Sodium laureth sulfate (SLES), an accepted contraction of sodium lauryl ether sulfate (SLES), also called sodium alkylethersulfate, is an anionic detergent and surfactant found in many personal care products and for industrial uses. SLES is an inexpensive and very effective foaming agent. SLES, sodium lauryl sulfate (SLS), ammonium lauryl sulfate (ALS), and sodium pareth sulfate are surfactants that are used in many cosmetic products for their cleaning and emulsifying properties. It is derived from palm kernel oil or coconut oil. In herbicides, it is used as a surfactant to improve absorption of the herbicidal chemicals and reduces time the product takes to be rainfast, when enough of the herbicidal agent will be absorbed.

Contact dermatitis is a type of acute or chronic inflammation of the skin caused by exposure to chemical or physical agents. Symptoms of contact dermatitis can include itchy or dry skin, a red rash, bumps, blisters, or swelling. These rashes are not contagious or life-threatening, but can be very uncomfortable.

In organic chemistry, quaternary ammonium cations, also known as quats, are positively-charged polyatomic ions of the structure [NR4]+, where R is an alkyl group, an aryl group or organyl group. Unlike the ammonium ion and the primary, secondary, or tertiary ammonium cations, the quaternary ammonium cations are permanently charged, independent of the pH of their solution. Quaternary ammonium salts or quaternary ammonium compounds are salts of quaternary ammonium cations. Polyquats are a variety of engineered polymer forms which provide multiple quat molecules within a larger molecule.
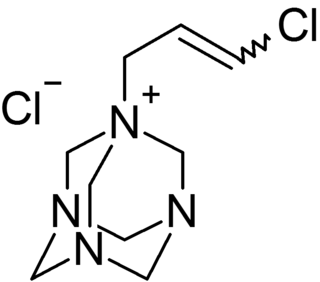
Quaternium-15 is a quaternary ammonium salt that has been used as a surfactant and preservative. It acts as an antimicrobial agent because it slowly releases formaldehyde, which is a preservative with biocidal properties.
Cetyl alcohol, also known as hexadecan-1-ol and palmityl alcohol, is a C-16 fatty alcohol with the formula CH3(CH2)15OH. At room temperature, cetyl alcohol takes the form of a waxy white solid or flakes. The name cetyl derives from the whale oil (cetacea oil, from Latin: cetus, lit. 'whale', from Ancient Greek: κῆτος, romanized: kētos, lit. 'huge fish') from which it was first isolated.
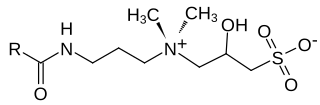
Hydroxysultaines are chemical compounds used in high-foaming shampoos, bath products and shower gels especially in conjunction with ether sulfates and alkyl sulfates. They are also used in industrial applications where high, stable foam is required. Chemically, hydroxysultaines are zwitterionic, typically containing covalently linked positive and negative ions.
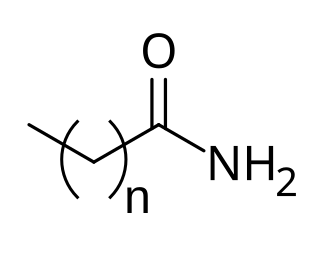
Cocamide is a mixture of amides manufactured from the fatty acids obtained from coconut oil. As coconut oil contains about 50% of lauric acid, in formulas only the 12-carbon chains tend to be considered. Therefore the formula of cocamide can be written as CH3(CH2)10CONH2, though the number of carbon atoms in the chains varies.
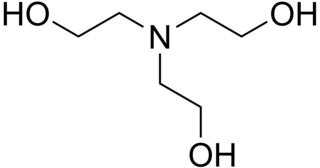
Triethanolamine, or TEOA, is an organic compound with the chemical formula N(CH2CH2OH)3. It is a colourless, viscous liquid. It is both a tertiary amine and a triol. A triol is a molecule with three alcohol groups. Approximately 150,000 tonnes were produced in 1999. It is a colourless compound although samples may appear yellow because of impurities.

Allergic contact dermatitis (ACD) is a form of contact dermatitis that is the manifestation of an allergic response caused by contact with a substance; the other type being irritant contact dermatitis (ICD).

Tetrazepam is a benzodiazepine derivative with anticonvulsant, anxiolytic, muscle relaxant and slightly hypnotic properties. It was formerly used mainly in Austria, France, Belgium, Germany and Spain to treat muscle spasm, anxiety disorders such as panic attacks, or more rarely to treat depression, premenstrual syndrome or agoraphobia. Tetrazepam has relatively little sedative effect at low doses while still producing useful muscle relaxation and anxiety relief. The Co-ordination Group for Mutual Recognition and Decentralised Procedures-Human endorsed the Pharmacovigilance Risk Assessment Committee (PRAC) recommendation to suspend the marketing authorisations of tetrazepam-containing medicines across the European Union (EU) in April 2013. The European Commission has confirmed the suspension of the marketing authorisations for Tetrazepam in Europe because of cutaneous toxicity, effective from the 1 August 2013.

Sodium lauroamphoacetate is a zwitterionic surfactant of the amphoacetate class. It is used as a very mild cleaning agent originally used in shampoos and body washes for infants but it now sees broader use in other personal care products.
Amidoamines are a class of chemical compounds that are formed from fatty acids and amines. They are used as intermediates in the synthesis of surfactants, such as cocamidopropyl betaine (CAPB), some of which are used in personal care products including soaps, shampoos, and cosmetics. Amidoamines can also serve as curing agents for epoxy resins. They are also used as oil well drilling fluids and also as corrosion inhibitors.

Dimethylaminopropylamine (DMAPA) is a diamine used in the preparation of some surfactants, such as cocamidopropyl betaine which is an ingredient in many personal care products including soaps, shampoos, and cosmetics. BASF, a major producer, claims that DMAPA-derivatives do not sting the eyes and makes a fine-bubble foam, making it appropriate in shampoos.
Eyelid dermatitis is commonly related to atopic dermatitis or allergic contact dermatitis. Volatile substances, tosylamide, epoxy hardeners, insect sprays, and lemon peel oil may be implicated, with many cases of eyelid contact dermatitis being caused by substances transferred by the hands to the eyelids.
Allergen of the Year is an annual "award" voted upon by the American Contact Dermatitis Society. The purpose of the award is "to draw attention to the agents causing the most significant clinical effects, those that are underrecognized and those that have become obsolete or for which exposure patterns have changed".
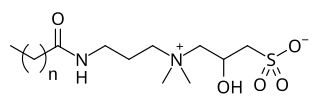
Cocamidopropyl hydroxysultaine (CAHS) is a synthetic amphoteric surfactant from the hydroxysultaine group. It is found in personal care products. It has uses as a foam booster, viscosity builder, and an antistatic agent.

Iodopropynyl Butyl Carbamate (IPBC) is a water-soluble preservative used globally in the paints & coatings, wood preservatives, personal care, and cosmetics industries. IPBC is a member of the carbamate family of biocides. IPBC was invented in the 1970s and has a long history of effective use as an antifungal technology.

Nickel allergy is any of several allergic conditions provoked by exposure to the chemical element nickel. Nickel allergy often takes the form of nickel allergic contact dermatitis (Ni-ACD), a form of allergic contact dermatitis (ACD). Ni-ACD typically causes a rash that is red and itchy and that may be bumpy or scaly. The main treatment for it is avoiding contact with nickel-releasing metals, such as inexpensive jewelry. Another form of nickel allergy is a systemic form: systemic nickel allergy syndrome (SNAS) can mimic some of the symptoms of irritable bowel syndrome (IBS) and also has a dermatologic component.
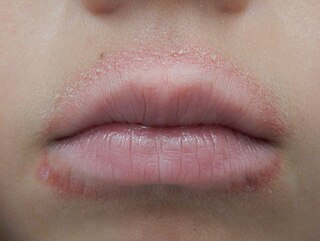
Lip licker's dermatitis is a type of skin inflammation around the lips due to damage by saliva from repetitive lip licking and is classified as a subtype of irritant contact cheilitis. The resulting scaling, redness, chapping, and crusting makes a well-defined ring around the lips. The rash may extend as far as the tongue can reach and usually does not occur at the corners of the mouth. It commonly occurs during winter months but some people can have it year-round if lip licking is a chronic habit.

Metal allergies inflame the skin after it has been in contact with metal. They are a form of allergic contact dermatitis. They are becoming more common, as of 2021, except in areas with regulatory countermeasures.
 [1]
[1] 












