Lipids are a broad group of naturally-occurring molecules which includes fats, waxes, sterols, fat-soluble vitamins, monoglycerides, diglycerides, phospholipids, and others. The functions of lipids include storing energy, signaling, and acting as structural components of cell membranes. Lipids have applications in the cosmetic and food industries, and in nanotechnology.
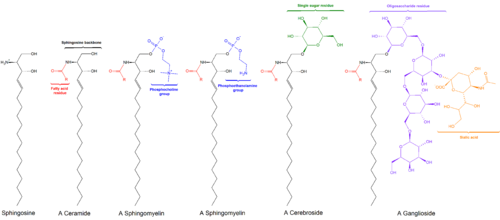
Sphingomyelin is a type of sphingolipid found in animal cell membranes, especially in the membranous myelin sheath that surrounds some nerve cell axons. It usually consists of phosphocholine and ceramide, or a phosphoethanolamine head group; therefore, sphingomyelins can also be classified as sphingophospholipids. In humans, SPH represents ~85% of all sphingolipids, and typically make up 10–20 mol % of plasma membrane lipids.
Sphingosine (2-amino-4-trans-octadecene-1,3-diol) is an 18-carbon amino alcohol with an unsaturated hydrocarbon chain, which forms a primary part of sphingolipids, a class of cell membrane lipids that include sphingomyelin, an important phospholipid.

Fumonisin B1 is the most prevalent member of a family of toxins, known as fumonisins, produced by several species of Fusarium molds, such as Fusarium verticillioides, which occur mainly in maize (corn), wheat and other cereals. Fumonisin B1 contamination of maize has been reported worldwide at mg/kg levels. Human exposure occurs at levels of micrograms to milligrams per day and is greatest in regions where maize products are the dietary staple.

Ceramides are a family of waxy lipid molecules. A ceramide is composed of N-acetylsphingosine and a fatty acid. Ceramides are found in high concentrations within the cell membrane of eukaryotic cells, since they are component lipids that make up sphingomyelin, one of the major lipids in the lipid bilayer. Contrary to previous assumptions that ceramides and other sphingolipids found in cell membrane were purely supporting structural elements, ceramide can participate in a variety of cellular signaling: examples include regulating differentiation, proliferation, and programmed cell death (PCD) of cells.

Sphingosine kinase (SphK) is a conserved lipid kinase that catalyzes formation sphingosine-1-phosphate (S1P) from the precursor sphingolipid sphingosine. Sphingolipid metabolites, such as ceramide, sphingosine and sphingosine-1-phosphate, are lipid second messengers involved in diverse cellular processes. There are two forms of SphK, SphK1 and SphK2. SphK1 is found in the cytosol of eukaryotic cells, and migrates to the plasma membrane upon activation. SphK2 is localized to the nucleus.

Lipid signaling, broadly defined, refers to any biological signaling event involving a lipid messenger that binds a protein target, such as a receptor, kinase or phosphatase, which in turn mediate the effects of these lipids on specific cellular responses. Lipid signaling is thought to be qualitatively different from other classical signaling paradigms because lipids can freely diffuse through membranes. One consequence of this is that lipid messengers cannot be stored in vesicles prior to release and so are often biosynthesized "on demand" at their intended site of action. As such, many lipid signaling molecules cannot circulate freely in solution but, rather, exist bound to special carrier proteins in serum.

Sphingomyelin phosphodiesterase is a hydrolase enzyme that is involved in sphingolipid metabolism reactions. SMase is a member of the DNase I superfamily of enzymes and is responsible for breaking sphingomyelin (SM) down into phosphocholine and ceramide. The activation of SMase has been suggested as a major route for the production of ceramide in response to cellular stresses.
Sphingosine-1-phosphate (S1P) is a signaling sphingolipid, also known as lysosphingolipid. It is also referred to as a bioactive lipid mediator. Sphingolipids at large form a class of lipids characterized by a particular aliphatic aminoalcohol, which is sphingosine.

The enzyme phosphatidate phosphatase (PAP, EC 3.1.3.4) is a key regulatory enzyme in lipid metabolism, catalyzing the conversion of phosphatidate to diacylglycerol:

In enzymology, a serine C-palmitoyltransferase (EC 2.3.1.50) is an enzyme that catalyzes the chemical reaction:
In enzymology, sphingosine N-acyltransferases (ceramide synthases (CerS), EC 2.3.1.24) are enzymes that catalyze the chemical reaction of synthesis of ceramide:
In enzymology, a ceramide kinase, also abbreviated as CERK, is an enzyme that catalyzes the chemical reaction:

Collagen type IV alpha-3-binding protein, also known as ceramide transfer protein (CERT) or StAR-related lipid transfer protein 11 (STARD11) is a protein that in humans is encoded by the COL4A3BP gene. The protein contains a pleckstrin homology domain at its amino terminus and a START domain towards the end of the molecule. It is a member of the StarD2 subfamily of START domain proteins.

Serine palmitoyltransferase, long chain base subunit 2, also known as SPTLC2, is a protein which in humans is encoded by the SPTLC2 gene. SPTLC2 belongs to the class-II pyridoxal-phosphate-dependent aminotransferase family.
Neutral ceramidase C also known as N-acylsphingosine amidohydrolase 2C or non-lysosomal ceramidase C or ASAH2C is a ceramidase enzyme which in humans is encoded by the ASAH2C gene.

Alkaline ceramidase 1 also known as ACER1 is a ceramidase enzyme which in humans is encoded by the ACER1 gene.

Ceramide synthase 3 (CersS3), also known as longevity assurance homologue 3, is an enzyme that is encoded in humans by the CERS3 gene.
Ceramide synthase 5 (CerS5) is the enzyme encoded in humans by the CERS5 gene.
The 1-deoxysphingolipids (1-deoxySLs) are an atypical and recently discovered class of sphingolipids (SLs). They are formed during the nove synthesis pathway and their essential C1-OH deficit causes the malfunctions of the following transformations to achieve complex sphingolipids. In general, sphingolipids are formed during a reaction that is catalyzed by the SPT enzyme (serine-palmitoyltransferase) where the condensation of serine and palmitoyl-CoA takes place. The origin of this rare sphingolipid, though, is due to a defect of the SPT which can also use alanine or glycine. This change is what forms the 1-deoxySL.