Related Research Articles
In biology, homeostasis is the state of steady internal, physical, chemical, and social conditions maintained by living systems. This is the condition of optimal functioning for the organism and includes many variables, such as body temperature and fluid balance, being kept within certain pre-set limits. Other variables include the pH of extracellular fluid, the concentrations of sodium, potassium, and calcium ions, as well as the blood sugar level, and these need to be regulated despite changes in the environment, diet, or level of activity. Each of these variables is controlled by one or more regulators or homeostatic mechanisms, which together maintain life.

In humans, the kidneys are two reddish-brown bean-shaped blood-filtering organs that are a multilobar multipapillary form of mammalian kidney, usually without signs of external lobulation. They are located on the left and right in the retroperitoneal space, and in adult humans are about 12 centimetres in length. They receive blood from the paired renal arteries; blood exits into the paired renal veins. Each kidney is attached to a ureter, a tube that carries excreted urine to the bladder.

The common ostrich, or simply ostrich, is a species of flightless bird native to certain large areas of Africa. It is one of two extant species of ostriches, the only living members of the genus Struthio in the ratite order of birds. The other is the Somali ostrich, which was recognized as a distinct species by BirdLife International in 2014 having been previously considered a distinctive subspecies of ostrich.

Diabetes insipidus (DI), alternately called arginine vasopressin deficiency (AVP-D) or arginine vasopressin resistance (AVP-R), is a condition characterized by large amounts of dilute urine and increased thirst. The amount of urine produced can be nearly 20 liters per day. Reduction of fluid has little effect on the concentration of the urine. Complications may include dehydration or seizures.

Oncotic pressure, or colloid osmotic-pressure, is a type of osmotic pressure induced by the plasma proteins, notably albumin, in a blood vessel's plasma that causes a pull on fluid back into the capillary. Participating colloids displace water molecules, thus creating a relative water molecule deficit with water molecules moving back into the circulatory system within the lower venous pressure end of capillaries.

In physiology, dehydration is a lack of total body water, with an accompanying disruption of metabolic processes. It occurs when free water loss exceeds free water intake, usually due to exercise, disease, or high environmental temperature. Mild dehydration can also be caused by immersion diuresis, which may increase risk of decompression sickness in divers.

The blood sugar level, blood sugar concentration, blood glucose level, or glycemia, is the measure of glucose concentrated in the blood. The body tightly regulates blood glucose levels as a part of metabolic homeostasis.
In physiology, body water is the water content of an animal body that is contained in the tissues, the blood, the bones and elsewhere. The percentages of body water contained in various fluid compartments add up to total body water (TBW). This water makes up a significant fraction of the human body, both by weight and by volume. Ensuring the right amount of body water is part of fluid balance, an aspect of homeostasis.

Renal physiology is the study of the physiology of the kidney. This encompasses all functions of the kidney, including maintenance of acid-base balance; regulation of fluid balance; regulation of sodium, potassium, and other electrolytes; clearance of toxins; absorption of glucose, amino acids, and other small molecules; regulation of blood pressure; production of various hormones, such as erythropoietin; and activation of vitamin D.

In the kidney, the loop of Henle is the portion of a nephron that leads from the proximal convoluted tubule to the distal convoluted tubule. Named after its discoverer, the German anatomist Friedrich Gustav Jakob Henle, the loop of Henle's main function is to create a concentration gradient in the medulla of the kidney.
Syndrome of inappropriate antidiuretic hormone secretion (SIADH) is characterized by excessive unsuppressible release of antidiuretic hormone (ADH) either from the posterior pituitary gland, or an abnormal non-pituitary source. Unsuppressed ADH causes an unrelenting increase in solute-free water being returned by the tubules of the kidney to the venous circulation.
Fluid balance is an aspect of the homeostasis of organisms in which the amount of water in the organism needs to be controlled, via osmoregulation and behavior, such that the concentrations of electrolytes in the various body fluids are kept within healthy ranges. The core principle of fluid balance is that the amount of water lost from the body must equal the amount of water taken in; for example, in humans, the output must equal the input. Euvolemia is the state of normal body fluid volume, including blood volume, interstitial fluid volume, and intracellular fluid volume; hypovolemia and hypervolemia are imbalances. Water is necessary for all life on Earth. Humans can survive for 4 to 6 weeks without food but only for a few days without water.
The trans-tubular potassium gradient (TTKG) is an index reflecting the conservation of potassium in the cortical collecting ducts (CCD) of the kidneys. It is useful in diagnosing the causes of hyperkalemia or hypokalemia. The TTKG estimates the ratio of potassium in the lumen of the CCD to that in the peritubular capillaries.
Osmol gap in medical science is the difference between measured serum osmolality and calculated serum osmolality.

Osmotic concentration, formerly known as osmolarity, is the measure of solute concentration, defined as the number of osmoles (Osm) of solute per litre (L) of solution. The osmolarity of a solution is usually expressed as Osm/L, in the same way that the molarity of a solution is expressed as "M". Whereas molarity measures the number of moles of solute per unit volume of solution, osmolarity measures the number of osmoles of solute particles per unit volume of solution. This value allows the measurement of the osmotic pressure of a solution and the determination of how the solvent will diffuse across a semipermeable membrane (osmosis) separating two solutions of different osmotic concentration.
Plasma osmolality measures the body's electrolyte–water balance. There are several methods for arriving at this quantity through measurement or calculation.
In the physiology of the kidney, free water clearance (CH2O) is the volume of blood plasma that is cleared of solute-free water per unit time. An example of its use is in the determination of an individual's state of hydration. Conceptually, free water clearance should be thought of relative to the production of isoosmotic urine, which would be equal to the osmolarity of the plasma. If an individual is producing urine more dilute than the plasma, there is a positive value for free water clearance, meaning pure water is lost in the urine in addition to a theoretical isoosmotic filtrate. If the urine is more concentrated than the plasma, then free water is being extracted from the urine, giving a negative value for free water clearance. A negative value is typical for free water clearance, as the kidney usually produces concentrated urine except in the cases of volume overload by the individual.
Volume contraction is a decrease in the volume of body fluid, including the dissolved substances that maintain osmotic balance (osmolytes). The loss of the water component of body fluid is specifically termed dehydration.
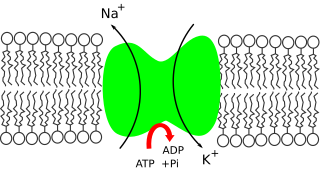
Sodium ions are necessary in small amounts for some types of plants, but sodium as a nutrient is more generally needed in larger amounts by animals, due to their use of it for generation of nerve impulses and for maintenance of electrolyte balance and fluid balance. In animals, sodium ions are necessary for the aforementioned functions and for heart activity and certain metabolic functions. The health effects of salt reflect what happens when the body has too much or too little sodium. Characteristic concentrations of sodium in model organisms are: 10 mM in E. coli, 30 mM in budding yeast, 10 mM in mammalian cell and 100 mM in blood plasma.

Potomania is a specific hypo-osmolality syndrome related to massive consumption of beer, which is poor in solutes and electrolytes. With little food or other sources of electrolytes, consumption of large amounts of beer or other dilute alcoholic drinks leads to electrolyte disturbances, where the body does not have enough nutrients known as electrolytes, namely sodium, potassium, and magnesium. The symptoms of potomania are similar to other causes of hyponatremia and include dizziness, muscular weakness, neurological impairment and seizures, all related to hyponatremia and hypokalaemia. While the symptoms of potomania are similar to other causes of hyponatremia and acute water intoxication, it should be considered an independent clinical entity because of its often chronic nature of onset, pathophysiology, and presentation of symptoms.
References
- ↑ "MedlinePlus Medical Encyclopedia: Osmolality - urine" . Retrieved 2009-03-29.
- 1 2 3 Sands, Jeff M.; Layton, Harold E. (2014-01-01). "Advances in Understanding the Urine-Concentrating Mechanism". Annual Review of Physiology. 76 (1): 387–409. doi: 10.1146/annurev-physiol-021113-170350 . PMID 24245944. – via Annual Reviews (subscription required)