Related Research Articles

The United States Food and Drug Administration is a federal agency of the Department of Health and Human Services. The FDA is responsible for protecting and promoting public health through the control and supervision of food safety, tobacco products, caffeine products, dietary supplements, prescription and over-the-counter pharmaceutical drugs (medications), vaccines, biopharmaceuticals, blood transfusions, medical devices, electromagnetic radiation emitting devices (ERED), cosmetics, animal foods & feed and veterinary products.

Gaucher's disease or Gaucher disease (GD) is a genetic disorder in which glucocerebroside accumulates in cells and certain organs. The disorder is characterized by bruising, fatigue, anemia, low blood platelet count and enlargement of the liver and spleen, and is caused by a hereditary deficiency of the enzyme glucocerebrosidase, which acts on glucocerebroside. When the enzyme is defective, glucocerebroside accumulates, particularly in white blood cells and especially in macrophages. Glucocerebroside can collect in the spleen, liver, kidneys, lungs, brain, and bone marrow.

The Food and Drug Administration's (FDA) New Drug Application (NDA) is the vehicle in the United States through which drug sponsors formally propose that the FDA approve a new pharmaceutical for sale and marketing. Some 30% or less of initial drug candidates proceed through the entire multi-year process of drug development, concluding with an approved NDA, if successful.
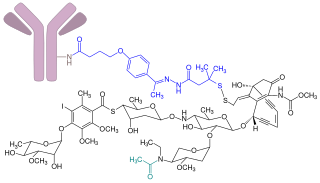
Gemtuzumab ozogamicin, sold under the brand name Mylotarg, is an antibody-drug conjugate that is used to treat acute myeloid leukemia.

Asparaginase is an enzyme that is used as a medication and in food manufacturing. As a medication, L-asparaginase is used to treat acute lymphoblastic leukemia (ALL) and lymphoblastic lymphoma (LBL). It is given by injection into a vein, muscle, or under the skin. A pegylated version is also available. In food manufacturing it is used to decrease acrylamide.

Peramivir is an antiviral drug developed by BioCryst Pharmaceuticals for the treatment of influenza. Peramivir is a neuraminidase inhibitor, acting as a transition-state analogue inhibitor of influenza neuraminidase and thereby preventing new viruses from emerging from infected cells. It is approved for intravenous administration.

β-Glucocerebrosidase is an enzyme with glucosylceramidase activity that cleaves by hydrolysis the β-glycosidic linkage of the chemical glucocerebroside, an intermediate in glycolipid metabolism that is abundant in cell membranes. It is localized in the lysosome, where it remains associated with the lysosomal membrane. β-Glucocerebrosidase is 497 amino acids in length and has a molecular mass of 59,700 Da.

Miglustat, sold under the brand name Zavesca among others, is a medication used to treat type I Gaucher disease (GD1) and Pompe disease. It is also known as N-butyldeoxynojirimycin, and is a derivative of the anti-diabetic 1-deoxynojirimycin. It was developed by Oxford GlycoSciences and is marketed by Actelion.
Alglucosidase alfa, sold under the brand name Myozyme among others, is an enzyme replacement therapy (ERT) orphan drug for treatment of Pompe disease, a rare lysosomal storage disorder (LSD). Chemically, the drug is an analog of the enzyme that is deficient in patients affected by Pompe disease, alpha-glucosidase. It is the first drug available to treat this disease.
Blinatumomab, sold under the brand name Blincyto, is a biopharmaceutical medication used as a second-line treatment for Philadelphia chromosome-negative relapsed or refractory acute lymphoblastic leukemia. It belongs to a class of constructed monoclonal antibodies, bi-specific T-cell engagers (BiTEs), that exert action selectively and direct the human immune system to act against tumor cells. Blinatumomab specifically targets the CD19 antigen present on B cells. In December 2014, it was approved by the US Food and Drug Administration under the accelerated approval program; marketing authorization depended on the outcome of clinical trials that were ongoing at the time of approval.
Velaglucerase alfa, sold under the brand name Vpriv, is a medication used for the treatment of Gaucher disease Type 1. It is a hydrolytic lysosomal glucocerebroside-specific enzyme, which is a recombinant form of glucocerebrosidase. It has an identical amino acid sequence to the naturally occurring enzyme. It is manufactured by Shire plc.

Apixaban, sold under the brand name Eliquis, is an anticoagulant medication used to treat and prevent blood clots and to prevent stroke in people with nonvalvular atrial fibrillation through directly inhibiting factor Xa. Specifically, it is used to prevent blood clots following hip or knee replacement and in those with a history of prior clots. It is used as an alternative to warfarin and does not require monitoring by blood tests or dietary restrictions. It is taken by mouth.

Protalix BioTherapeutics is an Israeli pharmaceutical company that manufactures a plant-based enzyme, taliglucerase alfa, which has received U.S. Food and Drug Administration approval for the treatment of Gaucher disease.

Efinaconazole, sold under the brand name Jublia among others, is a triazole antifungal medication. It is approved for use in the United States, Canada, and Japan as a 10% topical solution for the treatment of onychomycosis. Efinaconazole acts as a 14α-demethylase inhibitor.
Sebelipase alfa, sold under the brand name Kanuma, is a recombinant form of the enzyme lysosomal acid lipase (LAL) that is used as a medication for the treatment of lysosomal acid lipase deficiency (LAL-D). It is administered via intraveneous infusion. It was approved for medical use in the European Union and in the United States in 2015.

Migalastat, sold under the brand name Galafold, is a medication for the treatment of Fabry disease, a rare genetic disorder. It was developed by Amicus Therapeutics. The US Food and Drug Administration (FDA) granted it orphan drug status in 2004, and the European Commission followed in 2006. The European Medicines Agency's Committee for Medicinal Products for Human Use (CHMP) granted the drug a marketing approval under the name Galafold in May 2016.
Cerliponase alfa, marketed as Brineura, is an enzyme replacement treatment for Batten disease, a neurodegenerative lysosomal storage disease. Specifically, Cerliponase alfa is meant to slow loss of motor function in symptomatic children over three years old with late infantile neuronal ceroid lipofuscinosis type 2 (CLN2). The disease is also known as tripeptidyl peptidase-1 (TPP1) deficiency, a soluble lysosomal enzyme deficiency. Approved by the United States Food and Drug Administration (FDA) on 27 April 2017, this is the first treatment for a neuronal ceroid lipofuscinosis of its kind, acting to slow disease progression rather than palliatively treat symptoms by giving patients the TPP1 enzyme they are lacking.
Vestronidase alfa, sold under brand name Mepsevii, is a drug for the treatment of Sly syndrome. It is a recombinant form of the human enzyme beta-glucuronidase. It was approved in the United States in November 2017, to treat children and adults with an inherited metabolic condition called mucopolysaccharidosis type VII, also known as Sly syndrome. MPS VII is an extremely rare, progressive condition that affects most tissues and organs.
Avalglucosidase alfa, sold under the brand name Nexviazyme, is an enzyme replacement therapy medication used for the treatment of glycogen storage disease type II.
Olipudase alfa, sold under the brand name Xenpozyme, is a medication used for the treatment of non-central nervous system (CNS) manifestations of acid sphingomyelinase deficiency (ASMD) type A/B or type B.
References
- ↑ "Prescription medicines: registration of new chemical entities in Australia, 2014". Therapeutic Goods Administration (TGA). 21 June 2022. Retrieved 10 April 2023.
- ↑ Aviezer D, Brill-Almon E, Shaaltiel Y, Hashmueli S, Bartfeld D, Mizrachi S, et al. (2009). Ho PL (ed.). "A plant-derived recombinant human glucocerebrosidase enzyme--a preclinical and phase I investigation". PLOS ONE. 4 (3): e4792. Bibcode:2009PLoSO...4.4792A. doi: 10.1371/journal.pone.0004792 . PMC 2652073 . PMID 19277123.
- ↑ Clinical trial number NCT00962260 for "Expanded Access Trial of Plant Expressed Recombinant Glucocerebrosidase (prGCD) in Patients With Gaucher Disease" at ClinicalTrials.gov
- ↑ Kaiser J (April 2008). "Is the drought over for pharming?". Science. 320 (5875): 473–5. doi:10.1126/science.320.5875.473. PMID 18436771. S2CID 28407422.
- ↑ Maxmen A (2 May 2012). "First plant-made drug on the market". Nature, Biology & Biotechnology, Industry. Archived from the original on 18 October 2012. Retrieved 26 June 2012.
- ↑ Beitz J. "Elenyso (taliglucerase alfa) NDA Approval" (PDF). Center for Drug Evaluation and Research. U.S. Food and Drug Administration.
- ↑ "Elenyso (taliglucerase alfa): Highlights of Prescribing Information" (PDF). U.S. Food and Drug Administration.
- ↑ "Supplement Approval Fulfillment of Postmarketing Requirement" (PDF). U.S. Food and Drug Administration.
- ↑ "מאגר התרופות (Drug details for Elelyso)". Ministry of Health Israel. Archived from the original on 2017-09-08. Retrieved 2017-09-08.
- ↑ "Summary Basis of Decision (SBD): Elelyso". Health Canada. 2014-08-01. Archived from the original on 2014-08-06.
- ↑ "Medical Pharmacy Trend Report" (PDF). Magellan Rx Management (seventh ed.). 2016. Archived from the original (PDF) on 29 March 2019.