
Bromocriptine, originally marketed as Parlodel and subsequently under many brand names, is an ergoline derivative and dopamine agonist that is used in the treatment of pituitary tumors, Parkinson's disease, hyperprolactinaemia, neuroleptic malignant syndrome, and, as an adjunct, type 2 diabetes.

Dopamine receptors are a class of G protein-coupled receptors that are prominent in the vertebrate central nervous system (CNS). Dopamine receptors activate different effectors through not only G-protein coupling, but also signaling through different protein interactions. The neurotransmitter dopamine is the primary endogenous ligand for dopamine receptors.

(–)-2-β-Carbomethoxy-3-β-(4-fluorophenyl)tropane is a stimulant drug used in scientific research. CFT is a phenyltropane based dopamine reuptake inhibitor and is structurally derived from cocaine. It is around 3-10x more potent than cocaine and lasts around 7 times longer based on animal studies. While the naphthalenedisulfonate salt is the most commonly used form in scientific research due to its high solubility in water, the free base and hydrochloride salts are known compounds and can also be produced. The tartrate is another salt form that is reported.
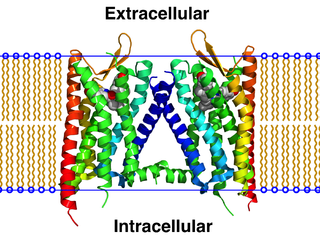
The κ-opioid receptor or kappa opioid receptor, abbreviated KOR or KOP for its ligand ketazocine, is a G protein-coupled receptor that in humans is encoded by the OPRK1 gene. The KOR is coupled to the G protein Gi/G0 and is one of four related receptors that bind opioid-like compounds in the brain and are responsible for mediating the effects of these compounds. These effects include altering nociception, consciousness, motor control, and mood. Dysregulation of this receptor system has been implicated in alcohol and drug addiction.

Berberine is a quaternary ammonium salt from the protoberberine group of benzylisoquinoline alkaloids

Desmetramadol (INN), also known as O-desmethyltramadol (O-DSMT), is an opioid analgesic and the main active metabolite of tramadol. Tramadol is demethylated by the liver enzyme CYP2D6 to desmetramadol in the same way as codeine, and so similarly to the variation in effects seen with codeine, individuals who have a less active form of CYP2D6 will tend to have reduced analgesic effects from tramadol. Because desmetramadol itself does not need to be metabolized to induce an analgesic effect, it can be used in individuals with low CYP2D6 activity unlike tramadol.

SB-277,011A is a drug which acts as a potent and selective dopamine D3 receptor antagonist, which is around 80-100x selective for D3 over D2, and lacks any partial agonist activity.

Glaucine(1,2,9,10-TetraMethoxyAporphine, Bromcholitin, Glauvent, Tusidil, Tussiglaucin) is an aporphine alkaloid found in several different plant species in the family Papaveraceae such as Glaucium flavum, Glaucium oxylobum and Corydalis yanhusuo, and in other plants like Croton lechleri in the family Euphorbiaceae.

(−)-Stepholidine is a protoberberine alkaloid found in the plant Stephania intermedia.

Oxaprotiline, also known as hydroxymaprotiline, is a norepinephrine reuptake inhibitor belonging to the tetracyclic antidepressant (TeCA) family and is related to maprotiline. Though investigated as an antidepressant, it was never marketed.

(S)-Canadine, also known as (S)-tetrahydroberberine and xanthopuccine, is a benzylisoquinoline alkaloid (BIA), of the protoberberine structural subgroup, and is present in many plants from the family Papaveraceae, such as Corydalis yanhusuo and C. turtschaninovii.

UH-232 ((+)-UH232) is a drug which acts as a subtype selective mixed agonist-antagonist for dopamine receptors, acting as a weak partial agonist at the D3 subtype, and an antagonist at D2Sh autoreceptors on dopaminergic nerve terminals. This causes dopamine release in the brain and has a stimulant effect, as well as blocking the behavioural effects of cocaine. It may also serve as a 5-HT2A receptor agonist, based on animal studies. It was investigated in clinical trials for the treatment of schizophrenia, but unexpectedly caused symptoms to become worse.

Nantenine is an alkaloid found in the plant Nandina domestica as well as some Corydalis species. It is an antagonist of both the α1-adrenergic receptor and the serotonin 5-HT2A receptor, and blocks both the behavioral and physiological effects of MDMA in animals.

An opiate, in classical pharmacology, is a substance derived from opium. In more modern usage, the term opioid is used to designate all substances, both natural and synthetic, that bind to opioid receptors in the brain. Opiates are alkaloid compounds naturally found in the opium poppy plant Papaver somniferum. The psychoactive compounds found in the opium plant include morphine, codeine, and thebaine. Opiates have long been used for a variety of medical conditions with evidence of opiate trade and use for pain relief as early as the eighth century AD. Opiates are considered drugs with moderate to high abuse potential and are listed on various "Substance-Control Schedules" under the Uniform Controlled Substances Act of the United States of America.

PD-128,907 is a drug used in scientific research which acts as a potent and selective agonist for the dopamine D2 and D3 receptors. It is used for studying the role of these receptors in the brain, in roles such as inhibitory autoreceptors that act to limit further dopamine release, as well as release of other neurotransmitters. In animal studies, it has been shown to reduce toxicity from cocaine overdose.

Osemozotan (MKC-242) is a selective 5-HT1A receptor agonist with some functional selectivity, acting as a full agonist at presynaptic and a partial agonist at postsynaptic 5-HT1A receptors. 5-HT1A receptor stimulation influences the release of various neurotransmitters including serotonin, dopamine, norepinephrine, and acetylcholine. 5-HT1A receptors are inhibitory G protein-coupled receptor. Osemozotan has antidepressant, anxiolytic, antiobsessional, serenic, and analgesic effects in animal studies, and is used to investigate the role of 5-HT1A receptors in modulating the release of dopamine and serotonin in the brain, and their involvement in addiction to abused stimulants such as cocaine and methamphetamine.
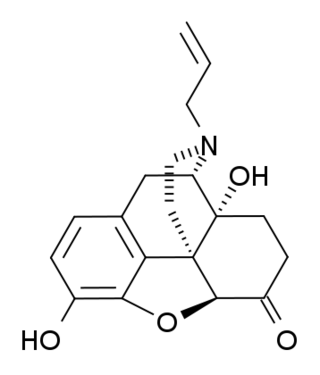
(+)-Naloxone (dextro-naloxone) is a drug which is the opposite enantiomer of the opioid antagonist drug (−)-naloxone. Unlike (-)-naloxone, (+)-naloxone has no significant affinity for opioid receptors, but instead has been discovered to act as a selective antagonist of Toll-like receptor 4. This receptor is involved in immune system responses, and activation of TLR4 induces glial activation and release of inflammatory mediators such as TNF-α and Interleukin-1.
Corydalis yanhusuo is a plant species in the genus Corydalis. The Chinese name for Corydalis yanhusuo is yan hu suo. The Japanese common name is engosaku (エンゴサク) and the Korean common name is hyeonhosaek (현호색). English common names include yanhusuo, corydalis, and Asian corydalis. The tuber of this plant, frequently mislabeled as the root, is an important therapeutic agent in traditional Chinese medicine. It is native to high-altitude grasslands across China including in the provinces of Anhui, Henan, Hubei, Hunan, Jiangsu, and Zhejiang, but is more widely cultivated.
Guineesine is a compound isolated from long pepper and black pepper.

Securinine is an alkaloid found in Securinega suffruticosa and Phyllanthus niruri.


















