A cancer vaccine is a vaccine that either treats existing cancer or prevents development of cancer. Vaccines that treat existing cancer are known as therapeutic cancer vaccines or tumor antigen vaccines. Some of the vaccines are "autologous", being prepared from samples taken from the patient, and are specific to that patient.
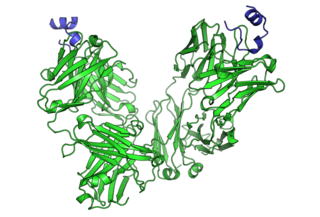
Cancer immunotherapy is the stimulation of the immune system to treat cancer, improving on the immune system's natural ability to fight the disease. It is an application of the fundamental research of cancer immunology and a growing subspecialty of oncology.
An oncolytic virus is a virus that preferentially infects and kills cancer cells. As the infected cancer cells are destroyed by oncolysis, they release new infectious virus particles or virions to help destroy the remaining tumour. Oncolytic viruses are thought not only to cause direct destruction of the tumour cells, but also to stimulate host anti-tumour immune system responses. Oncolytic viruses also have the ability to affect the tumor micro-environment in multiple ways.
Virotherapy is a treatment using biotechnology to convert viruses into therapeutic agents by reprogramming viruses to treat diseases. There are three main branches of virotherapy: anti-cancer oncolytic viruses, viral vectors for gene therapy and viral immunotherapy. These branches use three different types of treatment methods: gene overexpression, gene knockout, and suicide gene delivery. Gene overexpression adds genetic sequences that compensate for low to zero levels of needed gene expression. Gene knockout uses RNA methods to silence or reduce expression of disease-causing genes. Suicide gene delivery introduces genetic sequences that induce an apoptotic response in cells, usually to kill cancerous growths. In a slightly different context, virotherapy can also refer more broadly to the use of viruses to treat certain medical conditions by killing pathogens.

Tumor-infiltrating lymphocytes (TIL) are white blood cells that have left the bloodstream and migrated towards a tumor. They include T cells and B cells and are part of the larger category of ‘tumor-infiltrating immune cells’ which consist of both mononuclear and polymorphonuclear immune cells, in variable proportions. Their abundance varies with tumor type and stage and in some cases relates to disease prognosis.
Tigatuzumab (CS-1008) is a monoclonal antibody for the treatment of cancer. As of October 2009, a clinical trial for the treatment of pancreatic cancer, Phase II trials for colorectal cancer, non-small cell lung cancer, and ovarian cancer have been completed.
Glembatumumab vedotin is an antibody-drug conjugate (ADC) that targets cancer cells expressing transmembrane glycoprotein NMB (GPNMB).
Oncolytics Biotech Inc. is a Canadian company headquartered in Calgary, Alberta, that is developing an intravenously delivered immuno-oncolytic virus called pelareorep for the treatment of solid tumors and hematological malignancies. Pelareorep is a non-pathogenic, proprietary isolate of the unmodified reovirus that: induces selective tumor lysis and promotes an inflamed tumor phenotype through innate and adaptive immune responses.
Pelareorep is a proprietary isolate of the unmodified human reovirus being developed as a systemically administered immuno-oncological viral agent for the treatment of solid tumors and hematological malignancies. Pelareorep is an oncolytic virus, which means that it preferentially lyses cancer cells. Pelareorep also promotes an inflamed tumor phenotype through innate and adaptive immune responses. Preliminary clinical trials indicate that it may have anti-cancer effects across a variety of cancer types when administered alone and in combination with other cancer therapies.
JX-594 is an oncolytic virus is designed to target and destroy cancer cells. It is also known as Pexa-Vec, INN pexastimogene devacirepvec) and was constructed in Dr. Edmund Lattime's lab at Thomas Jefferson University, tested in clinical trials on melanoma patients, and licensed and further developed by SillaJen.
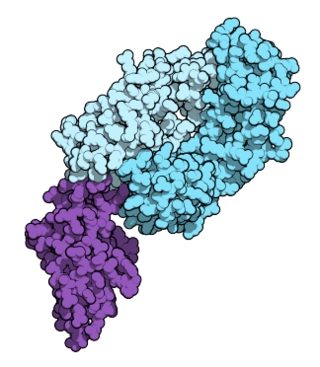
Nivolumab, sold under the brand name Opdivo, is a medication used to treat a number of types of cancer. This includes melanoma, lung cancer, malignant pleural mesothelioma, renal cell carcinoma, Hodgkin lymphoma, head and neck cancer, urothelial carcinoma, colon cancer, esophageal squamous cell carcinoma, liver cancer, gastric cancer, and esophageal or gastroesophageal junction (GEJ) cancer. It is used by slow injection into a vein.

The abscopal effect is a hypothesis in the treatment of metastatic cancer whereby shrinkage of untreated tumors occurs concurrently with shrinkage of tumors within the scope of the localized treatment. R.H. Mole proposed the term “abscopal” in 1953 to refer to effects of ionizing radiation “at a distance from the irradiated volume but within the same organism.”

Talimogene laherparepvec, sold under the brand name Imlygic, is a biopharmaceutical medication used to treat melanoma that cannot be operated on; it is injected directly into a subset of lesions which generates a systemic immune response against the recipient's cancer. The final four year analysis from the pivotal phase 3 study upon which TVEC was approved by the FDA showed a 31.5% response rate with a 16.9% complete response (CR) rate. There was also a substantial and statistically significant survival benefit in patients with earlier metastatic disease and in patients who hadn't received prior systemic treatment for melanoma. The earlier stage group had a reduction in the risk of death of approximately 50% with one in four patients appearing to have met, or be close to be reaching, the medical definition of cure. Real world use of talimogene laherparepvec have shown response rates of up to 88.5% with CR rates of up to 61.5%.

Many variants of herpes simplex virus have been considered for viral therapy of cancer; the early development of these was thoroughly reviewed in the journal Cancer Gene Therapy in 2002. This page describes the most notable variants—those tested in clinical trials: G207, HSV1716, NV1020 and Talimogene laherparepvec. These attenuated versions are constructed by deleting viral genes required for infecting or replicating inside normal cells but not cancer cells, such as ICP34.5, ICP6/UL39, and ICP47.

Pembrolizumab, sold under the brand name Keytruda, is a humanized antibody used in cancer immunotherapy that treats melanoma, lung cancer, head and neck cancer, Hodgkin lymphoma, stomach cancer, cervical cancer, and certain types of breast cancer. It is given by slow injection into a vein.
Eftilagimod alpha is a large-molecule cancer drug being developed by the clinical-stage biotechnology company Immutep. Efti is a soluble version of the immune checkpoint molecule LAG-3. It is an APC Activator used to increase an immune response to tumors, and is administered by subcutaneous injection. Efti has three intended clinical settings:
GL-ONC1 is an investigational therapeutic product consisting of the clinical grade formulation of the laboratory strain GLV-1h68, an oncolytic virus developed by Genelux Corporation. GL-ONC1 is currently under evaluation in Phase I/II human clinical trials in the United States and Europe.
Ieramilimab is a monoclonal antibody being developed by Novartis for the treatment of cancer. The antibody targets the immune checkpoint LAG-3, which is expressed on T cells and tends to down-regulate an immune response. In a June 2015 presentation Novartis management indicated that 'dosing is imminent' for the first clinical trials of LAG525.
Imugene Ltd is a clinical stage immuno-oncology company developing a range of new and novel immunotherapies that seek to activate the immune system of cancer patients to treat and eradicate tumours. Imugene's unique platform technologies seeks to harness the body's immune system against tumours, potentially achieving a similar or greater effect than synthetically manufactured monoclonal antibody and other immunotherapies.
Paul A. Hopper is an Australian bioentrepreneur who has been associated since 2003 with a number of biotechnology companies, most of them publicly traded.





