein receptor (LDLR). Yolk is a lipoprotein composed of proteins, phospholipids and neutral fats along with a small amount of glycogen. The yolk is synthesised in the liver of the female parent in soluble form. Through circulation it is transported to the follicle cells that surround the maturing ovum, and is deposited in the form of yolk platelets and granules in the ooplasm. The mitochondria and Golgi complex are said to bring about the conversion of the soluble form of yolk into insoluble granules or platelets.

Apolipoprotein C-III also known as apo-CIII, and apolipoprotein C3, is a protein that in humans is encoded by the APOC3 gene. Apo-CIII is secreted by the liver as well as the small intestine, and is found on triglyceride-rich lipoproteins such as chylomicrons, very low density lipoprotein (VLDL), and remnant cholesterol.

Hepatic lipase (HL), also called hepatic triglyceride lipase (HTGL) or LIPC (for "lipase, hepatic"), is a form of lipase, catalyzing the hydrolysis of triacylglyceride. Hepatic lipase is coded by chromosome 15 and its gene is also often referred to as HTGL or LIPC. Hepatic lipase is expressed mainly in liver cells, known as hepatocytes, and endothelial cells of the liver. The hepatic lipase can either remain attached to the liver or can unbind from the liver endothelial cells and is free to enter the body's circulation system. When bound on the endothelial cells of the liver, it is often found bound to heparan sulfate proteoglycans (HSPG), keeping HL inactive and unable to bind to HDL (high-density lipoprotein) or IDL (intermediate-density lipoprotein). When it is free in the bloodstream, however, it is found associated with HDL to maintain it inactive. This is because the triacylglycerides in HDL serve as a substrate, but the lipoprotein contains proteins around the triacylglycerides that can prevent the triacylglycerides from being broken down by HL.
Blood lipids are lipids in the blood, either free or bound to other molecules. They are mostly transported in a phospholipid capsule, and the type of protein embedded in this outer shell determines the fate of the particle and its influence on metabolism. Examples of these lipids include cholesterol and triglycerides. The concentration of blood lipids depends on intake and excretion from the intestine, and uptake and secretion from cells. Hyperlipidemia is the presence of elevated or abnormal levels of lipids and/or lipoproteins in the blood, and is a major risk factor for cardiovascular disease.

Apolipoprotein A-V is a protein that in humans is encoded by the APOA5 gene on chromosome 11. It is significantly expressed in liver. The protein encoded by this gene is an apolipoprotein and an important determinant of plasma triglyceride levels, a major risk factor for coronary artery disease. It is a component of several lipoprotein fractions including VLDL, HDL, chylomicrons. It is believed that apoA-V affects lipoprotein metabolism by interacting with LDL-R gene family receptors. Considering its association with lipoprotein levels, APOA5 is implicated in metabolic syndrome. The APOA5 gene also contains one of 27 SNPs associated with increased risk of coronary artery disease.

Microsomal triglyceride transfer protein large subunit is a protein that in humans is encoded by the MTTP gene.
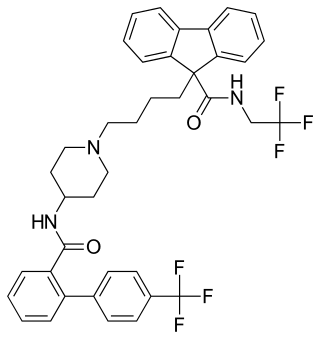
Lomitapide, sold under the brand name Juxtapid in the US and Lojuxta in the EU, is a medication used as a lipid-lowering agent for the treatment of familial hypercholesterolemia, developed by Aegerion Pharmaceuticals. It has been tested in clinical trials as single treatment and in combinations with atorvastatin, ezetimibe and fenofibrate.
Vitellin is a protein found in the egg yolk. It is a phosphoprotein. Vitellin is a generic name for major of many yolk proteins.

Apolipoprotein O also known as protein FAM121B is a protein that in humans is encoded by the APOO gene. APOO is a member of the apolipoprotein family.
In molecular biology, apovitellenin-1 is a family of proteins found in birds. As part of the avian reproductive effort, large quantities of triglyceride-rich very-low-density lipoprotein (VLDL) particles are transported by receptor-mediated endocytosis into the female germ cells, apovitellenin-1 is a protein component of this VLDL. Although the oocytes are surrounded by a layer of granulosa cells harbouring high levels of active lipoprotein lipase, non-lipolysed VLDL is transported into the yolk. This is because the VLDL particles are protected from lipolysis by apovitellenin-1a, which acts as a potent dimeric lipoprotein lipase inhibitor. Apo-VLDL-II is produced in the liver and secreted into the blood stream when induced by estrogen production in female birds.
References
- ↑ "Definition of VITELLOGENIN".
- ↑ Robinson, Richard (18 March 2008). "For Mammals, Loss of Yolk and Gain of Milk Went Hand in Hand". PLOS Biology. 6 (3): e77. doi: 10.1371/journal.pbio.0060077 . PMC 2267822 . PMID 20076706.
- ↑ Tran, Thi Kim Anh; Yu, Richard Man Kit; Islam, Rafiquel; Nguyen, Thi Hong Tham; Bui, Thi Lien Ha; Kong, Richard Yuen Chong; O'Connor, Wayne A.; Leusch, Frederic D.L.; Andrew-Priestley, Megan; MacFarlane, Geoff R. (May 2019). "The utility of vitellogenin as a biomarker of estrogenic endocrine disrupting chemicals in molluscs". Environmental Pollution. 248: 1067–1078. doi:10.1016/j.envpol.2019.02.056. hdl: 10072/386355 . PMID 31091639. S2CID 92464394.
- ↑ Price, E. R. (2017). "The physiology of lipid storage and use in reptiles". Biological Reviews. 92 (3): 1406–1426. doi: 10.1111/brv.12288 . PMID 27348513. S2CID 7570705.
- ↑ Finn, Roderick Nigel (1 June 2007). "Vertebrate Yolk Complexes and the Functional Implications of Phosvitins and Other Subdomains in Vitellogenins1". Biology of Reproduction. 76 (6): 926–35. doi: 10.1095/biolreprod.106.059766 . PMID 17314313.
- ↑ Thompson, James R.; Banaszak, Leonard J. (July 2002). "Lipid−Protein Interactions in Lipovitellin". Biochemistry. 41 (30): 9398–409. doi:10.1021/bi025674w. PMID 12135361.
- ↑ Anderson TA, Levitt DG, Banaszak LJ (July 1998). "The structural basis of lipid interactions in lipovitellin, a soluble lipoprotein". Structure. 6 (7): 895–909. doi: 10.1016/S0969-2126(98)00091-4 . PMID 9687371.
- ↑ Sagiv, Yuval; Bai, Li; Wei, Datsen G.; Agami, Reuven; Savage, Paul B.; Teyton, Luc; Bendelac, Albert (16 April 2007). "A distal effect of microsomal triglyceride transfer protein deficiency on the lysosomal recycling of CD1d". The Journal of Experimental Medicine. 204 (4): 921–8. doi:10.1084/jem.20061568. PMC 2118556 . PMID 17403933.
- ↑ Olofsson SO, Borèn J (November 2005). "Apolipoprotein B: a clinically important apolipoprotein which assembles atherogenic lipoproteins and promotes the development of atherosclerosis". Journal of Internal Medicine. 258 (5): 395–410. doi: 10.1111/j.1365-2796.2005.01556.x . PMID 16238675. S2CID 19885776.
- ↑ Huebbe P, Rimbach G (August 2017). "Evolution of human apolipoprotein E (APOE) isoforms: Gene structure, protein function and interaction with dietary factors". Ageing Research Reviews. 37: 146–161. doi:10.1016/j.arr.2017.06.002. PMID 28647612. S2CID 3758905.
- ↑ Kumar V, Butcher SJ, Öörni K, Engelhardt P, Heikkonen J, et al. (2011) Three-Dimensional cryoEM Reconstruction of Native LDL Particles to 16Å Resolution at Physiological Body Temperature.
- ↑ Oliver, Randy (August 2007). "Fat Bees Part 1". American Bee Journal.[ verification needed ]
- ↑ Randy, Oliver (Aug 2007). "Fat Bees - Part 1". American Bee Journal: 714.
- ↑ Engelmann F (1983). "Vitellogenesis controlled by juvenile hormone". In Downer RG, Laufer H (eds.). Endocrinology of Insects. New York: Alan R. Liss. pp. 259–270.
- ↑ Wyatt GR, Davey KG (1996). "Cellular and Molecular Actions of Juvenile Hormone. II. Roles of Juvenile Hormone in Adult Insects". Cellular and molecular actions of juvenile hormone. II. Roles of juvenile hormones in adult insects. pp. 1–155. doi:10.1016/S0065-2806(08)60030-2. ISBN 9780120242269.
{{cite book}}:|journal=ignored (help) - ↑ Hrassnigg, Norbert; Crailsheim, Karl (2005). "Differences in drone and worker physiology in honeybees (Apis mellifera)" (PDF). Apidologie. 36 (2): 255–277. doi: 10.1051/apido:2005015 .
- ↑ Zeng, Zhijiang; Huang, Zachary Y.; Qin, Yuchuan; Pang, Huizhong (1 April 2005). "Hemolymph Juvenile Hormone Titers in Worker Honey Bees under Normal and Preswarming Conditions". Journal of Economic Entomology. 98 (2): 274–278. doi: 10.1603/0022-0493-98.2.274 . PMID 15889713. S2CID 198130721.
- ↑ Brawand, David; Wahli, Walter; Kaessmann, Henrik; Phillippe, Hervé (18 March 2008). "Loss of Egg Yolk Genes in Mammals and the Origin of Lactation and Placentation". PLOS Biology. 6 (3): e63. doi: 10.1371/journal.pbio.0060063 . PMC 2267819 . PMID 18351802.
- ↑ Yang Zhou, Linda Shearwin-Whyatt, Guojie Zhang et al.: Platypus and echidna genomes reveal mammalian biology and evolution. In: Nature . 6 January 2021. doi:10.1038/s41586-020-03039-0. See also:
- Researchers Sequence Platypus and Echidna Genomes. On: sci-news. 7 January 2021