
Hypoxia is a condition in which the body or a region of the body is deprived of adequate oxygen supply at the tissue level. Hypoxia may be classified as either generalized, affecting the whole body, or local, affecting a region of the body. Although hypoxia is often a pathological condition, variations in arterial oxygen concentrations can be part of the normal physiology, for example, during strenuous physical exercise.

The diving reflex, also known as the diving response and mammalian diving reflex, is a set of physiological responses to immersion that overrides the basic homeostatic reflexes, and is found in all air-breathing vertebrates studied to date. It optimizes respiration by preferentially distributing oxygen stores to the heart and brain, enabling submersion for an extended time.
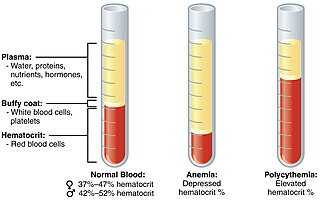
Polycythemia is a laboratory finding in which the hematocrit and/or hemoglobin concentration are increased in the blood. Polycythemia is sometimes called erythrocytosis, and there is significant overlap in the two findings, but the terms are not the same: polycythemia describes any increase in hematocrit and/or hemoglobin, while erythrocytosis describes an increase specifically in the number of red blood cells in the blood.

Altitude training is the practice by some endurance athletes of training for several weeks at high altitude, preferably over 2,400 metres (8,000 ft) above sea level, though more commonly at intermediate altitudes due to the shortage of suitable high-altitude locations. At intermediate altitudes, the air still contains approximately 20.9% oxygen, but the barometric pressure and thus the partial pressure of oxygen is reduced.

Glomus cells are the cell type mainly located in the carotid bodies and aortic bodies. Glomus type I cells are peripheral chemoreceptors which sense the oxygen, carbon dioxide and pH levels of the blood. When there is a decrease in the blood's pH, a decrease in oxygen (pO2), or an increase in carbon dioxide (pCO2), the carotid bodies and the aortic bodies signal the dorsal respiratory group in the medulla oblongata to increase the volume and rate of breathing. The glomus cells have a high metabolic rate and good blood perfusion and thus are sensitive to changes in arterial blood gas tension. Glomus type II cells are sustentacular cells having a similar supportive function to glial cells.
Hypoxic pulmonary vasoconstriction (HPV), also known as the Euler-Liljestrand mechanism, is a physiological phenomenon in which small pulmonary arteries constrict in the presence of alveolar hypoxia. By redirecting blood flow from poorly-ventilated lung regions to well-ventilated lung regions, HPV is thought to be the primary mechanism underlying ventilation/perfusion matching.
Hypoxia-inducible factors (HIFs) are transcription factors that respond to decreases in available oxygen in the cellular environment, or hypoxia.

Hypoxemia is an abnormally low level of oxygen in the blood. More specifically, it is oxygen deficiency in arterial blood. Hypoxemia has many causes, and often causes hypoxia as the blood is not supplying enough oxygen to the tissues of the body.
Chronic mountain sickness (CMS) is a disease in which the proportion of blood volume that is occupied by red blood cells increases (polycythaemia) and there is an abnormally low level of oxygen in the blood (hypoxemia). CMS typically develops after extended time living at high altitude. It is most common amongst native populations of high altitude nations. The most frequent symptoms of CMS are headache, dizziness, tinnitus, breathlessness, palpitations, sleep disturbance, fatigue, loss of appetite, confusion, cyanosis, and dilation of veins.
Peripheral chemoreceptors are so named because they are sensory extensions of the peripheral nervous system into blood vessels where they detect changes in chemical concentrations. As transducers of patterns of variability in the surrounding environment, carotid and aortic bodies count as chemosensors in a similar way as taste buds and photoreceptors. However, because carotid and aortic bodies detect variation within the body's internal organs, they are considered interoceptors. Taste buds, olfactory bulbs, photoreceptors, and other receptors associated with the five traditional sensory modalities, by contrast, are exteroceptors in that they respond to stimuli outside the body. The body also contains proprioceptors, which respond to the amount of stretch within the organ, usually muscle, that they occupy.

The effects of high altitude on humans are mostly the consequences of reduced partial pressure of oxygen in the atmosphere. The medical problems that are direct consequence of high altitude are caused by the low inspired partial pressure of oxygen, which is caused by the reduced atmospheric pressure, and the constant gas fraction of oxygen in atmospheric air over the range in which humans can survive. The other major effect of altitude is due to lower ambient temperature.

Hypoxia-inducible factor 1-alpha, also known as HIF-1-alpha, is a subunit of a heterodimeric transcription factor hypoxia-inducible factor 1 (HIF-1) that is encoded by the HIF1A gene. The Nobel Prize in Physiology or Medicine 2019 was awarded for the discovery of HIF.
Fabiola León-Velarde Servetto is a Peruvian physiologist who has devoted her research to the biology and physiology of high altitude adaptation. Born in Lima, Peru. She is the daughter of Carlos Leon-Velarde Gamarra and Juana Servetto Marti from Uruguay, and granddaughter of Angelica Gamarra. Under the mentorship of high altitude physiologist Carlos Monge Cassinelli, she obtained a BSc. in Biology (1979), an MSc (1981) and DSc (1986) in physiology at Cayetano Heredia University in Lima, Perú.
Endothelial PAS domain-containing protein 1 is a protein that is encoded by the EPAS1 gene in mammals. It is a type of hypoxia-inducible factor, a group of transcription factors involved in the physiological response to oxygen concentration. The gene is active under hypoxic conditions. It is also important in the development of the heart, and for maintaining the catecholamine balance required for protection of the heart. Mutation often leads to neuroendocrine tumors.
A hypoxicator is a medical device intended to provide a stimulus for the adaptation of an individual's cardiovascular system by means of breathing reduced oxygen hypoxic air and triggering mechanisms of compensation. The aim of intermittent hypoxic training or hypoxic therapy conducted with such a device is to obtain benefits in physical performance and wellbeing through improved oxygen metabolism.

Organisms can live at high altitude, either on land, in water, or while flying. Decreased oxygen availability and decreased temperature make life at such altitudes challenging, though many species have been successfully adapted via considerable physiological changes. As opposed to short-term acclimatisation, high-altitude adaptation means irreversible, evolved physiological responses to high-altitude environments, associated with heritable behavioural and genetic changes. Among vertebrates, only few mammals and certain birds are known to have completely adapted to high-altitude environments.
Fish are exposed to large oxygen fluctuations in their aquatic environment since the inherent properties of water can result in marked spatial and temporal differences in the concentration of oxygen. Fish respond to hypoxia with varied behavioral, physiological, and cellular responses to maintain homeostasis and organism function in an oxygen-depleted environment. The biggest challenge fish face when exposed to low oxygen conditions is maintaining metabolic energy balance, as 95% of the oxygen consumed by fish is used for ATP production releasing the chemical energy of nutrients through the mitochondrial electron transport chain. Therefore, hypoxia survival requires a coordinated response to secure more oxygen from the depleted environment and counteract the metabolic consequences of decreased ATP production at the mitochondria.
High-altitude adaptation in humans is an instance of evolutionary modification in certain human populations, including those of Tibet in Asia, the Andes of the Americas, and Ethiopia in Africa, who have acquired the ability to survive at altitudes above 2,500 meters. This adaptation means irreversible, long-term physiological responses to high-altitude environments, associated with heritable behavioural and genetic changes. While the rest of the human population would suffer serious health consequences, the indigenous inhabitants of these regions thrive well in the highest parts of the world. These humans have undergone extensive physiological and genetic changes, particularly in the regulatory systems of oxygen respiration and blood circulation, when compared to the general lowland population.
Cynthia Beall is an American physical anthropologist at the Case Western Reserve University, Cleveland, Ohio. Four decades of her research on people living in extremely high mountains became the frontier in understanding human evolution and high-altitude adaptation. Her groundbreaking works among the Andean, Tibetan and East African highlanders are the basis of our knowledge on adaptation to hypoxic condition and how it influences the evolutionary selection in modern humans. She is currently the Distinguished University Professor, and member of the U.S. National Academy of Sciences and the American Philosophical Society.
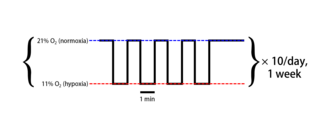
Intermittent hypoxia (also known as episodic hypoxia) is an intervention in which a person or animal undergoes alternating periods of normoxia and hypoxia. Normoxia is defined as exposure to oxygen levels normally found in Earth's atmosphere (~21% O2) and hypoxia as any oxygen levels lower than those of normoxia. Normally, exposure to hypoxia is negatively associated to physiological changes to the body, such as altitude sickness. However, when used in moderation, intermittent hypoxia may be used clinically as a means to alleviate various pathological conditions.














