
The Biginelli reaction is a multiple-component chemical reaction that creates 3,4-dihydropyrimidin-2(1H)-ones 4 from ethyl acetoacetate 1, an aryl aldehyde, and urea 3. It is named for the Italian chemist Pietro Biginelli.
Farnesyl pyrophosphate (FPP), also known as farnesyl diphosphate (FDP), is an intermediate in the biosynthesis of terpenes and terpenoids such as sterols and carotenoids. It is also used in the synthesis of CoQ, as well as dehydrodolichol diphosphate.
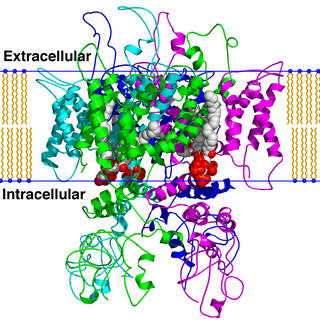
TRPV is a family of transient receptor potential cation channels in animals. All TRPVs are highly calcium selective.

Transient receptor potential cation channel, subfamily V, member 3, also known as TRPV3, is a human gene encoding the protein of the same name.

The adenosine A2B receptor, also known as ADORA2B, is a G-protein coupled adenosine receptor, and also denotes the human adenosine A2b receptor gene which encodes it.
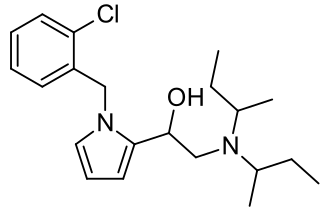
Viminol is an opioid analgesic developed by a team at the drug company Zambon in the 1960s. Viminol is based on the α-pyrryl-2-aminoethanol structure, unlike any other class of opioids.

NESS-0327 is a drug used in scientific research which acts as an extremely potent and selective antagonist of the cannabinoid receptor CB1. It is much more potent an antagonist, and more selective for the CB1 receptor over CB2, than the more commonly used ligand rimonabant, with a Ki at CB1 of 350fM (i.e. 0.00035nM) and a selectivity of over 60,000x for CB1 over CB2. Independently, two other groups have described only modest nanomolar CB1 affinity for this compound (125nM and 18.4nM). Also unlike rimonabant, NESS-0327 does not appear to act as an inverse agonist at higher doses, instead being a purely neutral antagonist which blocks the CB1 receptor but does not produce any physiological effect of its own.

Cyprodime is an opioid antagonist from the morphinan family of drugs.
TAN-67 (SB-205,607) is an opioid drug used in scientific research that acts as a potent and selective δ-opioid agonist, selective for the δ1 subtype. It has analgesic properties and induces dopamine release in nucleus accumbens. It also protects both heart and brain tissue from hypoxic tissue damage through multiple mechanisms involving among others an interaction between δ receptors and mitochondrial K(ATP) channels.

J-113,397 is an opioid drug which was the first compound found to be a highly selective antagonist for the nociceptin receptor, also known as the ORL-1 receptor. It is several hundred times selective for the ORL-1 receptor over other opioid receptors, and its effects in animals include preventing the development of tolerance to morphine, the prevention of hyperalgesia induced by intracerebroventricular administration of nociceptin, as well as the stimulation of dopamine release in the striatum, which increases the rewarding effects of cocaine, but may have clinical application in the treatment of Parkinson's disease.
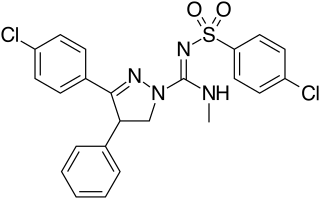
Ibipinabant (SLV319, BMS-646,256) is a drug used in scientific research which acts as a potent and highly selective CB1 antagonist. It has potent anorectic effects in animals, and was researched for the treatment of obesity, although CB1 antagonists as a class have now fallen out of favour as potential anorectics following the problems seen with rimonabant, and so ibipinabant is now only used for laboratory research, especially structure-activity relationship studies into novel CB1 antagonists. SLV330, which is a structural analogue of Ibipinabant, was reported active in animal models related to the regulation of memory, cognition, as well as in addictive behavior. An atom-efficient synthesis of ibipinabant has been reported.

Arylcyclohexylamines, also known as arylcyclohexamines or arylcyclohexanamines, are a chemical class of pharmaceutical, designer, and experimental drugs.

CSP-2503 is a potent and selective 5-HT1A receptor agonist, 5-HT2A receptor antagonist, and 5-HT3 receptor antagonist of the naphthylpiperazine class. First synthesized in 2003, it was designed based on computational models and QSAR studies. In rat studies, CSP-2503 has demonstrated anxiolytic effects, and thus has been suggested as a treatment for anxiety in humans with a multimodal mechanism of action.

SB-206553 is a drug which acts as a mixed antagonist for the 5-HT2B and 5-HT2C serotonin receptors. It has anxiolytic properties in animal studies and interacts with a range of other drugs. It has also been shown to act as a positive allosteric modulator of α7 nicotinic acetylcholine receptors. Modified derivatives of SB-206553 have been used to probe the structure of the 5-HT2B receptor.
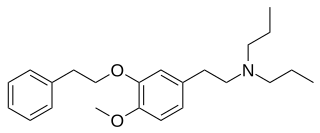
NE-100 or 4-methoxy-3-(2-phenylethoxy)-N,N-dipropylbenzeneethanamine is a selective sigma-1 receptor antagonist, with a reported binding affinity of Ki = 1.03 ± 0.01 nM, and more than 205 times selectivity over the sigma-2 receptor.

E-52862, also known as sigma-1 receptor antagonist, as well as MR-309, is a selective sigma-1 receptor antagonist, with a reported binding affinity of Ki = 17.0 ± 7.0 nM, selective over the sigma-2 receptor and against a panel of other 170 receptors, enzymes, transporters and ion channels. In preclinical studies, S1RA has demonstrated efficacy in relieving neuropathic pain and pain in other sensitizing conditions, associated with an improvement of the emotional negative state.

Aticaprant, also known by its developmental codes JNJ-67953964, CERC-501, and LY-2456302, is a κ-opioid receptor (KOR) antagonist which is under development for the treatment of major depressive disorder. A regulatory application for approval of the medication is expected to be submitted by 2025. Aticaprant is taken by mouth.

WB-4101 is a compound which acts as an antagonist at the α1B-adrenergic receptor. It was one of the first selective antagonists developed for this receptor and was invented in 1969, but is still commonly used in research into adrenergic receptors, especially as a lead compound from which to develop more selective drugs.

Drofenine is an antimuscarinic antispasmodic drug used for relaxing smooth muscle, thereby treating conditions, such as: dysmenorrhea, and pain in the gastrointestinal tract, biliary passages, and urogenital tract. Drofenine is assumed to work by increasing the levels of the protein TRPV3.

AZD 9272 is a drug which acts as a selective antagonist for the metabotropic glutamate receptor subtype mGluR5. It was unsuccessful in human trials as an analgesic, but continues to be widely used in research especially as its radiolabelled forms.



















