
Erbium is a chemical element; it has symbol Er and atomic number 68. A silvery-white solid metal when artificially isolated, natural erbium is always found in chemical combination with other elements. It is a lanthanide, a rare-earth element, originally found in the gadolinite mine in Ytterby, Sweden, which is the source of the element's name.

Holmium is a chemical element; it has symbol Ho and atomic number 67. It is a rare-earth element and the eleventh member of the lanthanide series. It is a relatively soft, silvery, fairly corrosion-resistant and malleable metal. Like many other lanthanides, holmium is too reactive to be found in native form, as pure holmium slowly forms a yellowish oxide coating when exposed to air. When isolated, holmium is relatively stable in dry air at room temperature. However, it reacts with water and corrodes readily, and also burns in air when heated.
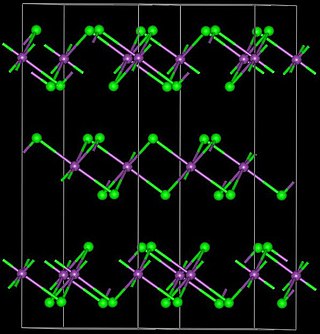
Yttrium(III) bromide is an inorganic compound with the chemical formula YBr3. It is a white solid. Anhydrous yttrium(III) bromide can be produced by reacting yttrium oxide or yttrium(III) bromide hydrate and ammonium bromide. The reaction proceeds via the intermediate (NH4)3YBr6. Another method is to react yttrium carbide (YC2) and elemental bromine. Yttrium(III) bromide can be reduced by yttrium metal to YBr or Y2Br3. It can react with osmium to produce Y4Br4Os.

Terbium(III) bromide (TbBr3) is a crystalline chemical compound.

Yttrium arsenide is an inorganic compound of yttrium and arsenic with the chemical formula YAs. It can be prepared by reacting yttrium and arsenic at high temperature. Some literature has done research on the eutectic system of it and zinc arsenide.
Tellurium compounds are compounds containing the element tellurium (Te). Tellurium belongs to the chalcogen family of elements on the periodic table, which also includes oxygen, sulfur, selenium and polonium: Tellurium and selenium compounds are similar. Tellurium exhibits the oxidation states −2, +2, +4 and +6, with +4 being most common.
Praseodymium(III) fluoride is an inorganic compound with the formula PrF3, being the most stable fluoride of praseodymium.
Thullium(III) fluoride is an inorganic compound with the chemical formula TmF3.
Erbium(III) fluoride is the fluoride of erbium, a rare earth metal, with the chemical formula ErF3. It can be used to make infrared light-transmitting materials and up-converting luminescent materials.

Neodymium tantalate is an inorganic compound with the chemical formula NdTaO4. It is prepared by reacting neodymium oxide and tantalum pentoxide at 1200 °C. It reacts with a mixture of tantalum pentoxide and chlorine gas at high temperature to obtain Nd2Ta2O7Cl2. It is ammonolyzed at high temperature to obtain oxynitrides of Nd-Ta.

Europium(III) acetate is an inorganic salt of europium and acetic acid with the chemical formula of Eu(CH3COO)3. In this compound, europium exhibits the +3 oxidation state. It can exist in the anhydrous form, sesquihydrate and tetrahydrate. Its hydrate molecule is a dimer.
Europium(III) chromate is a chemical compound composed of europium, chromium and oxygen with europium in the +3 oxidation state, chromium in the +5 oxidation state and oxygen in the −2 oxidation state. It has the chemical formula of EuCrO4.

Europium compounds are compounds formed by the lanthanide metal europium (Eu). In these compounds, europium generally exhibits the +3 oxidation state, such as EuCl3, Eu(NO3)3 and Eu(CH3COO)3. Compounds with europium in the +2 oxidation state are also known. The +2 ion of europium is the most stable divalent ion of lanthanide metals in aqueous solution. Many europium compounds fluoresce under ultraviolet light due to the excitation of electrons to higher energy levels. Lipophilic europium complexes often feature acetylacetonate-like ligands, e.g., Eufod.

Dysprosium(III) bromide is an inorganic compound of bromine and dysprosium, with the chemical formula of DyBr3.

Gadolinium diiodide is an inorganic compound, with the chemical formula of GdI2. It is an electride, with the ionic formula of Gd3+(I−)2e−, and therefore not a true gadolinium(II) compound. It is ferromagnetic at 276 K with a saturation magnetization of 7.3 B; it exhibits a large negative magnetoresistance (~70%) at 7 T near room temperature. It can be obtained by reacting gadolinium and gadolinium(III) iodide at a high temperature:
Erbium compounds are compounds containing the element erbium (Er). These compounds are usually dominated by erbium in the +3 oxidation state, although the +2, +1 and 0 oxidation states have also been reported.
Lutetium compounds are compounds formed by the lanthanide metal lutetium (Lu). In these compounds, lutetium generally exhibits the +3 oxidation state, such as LuCl3, Lu2O3 and Lu2(SO4)3. Aqueous solutions of most lutetium salts are colorless and form white crystalline solids upon drying, with the common exception of the iodide. The soluble salts, such as nitrate, sulfate and acetate form hydrates upon crystallization. The oxide, hydroxide, fluoride, carbonate, phosphate and oxalate are insoluble in water.
Erbium(III) selenide is an inorganic compound with a chemical formula of Er2Se3.

Erbium(III) selenate is an inorganic compound, with the chemical formula Er2(SeO4)3. It exists as an anhydrate or an octahydrate.
Ytterbium monotelluride is an inorganic compound, one of the tellurides of ytterbium, with the chemical formula YbTe.









