Related Research Articles

Cowpox is an infectious disease caused by the cowpox virus (CPXV). It presents with large blisters in the skin, a fever and swollen glands, historically typically following contact with an infected cow, though in the last several decades more often from infected cats. The hands and face are most frequently affected and the spots are generally very painful.

The smallpox vaccine is the first vaccine to have been developed against a contagious disease. In 1796, British physician Edward Jenner demonstrated that an infection with the relatively mild cowpox virus conferred immunity against the deadly smallpox virus. Cowpox served as a natural vaccine until the modern smallpox vaccine emerged in the 20th century. From 1958 to 1977, the World Health Organization (WHO) conducted a global vaccination campaign that eradicated smallpox, making it the only human disease to be eradicated. Although routine smallpox vaccination is no longer performed on the general public, the vaccine is still being produced to guard against bioterrorism, biological warfare, and mpox.

Mpox is an infectious viral disease that can occur in humans and other animals. Symptoms include a rash that forms blisters and then crusts over, fever, and swollen lymph nodes. The illness is usually mild and most of those infected will recover within a few weeks without treatment. The time from exposure to onset of symptoms ranges from five to twenty-one days and symptoms typically last from two to four weeks. Cases may be severe, especially in children, pregnant women or people with suppressed immune systems.
In biology, immunity is the state of being insusceptible or resistant to a noxious agent or process, especially a pathogen or infectious disease. Immunity may occur naturally or be produced by prior exposure or immunization.

Vaccinia virus is a large, complex, enveloped virus belonging to the poxvirus family. It has a linear, double-stranded DNA genome approximately 190 kbp in length, which encodes approximately 250 genes. The dimensions of the virion are roughly 360 × 270 × 250 nm, with a mass of approximately 5–10 fg. The vaccinia virus is the source of the modern smallpox vaccine, which the World Health Organization (WHO) used to eradicate smallpox in a global vaccination campaign in 1958–1977. Although smallpox no longer exists in the wild, vaccinia virus is still studied widely by scientists as a tool for gene therapy and genetic engineering.
Post-exposure prophylaxis, also known as post-exposure prevention (PEP), is any preventive medical treatment started after exposure to a pathogen in order to prevent the infection from occurring.
This is a list of AIDS-related topics, many of which were originally taken from the public domain U.S. Department of Health Glossary of HIV/AIDS-Related Terms, 4th Edition.

Eczema vaccinatum is a rare severe adverse reaction to smallpox vaccination.
Modified vaccinia Ankara (MVA) is an attenuated (weakened) strain of the vaccinia virus. It is being used as a vaccine against smallpox and mpox, having fewer side effects than smallpox vaccines derived from other poxviruses.

X-linked agammaglobulinemia (XLA) is a rare genetic disorder discovered in 1952 that affects the body's ability to fight infection. As the form of agammaglobulinemia that is X-linked, it is much more common in males. In people with XLA, the white blood cell formation process does not generate mature B cells, which manifests as a complete or near-complete lack of proteins called gamma globulins, including antibodies, in their bloodstream. B cells are part of the immune system and normally manufacture antibodies, which defend the body from infections by sustaining a humoral immunity response. Patients with untreated XLA are prone to develop serious and even fatal infections. A mutation occurs at the Bruton's tyrosine kinase (Btk) gene that leads to a severe block in B cell development and a reduced immunoglobulin production in the serum. Btk is particularly responsible for mediating B cell development and maturation through a signaling effect on the B cell receptor BCR. Patients typically present in early childhood with recurrent infections, in particular with extracellular, encapsulated bacteria. XLA is deemed to have a relatively low incidence of disease, with an occurrence rate of approximately 1 in 200,000 live births and a frequency of about 1 in 100,000 male newborns. It has no ethnic predisposition. XLA is treated by infusion of human antibody. Treatment with pooled gamma globulin cannot restore a functional population of B cells, but it is sufficient to reduce the severity and number of infections due to the passive immunity granted by the exogenous antibodies.
Orthopoxvirus is a genus of viruses in the family Poxviridae and subfamily Chordopoxvirinae. Vertebrates, including mammals and humans, and arthropods serve as natural hosts. There are 12 species in this genus. Diseases associated with this genus include smallpox, cowpox, horsepox, camelpox, and mpox. The most widely known member of the genus is Variola virus, which causes smallpox. It was eradicated globally by 1977, through the use of Vaccinia virus as a vaccine. The most recently described species is the Alaskapox virus, first isolated in 2015.
In immunology, passive immunity is the transfer of active humoral immunity of ready-made antibodies. Passive immunity can occur naturally, when maternal antibodies are transferred to the fetus through the placenta, and it can also be induced artificially, when high levels of antibodies specific to a pathogen or toxin are transferred to non-immune persons through blood products that contain antibodies, such as in immunoglobulin therapy or antiserum therapy. Passive immunization is used when there is a high risk of infection and insufficient time for the body to develop its own immune response, or to reduce the symptoms of ongoing or immunosuppressive diseases. Passive immunization can be provided when people cannot synthesize antibodies, and when they have been exposed to a disease that they do not have immunity against.
An attenuated vaccine is a vaccine created by reducing the virulence of a pathogen, but still keeping it viable. Attenuation takes an infectious agent and alters it so that it becomes harmless or less virulent. These vaccines contrast to those produced by "killing" the pathogen.
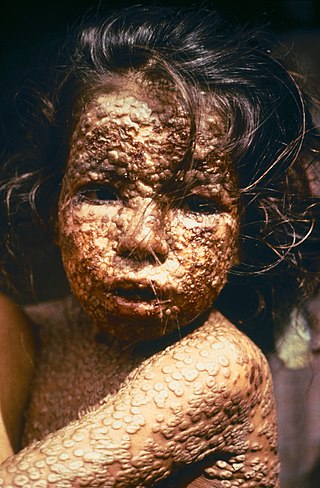
Smallpox was an infectious disease caused by variola virus, which belongs to the genus Orthopoxvirus. The last naturally occurring case was diagnosed in October 1977, and the World Health Organization (WHO) certified the global eradication of the disease in 1980, making smallpox the only human disease to have been eradicated to date.

Progressive vaccinia is a rare cutaneous condition caused by the vaccinia virus, characterized by painless but progressive necrosis and ulceration.

Tecovirimat, sold under the brand name Tpoxx among others, is an antiviral medication with activity against orthopoxviruses such as smallpox and mpox. In 2018 it became the first antipoxviral drug approved in the United States.

The monkeypox virus, is a species of double-stranded DNA virus that causes mpox disease in humans and other mammals. The monkeypox virus is a zoonotic virus belonging to the orthopoxvirus genus, making it closely related to the variola, cowpox, and vaccinia viruses. MPV is oval-shaped with a lipoprotein outer membrane. The genome is approximately 190 kb.

Beginning in May 2003, by July a total of 71 cases of human monkeypox were found in six Midwestern states including Wisconsin, Indiana (16), Illinois (12), Kansas (1), Missouri (2), and Ohio (1). The cause of the outbreak was traced to three species of African rodents imported from Ghana on April 9, 2003, into the United States by an exotic animal importer in Texas. These were shipped from Texas to an Illinois distributor, who housed them with prairie dogs, which then became infected.
Immunoglobulin therapy is the use of a mixture of antibodies to treat several health conditions. These conditions include primary immunodeficiency, immune thrombocytopenic purpura, chronic inflammatory demyelinating polyneuropathy, Kawasaki disease, certain cases of HIV/AIDS and measles, Guillain–Barré syndrome, and certain other infections when a more specific immunoglobulin is not available. Depending on the formulation it can be given by injection into muscle, a vein, or under the skin. The effects last a few weeks.
Raccoonpox virus (RCN) is a double-stranded DNA virus and a member of the orthopoxviruses in the family Poxviridae and subfamily Chordopoxvirinae which consists of eight genera: Avipoxvirus, Capripoxvirus, Leporipoxvirus, Molluscipoxvirus, Orthopoxvirus, Parapoxvirus, Suipoxvirus and Yatapoxvirus Vertebrates are the natural host of Chordopoxvirinae subfamily viruses. More specifically, raccoons are the natural hosts of RCN. RCN was isolated in 1961 from the upper respiratory tissues of 2 raccoons in a group of 92 observably healthy raccoons trapped close to Aberdeen, Maryland.
References
- ↑ "CDC Smallpox | Investigational Vaccinia Immune Globulin (VIG) Information". Bt.cdc.gov. Archived from the original on 2013-05-10. Retrieved 2013-06-16.
- ↑ Wharton, M; Strikas, RA; Harpaz, R; Rotz, LD; Schwartz, B; Casey, CG; Pearson, ML; Anderson, LJ (4 April 2003). "Recommendations for using smallpox vaccine in a pre-event vaccination program. Supplemental recommendations of the Advisory Committee on Immunization Practices (ACIP) and the Healthcare Infection Control Practices Advisory Committee (HICPAC)". MMWR. Recommendations and Reports. 52 (RR-7): 1–16. PMID 12710832.
- ↑ "Vaccinia Immune Globulin IV Monograph for Professionals". Drugs.com.
- ↑ "Monkeypox | Agent Fact Sheet". 17 August 2023.
- ↑ Lane, J.Michael; Goldstein, Joel (July 2003). "Adverse events occurring after smallpox vaccination". Seminars in Pediatric Infectious Diseases. 14 (3): 189–195. doi:10.1016/s1045-1870(03)00032-3. PMID 12913830.
- ↑ Frelinger, JA; Garba, ML (29 August 2002). "Responses to smallpox vaccine". The New England Journal of Medicine. 347 (9): 689–90, author reply 689-90. doi:10.1056/NEJM200208293470913. PMID 12201305.
- ↑ Møller-Larsen, A; Haahr, S (January 1978). "Humoral and cell-mediated immune responses in humans before and after revaccination with vaccinia virus". Infection and Immunity. 19 (1): 34–39. doi:10.1128/iai.19.1.34-39.1978. PMC 414044 . PMID 624594.
- ↑ Cohen, Jon (2 November 2001). "Smallpox Vaccinations: How Much Protection Remains?". Science. 294 (5544): 985. doi:10.1126/science.294.5544.985. PMID 11691969. S2CID 28270372.
- ↑ "Vaccinia immune globulin definition - Medical Dictionary definitions of popular medical terms easily defined on MedTerms". Medterms.com. 2012-06-14. Archived from the original on 2012-08-09. Retrieved 2013-06-16.
- ↑ "CDC Smallpox | Medical Management of Smallpox (Vaccinia) Vaccine Adverse Reactions (Info for Clinicians and Public Health Professionals)". Bt.cdc.gov. Archived from the original on 2013-06-02. Retrieved 2013-06-16.
- ↑ Bray, M. (15 September 2004). "Henry Kempe and the Birth of Vaccinia Immune Globulin". Clinical Infectious Diseases. 39 (6): 767–769. doi: 10.1086/423005 . PMID 15472805.
- ↑ Wittek, Riccardo (1 May 2006). "Vaccinia immune globulin: current policies, preparedness, and product safety and efficacy". International Journal of Infectious Diseases. 10 (3): 193–201. doi: 10.1016/j.ijid.2005.12.001 . PMID 16564720.
- ↑ "DynPort Vaccine Company | GDIT".
- ↑ "DailyMed - CNJ-016 (Vaccinia immune globulin- human injection".
- ↑ Leendertz, Siv; Stern, Daniel; Theophil, Dennis; Anoh, Etile; Mossoun, Arsène; Schubert, Grit; Wiersma, Lidewij; Akoua-Koffi, Chantal; Couacy-Hymann, Emmanuel; Muyembe-Tamfum, Jean-Jacques; Karhemere, Stomy; Pauly, Maude; Schrick, Livia; Leendertz, Fabian; Nitsche, Andreas (29 September 2017). "A Cross-Sectional Serosurvey of Anti-Orthopoxvirus Antibodies in Central and Western Africa". Viruses. 9 (10): 278. doi: 10.3390/v9100278 . PMC 5691630 . PMID 28961172.
- ↑ Clinical trial number NCT01374984 for "VA-005 Clinical Outcomes of VIGIV Treatment of Smallpox Vaccination Complications or Vaccinia Infection" at ClinicalTrials.gov
- ↑ "Medical Management of Adverse Reactions to Vaccinia Virus Vaccination | Smallpox | CDC". 2 November 2021.
- ↑ "Progressive Vaccinia - an overview | ScienceDirect Topics".
- ↑ "VIGIV (Vaccinia immune globulin intravenous) dosing, indications, interactions, adverse effects, and more".
- ↑ Eggleton, J. S.; Nagalli, S. (2022). "Highly Active Antiretroviral Therapy (HAART)". StatPearls. StatPearls. PMID 32119420.
- ↑ Izzedine, Hassane; Launay-Vacher, Vincent; Baumelou, Alain; Deray, Gilbert (August 2004). "Antiretroviral and immunosuppressive drug-drug interactions: An update". Kidney International. 66 (2): 532–541. doi: 10.1111/j.1523-1755.2004.00772.x . PMID 15253704.
- ↑ "Medical Management of Adverse Reactions to Vaccinia Virus Vaccination | Smallpox | CDC". 2 November 2021.
- ↑ Lederman, E. R.; Davidson, W.; Groff, H. L.; Smith, S. K.; Warkentien, T.; Li, Y.; Wilkins, K. A.; Karem, K. L.; Akondy, R. S.; Ahmed, R.; Frace, M.; Shieh, W.-J.; Zaki, S.; Hruby, D. E.; Painter, W. P.; Bergman, K. L.; Cohen, J. I.; Damon, I. K. (1 November 2012). "Progressive Vaccinia: Case Description and Laboratory-Guided Therapy With Vaccinia Immune Globulin, ST-246, and CMX001". Journal of Infectious Diseases. 206 (9): 1372–1385. doi:10.1093/infdis/jis510. PMC 3529603 . PMID 22904336.
- ↑ Laiton-Donato, Katherine; Ávila-Robayo, Paola; Páez-Martinez, Andrés; Benjumea-Nieto, Paula; Usme-Ciro, José A.; Pinzón-Nariño, Nicole; Giraldo, Ivan; Torres-Castellanos, Diego; Nakazawa, Yoshinori; Patel, Nishi; Wilkins, Kimberly; Li, Yu; Davidson, Whitni; Burgado, Jillybeth; Satheshkumar, Panayampalli Subbian; Styczynski, Ashley; Mauldin, Matthew R.; Gracia-Romero, Martha; Petersen, Brett W. (March 2020). "Progressive Vaccinia Acquired through Zoonotic Transmission in a Patient with HIV/AIDS, Colombia". Emerging Infectious Diseases. 26 (3): 601–605. doi:10.3201/eid2603.191365. PMC 7045850 . PMID 32091366.
- ↑ Bray, Mike; Wright, Mary E. (15 March 2003). "Progressive Vaccinia". Clinical Infectious Diseases. 36 (6): 766–774. doi: 10.1086/374244 . PMID 12627361.
- ↑ "Specific Antibody Deficiency | Immune Deficiency Foundation".
- ↑ Earl, Patricia L.; Americo, Jeffrey L.; Wyatt, Linda S.; Anne Eller, Leigh; Montefiori, David C.; Byrum, Russ; Piatak, Michael; Lifson, Jeffrey D.; Rao Amara, Rama; Robinson, Harriet L.; Huggins, John W.; Moss, Bernard (15 September 2007). "Recombinant modified vaccinia virus Ankara provides durable protection against disease caused by an immunodeficiency virus as well as long-term immunity to an orthopoxvirus in a non-human primate". Virology. 366 (1): 84–97. doi:10.1016/j.virol.2007.02.041. PMC 2077303 . PMID 17499326.
- ↑ Berger, Melvin (1 May 2008). "Principles of and Advances in Immunoglobulin Replacement Therapy for Primary Immunodeficiency". Immunology and Allergy Clinics of North America. 28 (2): 413–437. doi:10.1016/j.iac.2008.01.008. PMC 7127239 . PMID 18424340.