Related Research Articles
In chemistry, a heteroatom is, strictly, any atom that is not carbon or hydrogen.

A heterocyclic compound or ring structure is a cyclic compound that has atoms of at least two different elements as members of its ring(s). Heterocyclic organic chemistry is the branch of organic chemistry dealing with the synthesis, properties, and applications of organic heterocycles.

In chemistry, a double bond is a covalent bond between two atoms involving four bonding electrons as opposed to two in a single bond. Double bonds occur most commonly between two carbon atoms, for example in alkenes. Many double bonds exist between two different elements: for example, in a carbonyl group between a carbon atom and an oxygen atom. Other common double bonds are found in azo compounds (N=N), imines (C=N), and sulfoxides (S=O). In a skeletal formula, a double bond is drawn as two parallel lines (=) between the two connected atoms; typographically, the equals sign is used for this. Double bonds were introduced in chemical notation by Russian chemist Alexander Butlerov.

In chemistry, catenation is the bonding of atoms of the same element into a series, called a chain. A chain or a ring shape may be open if its ends are not bonded to each other, or closed if they are bonded in a ring. The words to catenate and catenation reflect the Latin root catena, "chain".
The skeletal formula, line-angle formula, or shorthand formula of an organic compound is a type of molecular structural formula that serves as a shorthand representation of a molecule's bonding and some details of its molecular geometry. A skeletal formula shows the skeletal structure or skeleton of a molecule, which is composed of the skeletal atoms that make up the molecule. It is represented in two dimensions, as on a piece of paper. It employs certain conventions to represent carbon and hydrogen atoms, which are the most common in organic chemistry.
In organic chemistry, a carbene is a molecule containing a neutral carbon atom with a valence of two and two unshared valence electrons. The general formula is R−:C−R' or R=C: where the R represents substituents or hydrogen atoms.
Organosulfur chemistry is the study of the properties and synthesis of organosulfur compounds, which are organic compounds that contain sulfur. They are often associated with foul odors, but many of the sweetest compounds known are organosulfur derivatives, e.g., saccharin. Nature is abound with organosulfur compounds—sulfur is vital for life. Of the 20 common amino acids, two are organosulfur compounds, and the antibiotics penicillin and sulfa drugs both contain sulfur. While sulfur-containing antibiotics save many lives, sulfur mustard is a deadly chemical warfare agent. Fossil fuels, coal, petroleum, and natural gas, which are derived from ancient organisms, necessarily contain organosulfur compounds, the removal of which is a major focus of oil refineries.

A polyyne is any organic compound with alternating single and triple bonds; that is, a series of consecutive alkynes, (−C≡C−)n with n greater than 1. These compounds are also called polyacetylenes, especially in the natural products and chemical ecology literature, even though this nomenclature more properly refers to acetylene polymers composed of alternating single and double bonds (−CR=CR′−)n with n greater than 1. They are also sometimes referred to as oligoynes, or carbinoids after "carbyne" (−C≡C−)∞, the hypothetical allotrope of carbon that would be the ultimate member of the series. The synthesis of this substance has been claimed several times since the 1960s, but those reports have been disputed. Indeed, the substances identified as short chains of "carbyne" in many early organic synthesis attempts would be called polyynes today.
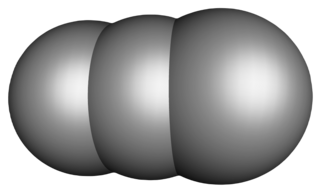
Tricarbon is an inorganic compound with the chemical formula C
2(μ-C). It is a colourless gas that only persists in dilution or solution as an adduct. It is one of the simplest unsaturated carbenes. Tricarbon can be found in interstellar space and can be produced in the laboratory by a process called laser ablation.
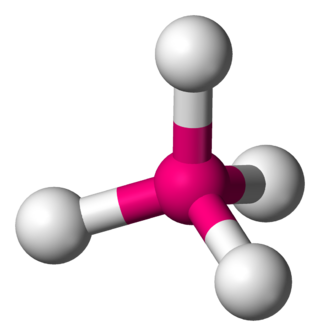
In a tetrahedral molecular geometry, a central atom is located at the center with four substituents that are located at the corners of a tetrahedron. The bond angles are cos−1(−1⁄3) = 109.4712206...° ≈ 109.5° when all four substituents are the same, as in methane as well as its heavier analogues. Methane and other perfectly symmetrical tetrahedral molecules belong to point group Td, but most tetrahedral molecules have lower symmetry. Tetrahedral molecules can be chiral.

Dicarbon monoxide is a molecule that contains two carbon atoms and one oxygen atom. It is a linear molecule that, because of its simplicity, is of interest in a variety of areas. It is, however, so extremely reactive that it is not encountered in everyday life. It is classified as a carbene, cumulene and an oxocarbon.

Linear acetylenic carbon (LAC), also known as carbyne or Linear Carbon Chain (LCC), is an allotrope of carbon that has the chemical structure (−C≡C−)n as a repeat unit, with alternating single and triple bonds. It would thus be the ultimate member of the polyyne family.

In chemistry, an oxocarbon or oxide of carbon is a chemical compound consisting only of carbon and oxygen. The simplest and most common oxocarbons are carbon monoxide (CO) and carbon dioxide. Many other stable or metastable oxides of carbon are known, but they are rarely encountered, such as carbon suboxide and mellitic anhydride.

Disulfur dioxide, dimeric sulfur monoxide or SO dimer is an oxide of sulfur with the formula S2O2. The solid is unstable with a lifetime of a few seconds at room temperature.
Hydrogen chalcogenides are binary compounds of hydrogen with chalcogen atoms. Water, the first chemical compound in this series, contains one oxygen atom and two hydrogen atoms, and is the most common compound on the Earth's surface.

A cubane-type cluster is an arrangement of atoms in a molecular structure that forms a cube. In the idealized case, the eight vertices are symmetry equivalent and the species has Oh symmetry. Such a structure is illustrated by the hydrocarbon cubane. With chemical formula C8H8, cubane has carbon atoms at the corners of a cube and covalent bonds forming the edges. Most cubanes have more complicated structures, usually with nonequivalent vertices. They may be simple covalent compounds or macromolecular or supramolecular cluster compounds.

Tricarbon monoxide C3O is a reactive radical oxocarbon molecule found in space, and which can be made as a transient substance in the laboratory. It can be trapped in an inert gas matrix or made as a short lived gas. C3O can be classified as a ketene or an oxocumulene a kind of heterocumulene.

Sulfoxylic acid (H2SO2) (also known as hyposulfurous acid or sulfur dihydroxide) is an unstable oxoacid of sulfur in an intermediate oxidation state between hydrogen sulfide and dithionous acid. It consists of two hydroxy groups attached to a sulfur atom. Sulfoxylic acid contains sulfur in an oxidation state of +2. Sulfur monoxide (SO) can be considered as a theoretical anhydride for sulfoxylic acid, but it is not actually known to react with water.
Tricarbon monosulfide (C3S) or tricarbon sulfur is a reactive molecular substance that has been detected in outer space. Tricarbon monosulfide is a heterocumulene or thiocumulene, consisting of a straight chain of three carbon atoms and a terminal sulfur atom.
Ethenedithione or ethylenedithone is an unstable chemical substance with formula S=C=C=S made from carbon and sulfur.
References
- ↑ IUPAC , Compendium of Chemical Terminology , 2nd ed. (the "Gold Book") (1997). Online corrected version: (2006–) " heterocumulenes ". doi : 10.1351/goldbook.H02797
- ↑ Kumar, Akshai; Samuelson, Ashoka G. (January 2011). "Metathesis of carbon dioxide and phenyl isocyanate catalysed by group(IV) metal alkoxides: An experimental and computational study" (PDF). Journal of Chemical Sciences. 123 (1): 29–36. doi: 10.1007/s12039-011-0069-4 .
- 1 2 3 Wang, Tianfang; Bowie, John H. (November 2011). "Studies of cyclization reactions of linear cumulenes and heterocumulenes using the neutralization-reionization procedure and/or ab initio calculations". Mass Spectrometry Reviews. 30 (6): 1225–1241. Bibcode:2011MSRv...30.1225W. doi:10.1002/mas.20328. PMID 21400561.
- ↑ Yamamoto, Satoshi; Saito, Shuji; Kawaguchi, Kentarou; Kaifu, Norio; Suzuki, Hiroko (June 1987). "Laboratory detection of a new carbon-chain molecule C3S and its astronomical identification". The Astrophysical Journal. 317: L119. Bibcode:1987ApJ...317L.119Y. doi:10.1086/184924.
- ↑ Burnin, Andrei; BelBruno, Joseph J. (November 2003). "SCnS Linear Chain Production by Direct Laser Ablation". The Journal of Physical Chemistry A. 107 (45): 9547–9553. Bibcode:2003JPCA..107.9547B. doi:10.1021/jp0304071.
- 1 2 3 4 5 6 Botschwina, Peter (2003). "Spectroscopic properties of interstellar molecules: Theory and experiment". Physical Chemistry Chemical Physics. 5 (16): 3337. Bibcode:2003PCCP....5.3337B. doi:10.1039/b303753n.
- 1 2 3 4 Ohshima, Yasuhiro; Endo, Yasuki; Ogata, Teruhiko (22 January 1995). "Fourier‐transform microwave spectroscopy of triplet carbon monoxides, C2O, C4O, C6O, and C8O". The Journal of Chemical Physics. 102 (4): 1493–1500. Bibcode:1995JChPh.102.1493O. doi:10.1063/1.468881.
- 1 2 3 4 Szczepanski, Jan; Hodyss, Robert; Fuller, Jason; Vala, Martin (April 1999). "Infrared Absorption Spectroscopy of Small Carbon−Sulfur Clusters Isolated in Solid Ar". The Journal of Physical Chemistry A. 103 (16): 2975–2981. Bibcode:1999JPCA..103.2975S. doi:10.1021/jp984700q.
- ↑ Thorwirth, S.; Salomon, T.; Fanghänel, S.; Kozubal, J.R.; Dudek, J.B. (September 2017). "High-resolution infrared fingerprints of carbon-sulfur clusters: The ν1 band of C5S". Chemical Physics Letters. 684: 262–266. Bibcode:2017CPL...684..262T. doi:10.1016/j.cplett.2017.06.032.
- 1 2 3 Wang, Haiyan; Szczepanski, Jan; Cooke, Andrew; Brucat, Philip; Vala, Martin (2005). "Vibrational absorption spectra of CnS (n = 2, 6) and CnS2 (n = 7, 9, 11, 13, 15) linear carbon-sulfur clusters". International Journal of Quantum Chemistry. 102 (5): 806–819. Bibcode:2005IJQC..102..806W. doi:10.1002/qua.20383.
- ↑ Pu, Liang; Zhao, Xiao; Zhang, Zhong; King, R. Bruce (May 2017). "Heavier Carbon Subchalcogenides as C3 Sources for Tungsten-Capped Cumulenes: A Theoretical Study". Inorganic Chemistry. 56 (10): 5567–5576. doi:10.1021/acs.inorgchem.6b02958. PMID 28459557.
- 1 2 3 Liu, Xuegang; Li, Gang; Liu, Zhiling; Yang, Wenshao; Fan, Hongjun; Jiang, Ling; Xie, Hua (2021-12-23). "Isoelectronic IrC 3 – , PtC 3 , and AuC 3 + Clusters Featuring the Structural and Bonding Resemblance to OC 3". The Journal of Physical Chemistry Letters. 13 (1): 12–17. doi:10.1021/acs.jpclett.1c03754. ISSN 1948-7185. PMID 34941270. S2CID 245444740.
- 1 2 McCarthy, M. C.; Cooksy, A. L.; Mohamed, S.; Gordon, V. D.; Thaddeus, P. (February 2003). "Rotational Spectra of the Nitrogen‐Sulfur Carbon Chains NCnS, n = 1–7" (PDF). The Astrophysical Journal Supplement Series. 144 (2): 287–297. Bibcode:2003ApJS..144..287M. doi:10.1086/344727. hdl: 2152/26169 . S2CID 122233232.