Related Research Articles

Europium is a chemical element; it has symbol Eu and atomic number 63. Europium is a silvery-white metal of the lanthanide series that reacts readily with air to form a dark oxide coating. It is the most chemically reactive, least dense, and softest of the lanthanide elements. It is soft enough to be cut with a knife. Europium was isolated in 1901 and named after the continent of Europe. Europium usually assumes the oxidation state +3, like other members of the lanthanide series, but compounds having oxidation state +2 are also common. All europium compounds with oxidation state +2 are slightly reducing. Europium has no significant biological role and is relatively non-toxic compared to other heavy metals. Most applications of europium exploit the phosphorescence of europium compounds. Europium is one of the rarest of the rare-earth elements on Earth.
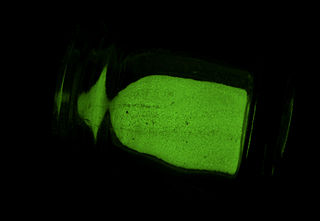
A phosphor is a substance that exhibits the phenomenon of luminescence; it emits light when exposed to some type of radiant energy. The term is used both for fluorescent or phosphorescent substances which glow on exposure to ultraviolet or visible light, and cathodoluminescent substances which glow when struck by an electron beam in a cathode-ray tube.

Pyrochlore2Nb2O6(OH,F) is a mineral group of the niobium end member of the pyrochlore supergroup. Pyrochlore is also a term for the crystal structure Fd3m. The name is from the Greek πῦρ, fire, and χλωρός, green because it typically turns green on ignition in classic blowpipe analysis.

Cerium(IV) oxide, also known as ceric oxide, ceric dioxide, ceria, cerium oxide or cerium dioxide, is an oxide of the rare-earth metal cerium. It is a pale yellow-white powder with the chemical formula CeO2. It is an important commercial product and an intermediate in the purification of the element from the ores. The distinctive property of this material is its reversible conversion to a non-stoichiometric oxide.

A ferrite is an iron oxide-containing magnetic ceramic material. They are ferrimagnetic, meaning they are attracted by magnetic fields and can be magnetized to become permanent magnets. Unlike many ferromagnetic materials, most ferrites are not electrically conductive, making them useful in applications like magnetic cores for transformers to suppress eddy currents.
Phosphor thermometry is an optical method for surface temperature measurement. The method exploits luminescence emitted by phosphor material. Phosphors are fine white or pastel-colored inorganic powders which may be stimulated by any of a variety of means to luminesce, i.e. emit light. Certain characteristics of the emitted light change with temperature, including brightness, color, and afterglow duration. The latter is most commonly used for temperature measurement.

Hafnium(IV) chloride is the inorganic compound with the formula HfCl4. This colourless solid is the precursor to most hafnium organometallic compounds. It has a variety of highly specialized applications, mainly in materials science and as a catalyst.

Strontium aluminate is an aluminate compound with the chemical formula SrAl2O4. It is a pale yellow, monoclinic crystalline powder that is odourless and non-flammable. When activated with a suitable dopant, it acts as a photoluminescent phosphor with long persistence of phosphorescence.

Hafnium carbide (HfC) is a chemical compound of hafnium and carbon. Previously the material was estimated to have a melting point of about 3,900 °C. More recent tests have been able to conclusively prove that the substance has an even higher melting point of 3,958 °C exceeding those of tantalum carbide and tantalum hafnium carbide which were both previously estimated to be higher. However, it has a low oxidation resistance, with the oxidation starting at temperatures as low as 430 °C. Experimental testing in 2018 confirmed the higher melting point yielding a result of 3,982 (±30°C) with a small possibility that the melting point may even exceed 4,000°C.

Hafnium(IV) oxide is the inorganic compound with the formula HfO
2. Also known as hafnium dioxide or hafnia, this colourless solid is one of the most common and stable compounds of hafnium. It is an electrical insulator with a band gap of 5.3~5.7 eV. Hafnium dioxide is an intermediate in some processes that give hafnium metal.
Tantalum hafnium carbide is a refractory chemical compound with a general formula TaxHfyCx+y, which can be considered as a solid solution of tantalum carbide and hafnium carbide. It was originally thought to have the highest melting of any known substance but new research has proven that hafnium carbonitride has a higher melting point.

Zirconium diboride (ZrB2) is a highly covalent refractory ceramic material with a hexagonal crystal structure. ZrB2 is an ultra-high temperature ceramic (UHTC) with a melting point of 3246 °C. This along with its relatively low density of ~6.09 g/cm3 (measured density may be higher due to hafnium impurities) and good high temperature strength makes it a candidate for high temperature aerospace applications such as hypersonic flight or rocket propulsion systems. It is an unusual ceramic, having relatively high thermal and electrical conductivities, properties it shares with isostructural titanium diboride and hafnium diboride.

Lutetium tantalate is a chemical compound of lutetium, tantalum and oxygen with the formula LuTaO4. With a density of 9.81 g/cm3, this salt is the densest known white stable material. (Although thorium dioxide ThO2 is also white and has a higher density of 10 g/cm3, it is radioactively unstable; while not radioactive enough to make it unstable as a material, even its low rate of decay is still too much for certain uses such as phosphors for detecting ionising radiation.) The white color and high density of LuTaO4 make it ideal for phosphor applications, though the high cost of lutetium is a hindrance.
Ultra-high-temperature ceramics (UHTCs) are a type of refractory ceramics that can withstand extremely high temperatures without degrading, often above 2,000 °C. They also often have high thermal conductivities and are highly resistant to thermal shock, meaning they can withstand sudden and extreme changes in temperature without cracking or breaking. Chemically, they are usually borides, carbides, nitrides, and oxides of early transition metals.

Europium acetylacetonate is a coordination complex with formula Eu(C5H7O2)3. Although this anhydrous acetylacetonate complex is widely discussed, some sources suggest that it is really the dihydrate Eu(C5H7O2)3(H2O)2.
Lanthanum manganite is an inorganic compound with the formula LaMnO3, often abbreviated as LMO. Lanthanum manganite is formed in the perovskite structure, consisting of oxygen octahedra with a central Mn atom. The cubic perovskite structure is distorted into an orthorhombic structure by a strong Jahn–Teller distortion of the oxygen octahedra.

Hafnium disulfide is an inorganic compound of hafnium and sulfur. It is a layered dichalcogenide with the chemical formula is HfS2. A few atomic layers of this material can be exfoliated using the standard Scotch Tape technique (see graphene) and used for the fabrication of a field-effect transistor. High-yield synthesis of HfS2 has also been demonstrated using liquid phase exfoliation, resulting in the production of stable few-layer HfS2 flakes. Hafnium disulfide powder can be produced by reacting hydrogen sulfide and hafnium oxides at 500–1300 °C.

High-entropy oxides (HEOs) are complex oxides that contain five or more principal metal cations and have a single-phase crystal structure. The first HEO, (MgNiCuCoZn)0.2O in a rock salt structure, was reported in 2015 by Rost et al. HEOs have been successfully synthesized in many structures, including fluorites, perovskites, and spinels. HEOs are currently being investigated for applications as functional materials.
Silicon quantum dots are metal-free biologically compatible quantum dots with photoluminescence emission maxima that are tunable through the visible to near-infrared spectral regions. These quantum dots have unique properties arising from their indirect band gap, including long-lived luminescent excited-states and large Stokes shifts. A variety of disproportionation, pyrolysis, and solution protocols have been used to prepare silicon quantum dots, however it is important to note that some solution-based protocols for preparing luminescent silicon quantum dots actually yield carbon quantum dots instead of the reported silicon. The unique properties of silicon quantum dots lend themselves to an array of potential applications: biological imaging, luminescent solar concentrators, light emitting diodes, sensors, and lithium-ion battery anodes.
Hafnium compounds are compounds containing the element hafnium (Hf). Due to the lanthanide contraction, the ionic radius of hafnium(IV) (0.78 ångström) is almost the same as that of zirconium(IV) (0.79 angstroms). Consequently, compounds of hafnium(IV) and zirconium(IV) have very similar chemical and physical properties. Hafnium and zirconium tend to occur together in nature and the similarity of their ionic radii makes their chemical separation rather difficult. Hafnium tends to form inorganic compounds in the oxidation state of +4. Halogens react with it to form hafnium tetrahalides. At higher temperatures, hafnium reacts with oxygen, nitrogen, carbon, boron, sulfur, and silicon. Some compounds of hafnium in lower oxidation states are known.
References
- 1 2 None Available (2020). "Materials Data on La2Hf2O7 by Materials Project". Materials Project. doi:10.17188/1188964 . Retrieved 30 April 2021.
{{cite journal}}: Cite journal requires|journal=(help) - 1 2 Ji, Yaming; Jiang, Danyu; Fen, Tao; Shi, Jianlin (March 2005). "Fabrication of transparent La2Hf2O7 ceramics from combustion synthesized powders". Materials Research Bulletin. 40 (3): 553–559. doi:10.1016/j.materresbull.2004.10.010.
- 1 2 Popov, V V; Menushenkov, A P; Yastrebtsev, A A; Zubavichus, Ya V (September 2016). "La 2 Hf 2 O 7 crystal and local structure changes on the fluorite - pyrochlore phase transition". Journal of Physics: Conference Series. 747 (1): 012043. Bibcode:2016JPhCS.747a2043P. doi: 10.1088/1742-6596/747/1/012043 .
- ↑ Blanchard, Peter E. R.; Liu, Sam; Kennedy, Brendan J.; Ling, Chris D.; Avdeev, Max; Aitken, Jade B.; Cowie, Bruce C. C.; Tadich, Anton (7 February 2013). "Investigating the Local Structure of Lanthanoid Hafnates Ln 2 Hf 2 O 7 via Diffraction and Spectroscopy". The Journal of Physical Chemistry C. 117 (5): 2266–2273. doi:10.1021/jp311329q.
- ↑ Garg, Nandini; Pandey, K. K.; Murli, Chitra; Shanavas, K. V.; Mandal, Balaji P.; Tyagi, A. K.; Sharma, Surinder M. (13 June 2008). "Decomposition of lanthanum hafnate at high pressures". Physical Review B. 77 (21): 214105. Bibcode:2008PhRvB..77u4105G. doi:10.1103/PhysRevB.77.214105.
- ↑ Eagleman, Yetta; Weber, Marvin; Derenzo, Stephen (May 2013). "Luminescence study of oxygen vacancies in lanthanum hafnium oxide, La2Hf2O7". Journal of Luminescence. 137: 93–97. Bibcode:2013JLum..137...93E. doi:10.1016/j.jlumin.2012.10.034.
- ↑ Gupta, Santosh K.; Zuniga, Jose P.; Abdou, Maya; Thomas, Melonie P.; De Alwis Goonatilleke, Manisha; Guiton, Beth S.; Mao, Yuanbing (January 2020). "Lanthanide-doped lanthanum hafnate nanoparticles as multicolor phosphors for warm white lighting and scintillators". Chemical Engineering Journal. 379: 122314. Bibcode:2020ChEnJ.37922314G. doi: 10.1016/j.cej.2019.122314 . S2CID 199650816.
- ↑ Trojan-Piegza, Joanna; Zych, Eugeniusz (March 2021). "White persistent luminescence of La2Hf2O7:Ti,Pr". Optical Materials. 113: 110896. Bibcode:2021OptMa.11310896T. doi:10.1016/j.optmat.2021.110896. S2CID 233842128.
- ↑ Wahid, Kareem; Pokhrel, Madhab; Mao, Yuanbing (January 2017). "Structural, photoluminescence and radioluminescence properties of Eu3+ doped La2Hf2O7 nanoparticles". Journal of Solid State Chemistry. 245: 89–97. Bibcode:2017JSSCh.245...89W. doi: 10.1016/j.jssc.2016.10.004 .