Related Research Articles

The alkali metals consist of the chemical elements lithium (Li), sodium (Na), potassium (K), rubidium (Rb), caesium (Cs), and francium (Fr). Together with hydrogen they constitute group 1, which lies in the s-block of the periodic table. All alkali metals have their outermost electron in an s-orbital: this shared electron configuration results in their having very similar characteristic properties. Indeed, the alkali metals provide the best example of group trends in properties in the periodic table, with elements exhibiting well-characterised homologous behaviour. This family of elements is also known as the lithium family after its leading element.

The atomic number or nuclear charge number of a chemical element is the charge number of an atomic nucleus. For ordinary nuclei composed of protons and neutrons, this is equal to the proton number (np) or the number of protons found in the nucleus of every atom of that element. The atomic number can be used to uniquely identify ordinary chemical elements. In an ordinary uncharged atom, the atomic number is also equal to the number of electrons.
A chemical element is a chemical substance that cannot be broken down into other substances by chemical reactions. The basic particle that constitutes a chemical element is the atom. Chemical elements are identified by the number of protons in the nuclei of their atoms, known as the element's atomic number. For example, oxygen has an atomic number of 8, meaning that each oxygen atom has 8 protons in its nucleus. Two or more atoms of the same element can combine to form molecules, in contrast to chemical compounds or mixtures, which contain atoms of different elements. Atoms can be transformed into different elements in nuclear reactions, which change an atom's atomic number.
Electronegativity, symbolized as χ, is the tendency for an atom of a given chemical element to attract shared electrons when forming a chemical bond. An atom's electronegativity is affected by both its atomic number and the distance at which its valence electrons reside from the charged nucleus. The higher the associated electronegativity, the more an atom or a substituent group attracts electrons. Electronegativity serves as a simple way to quantitatively estimate the bond energy, and the sign and magnitude of a bond's chemical polarity, which characterizes a bond along the continuous scale from covalent to ionic bonding. The loosely defined term electropositivity is the opposite of electronegativity: it characterizes an element's tendency to donate valence electrons.
Francium is a chemical element; it has symbol Fr and atomic number 87. It is extremely radioactive; its most stable isotope, francium-223, has a half-life of only 22 minutes. It is the second-most electropositive element, behind only caesium, and is the second rarest naturally occurring element. Francium's isotopes decay quickly into astatine, radium, and radon. The electronic structure of a francium atom is [Rn] 7s1; thus, the element is classed as an alkali metal.
A molecule is a group of two or more atoms held together by attractive forces known as chemical bonds; depending on context, the term may or may not include ions which satisfy this criterion. In quantum physics, organic chemistry, and biochemistry, the distinction from ions is dropped and molecule is often used when referring to polyatomic ions.

The noble gases are the naturally occurring members of group 18 of the periodic table: helium (He), neon (Ne), argon (Ar), krypton (Kr), xenon (Xe), and radon (Rn). Under standard conditions, these elements are odorless, colorless, monatomic gases with very low chemical reactivity and cryogenic boiling points.

Neon is a chemical element; it has symbol Ne and atomic number 10. It is the second noble gas in the periodic table. Neon is a colorless, odorless, inert monatomic gas under standard conditions, with approximately two-thirds the density of air.

The periodic table, also known as the periodic table of the elements, is an ordered arrangement of the chemical elements into rows ("periods") and columns ("groups"). It is an icon of chemistry and is widely used in physics and other sciences. It is a depiction of the periodic law, which states that when the elements are arranged in order of their atomic numbers an approximate recurrence of their properties is evident. The table is divided into four roughly rectangular areas called blocks. Elements in the same group tend to show similar chemical characteristics.

Chemical symbols are the abbreviations used in chemistry, mainly for chemical elements; but also for functional groups, chemical compounds, and other entities. Element symbols for chemical elements normally consist of one or two letters from the Latin alphabet and are written with the first letter capitalised.
Tennessine is a synthetic chemical element; it has symbol Ts and atomic number 117. It has the second-highest atomic number and joint-highest atomic mass of all known elements, and is the penultimate element of the 7th period of the periodic table.

Nonmetals are chemical elements that mostly lack distinctive metallic properties. They range from colorless gases like hydrogen to shiny crystals like iodine. Physically, they are usually lighter than metals; brittle or crumbly if solid; and often poor conductors of heat and electricity. Chemically, nonmetals have high electronegativity ; and their oxides tend to be acidic.
A period 2 element is one of the chemical elements in the second row of the periodic table of the chemical elements. The periodic table is laid out in rows to illustrate recurring (periodic) trends in the chemical behavior of the elements as their atomic number increases; a new row is started when chemical behavior begins to repeat, creating columns of elements with similar properties.
A period 1 element is one of the chemical elements in the first row of the periodic table of the chemical elements. The periodic table is laid out in rows to illustrate periodic (recurring) trends in the chemical behaviour of the elements as their atomic number increases: a new row is begun when chemical behaviour begins to repeat, meaning that analog elements fall into the same vertical columns. The first period contains fewer elements than any other row in the table, with only two: hydrogen and helium. This situation can be explained by modern theories of atomic structure. In a quantum mechanical description of atomic structure, this period corresponds to the filling of the 1s orbital. Period 1 elements obey the duet rule in that they need two electrons to complete their valence shell.
Chemistry is the physical science concerned with the composition, structure, and properties of matter, as well as the changes it undergoes during chemical reactions.
Core electrons are the electrons in an atom that are not valence electrons and do not participate in chemical bonding. The nucleus and the core electrons of an atom form the atomic core. Core electrons are tightly bound to the nucleus. Therefore, unlike valence electrons, core electrons play a secondary role in chemical bonding and reactions by screening the positive charge of the atomic nucleus from the valence electrons.
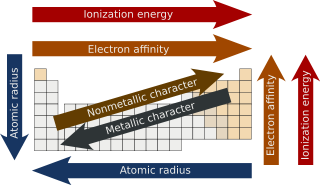
In chemistry, periodic trends are specific patterns that are present in the periodic table that illustrate different aspects of certain elements when grouped by period and/or group. They were discovered by the Russian chemist Dmitri Mendeleev in 1863. Major periodic trends include atomic radius, ionization energy, electron affinity, electronegativity, valency and metallic character. These trends exist because of the similar electron configurations of the elements within their respective groups or periods; they reflect the periodic nature of the elements. These trends give a qualitative assessment of the properties of each element.
This glossary of chemistry terms is a list of terms and definitions relevant to chemistry, including chemical laws, diagrams and formulae, laboratory tools, glassware, and equipment. Chemistry is a physical science concerned with the composition, structure, and properties of matter, as well as the changes it undergoes during chemical reactions; it features an extensive vocabulary and a significant amount of jargon.
There are currently 118 known chemical elements with a wide range of physical and chemical properties. Amongst this diversity, scientists have found it useful to apply names for various sets of elements that have similar properties, to varying degrees. Many of these sets are formally recognized by the standards body IUPAC.

Isotopes are distinct nuclear species of the same chemical element. They have the same atomic number and position in the periodic table, but differ in nucleon numbers due to different numbers of neutrons in their nuclei. While all isotopes of a given element have similar chemical properties, they have different atomic masses and physical properties.
References
- ↑ IUPAC , Compendium of Chemical Terminology , 2nd ed. (the "Gold Book") (1997). Online corrected version: (2006–) " chemical element ". doi : 10.1351/goldbook.C01022
- ↑ "Periodic Table – Royal Society of Chemistry". www.rsc.org.
- ↑ "Online Etymology Dictionary". etymonline.com.
- ↑ "beryl". Merriam-Webster . Archived from the original on 9 October 2013. Retrieved 27 January 2014.
- ↑ van der Krogt, Peter. "Wolframium Wolfram Tungsten". Elementymology & Elements Multidict. Archived from the original on 2010-01-23. Retrieved 2010-03-11.
- ↑ Originally assessed as 0.7 by Pauling but never revised after other elements' electronegativities were updated for precision. Predicted to be higher than that of caesium.
- ↑ Konings, Rudy J. M.; Beneš, Ondrej. "The Thermodynamic Properties of the 𝑓-Elements and Their Compounds. I. The Lanthanide and Actinide Metals". Journal of Physical and Chemical Reference Data. doi:10.1063/1.3474238.
- ↑ "Fermium". RSC.