Polyphenols are a large family of naturally occurring phenols. They are abundant in plants and structurally diverse. Polyphenols include flavonoids, tannic acid, and ellagitannin, some of which have been used historically as dyes and for tanning garments.

Lipid emulsion or fat emulsion refers to an emulsion of fat for human intravenous use, to administer nutrients to critically-ill patients that cannot consume food. It is often referred to by the brand name of the most commonly used version, Intralipid, which is an emulsion containing soybean oil, egg phospholipids and glycerin, and is available in 10%, 20% and 30% concentrations. The 30% concentration is not approved for direct intravenous infusion, but should be mixed with amino acids and dextrose as part of a total nutrient admixture.
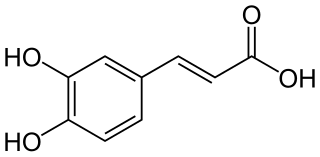
Caffeic acid is an organic compound that is classified as a hydroxycinnamic acid. This yellow solid consists of both phenolic and acrylic functional groups. It is found in all plants because it is an intermediate in the biosynthesis of lignin, one of the principal components of woody plant biomass and its residues.

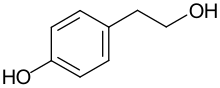
Hydroxytyrosol is an organic compound with the formula (HO)2C6H3CH2CH2OH. It is a phenylethanoid, i.e. a relative of phenethyl alcohol. Its derivatives are found in a variety of natural sources, notably olive oils and wines. Hydroxytyrosol is a colorless solid, although samples often turn beige during storage. It is a derivative, formally speaking, of catechol.

Oleuropein is a glycosylated seco-iridoid, a type of phenolic bitter compound found in green olive skin, flesh, seeds, and leaves. The term oleuropein is derived from the botanical name of the olive tree, Olea europaea.

Olive leaf is the leaf of the olive tree. Although olive oil is well known for its flavor and possible health benefits, the leaf and its extracts remain under preliminary research with unknown effects on human health.

Anthocyanins, also called anthocyans, are water-soluble vacuolar pigments that, depending on their pH, may appear red, purple, blue, or black. In 1835, the German pharmacist Ludwig Clamor Marquart gave the name Anthokyan to a chemical compound that gives flowers a blue color for the first time in his treatise "Die Farben der Blüthen". Food plants rich in anthocyanins include the blueberry, raspberry, black rice, and black soybean, among many others that are red, blue, purple, or black. Some of the colors of autumn leaves are derived from anthocyanins.

Elenolic acid is a component of olive oil, olive infusion and olive leaf extract. It can be considered as a marker for maturation of olives.
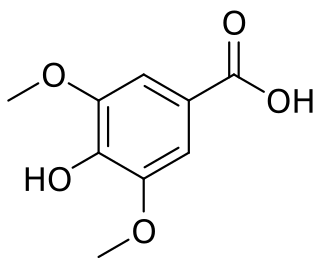
Syringic acid is a naturally occurring phenolic compound and dimethoxybenzene that is commonly found as a plant metabolite.

BRL-52537 is a drug which acts as a potent and highly selective κ-opioid agonist. It has neuroprotective effects in animal studies, and is used for research into potential treatments for stroke and heart attack as well as more general brain research.

In biochemistry, naturally occurring phenols are natural products containing at least one phenol functional group. Phenolic compounds are produced by plants and microorganisms. Organisms sometimes synthesize phenolic compounds in response to ecological pressures such as pathogen and insect attack, UV radiation and wounding. As they are present in food consumed in human diets and in plants used in traditional medicine of several cultures, their role in human health and disease is a subject of research. Some phenols are germicidal and are used in formulating disinfectants.
Vinyldithiins, more precisely named 3-vinyl-4H-1,2-dithiin and 2-vinyl-4H-1,3-dithiin, are organosulfur phytochemicals formed in the breakdown of allicin from crushed garlic (Allium sativum). Vinyldithiins are Diels-Alder dimers of thioacrolein, H2C=CHCH=S, formed in turn by decomposition of allicin. In garlic supplements, vinyldithiins are only found in garlic oil macerates that are made by incubation of crushed garlic in oil.

Miquelianin is a flavonol glucuronide, a type of phenolic compound present in wine, in species of St John's wort, like Hypericum hirsutum, in Nelumbo nucifera or in green beans.

Rottlerin (mallotoxin) is a polyphenol natural product isolated from the Asian tree Mallotus philippensis. Rottlerin displays a complex spectrum of pharmacology.

Lambertianin C is an ellagitannin.

Sanguiin H-6 is an ellagitannin.
Diallyl trisulfide (DATS), also known as Allitridin, is an organosulfur compound with the formula S(SCH2CH=CH2)2. It is one of several compounds produced by hydrolysis of allicin, including diallyl disulfide and diallyl tetrasulfide; DATS is one of the most potent.

Urolithin B (UB) is an urolithin, a type of phenolic compounds produced in the human gut after absorption of ellagitannins-containing food such as pomegranate, strawberries, red raspberries, walnuts or oak-aged red wine. Urolithin B is found in the urine in the form of urolithin B glucuronide.
Cardioprotection includes all mechanisms and means that contribute to the preservation of the heart by reducing or even preventing myocardial damage. Cardioprotection encompasses several regimens that have shown to preserve function and viability of cardiac muscle cell tissue subjected to ischemic insult or reoxygenation. Cardioprotection includes strategies that are implemented before an ischemic event, during an ischemic event and after the event and during reperfusion. These strategies can be further stratified by performing the intervention locally or remotely, creating classes of conditioning known as remote ischemic PC (RIPC), remote ischemic PostC and remote ischemic PerC. Classical (local) preconditioning has an early phase with an immediate onset lasting 2–3 hours that protects against myocardial infarction. The early phase involves post-translational modification of preexisting proteins, brought about by the activation of G protein-coupled receptors as well as downstream MAPK's and PI3/Akt. These signaling events act on the ROS-generating mitochondria, activate PKCε and the Reperfusion Injury Salvage Kinase (RISK) pathway, preventing mitochondrial permeability transition pore (MTP) opening. The late phase with an onset of 12–24 hours that lasts 3–4 days and protects against both infarction and reversible postischemic contractile dysfunction, termed myocardial stunning. This phase involves the synthesis of new cardioprotective proteins stimulated by nitric oxide (NO), ROS and adenosine acting on kinases such as PKCε and Src, which in turn activate gene transcription and upregulation of late PC molecular players.
Remote ischemic conditioning (RIC) is an experimental medical procedure that aims to reduce the severity of ischaemic injury to an organ such as the heart or the brain, most commonly in the situation of a heart attack or a stroke, or during procedures such as heart surgery when the heart may temporary suffer ischaemia during the operation, by triggering the body's natural protection against tissue injury. Although noted to have some benefits in experimental models in animals, this is still an experimental procedure in humans and initial evidence from small studies have not been replicated in larger clinical trials. Successive clinical trials have failed to identify evidence supporting a protective role in humans.