Drugs used in diabetes treat diabetes mellitus by decreasing the glucose level in the blood. With the exception of insulin, most GLP receptor agonists, and pramlintide, all are administered orally and are thus also called oral hypoglycemic agents or oral antihyperglycemic agents. There are different classes of hypoglycemic drugs, and their selection depends on the nature of diabetes, age, and situation of the person, as well as other factors.

Incretins are a group of metabolic hormones that stimulate a decrease in blood glucose levels. Incretins are released after eating and augment the secretion of insulin released from pancreatic beta cells of the islets of Langerhans by a blood-glucose–dependent mechanism.

Exenatide, sold under the brand name Byetta and Bydureon among others, is a medication used to treat diabetes mellitus type 2. It is used together with diet, exercise, and potentially other antidiabetic medication. It is a treatment option after metformin and sulfonylureas. It is given by injection under the skin twice daily or once weekly.

Vildagliptin, sold under the brand name Galvus and others, is an oral anti-hyperglycemic agent of the dipeptidyl peptidase-4 (DPP-4) inhibitor class of drugs. Vildagliptin inhibits the inactivation of GLP-1 and GIP by DPP-4, allowing GLP-1 and GIP to potentiate the secretion of insulin in the beta cells and suppress glucagon release by the alpha cells of the islets of Langerhans in the pancreas.

Inhibitors of dipeptidyl peptidase 4 are a class of oral hypoglycemics that block the enzyme dipeptidyl peptidase-4 (DPP-4). They can be used to treat diabetes mellitus type 2.

Sitagliptin, sold under the brand name Januvia among others, is an anti-diabetic medication used to treat type 2 diabetes. In the United Kingdom it is listed as less preferred than metformin or a sulfonylurea. It is taken by mouth. It is also available in the fixed-dose combination medication sitagliptin/metformin.

Glucagon-like peptide-1 (GLP-1) is a 30- or 31-amino-acid-long peptide hormone deriving from the tissue-specific posttranslational processing of the proglucagon peptide. It is produced and secreted by intestinal enteroendocrine L-cells and certain neurons within the nucleus of the solitary tract in the brainstem upon food consumption. The initial product GLP-1 (1–37) is susceptible to amidation and proteolytic cleavage, which gives rise to the two truncated and equipotent biologically active forms, GLP-1 (7–36) amide and GLP-1 (7–37). Active GLP-1 protein secondary structure includes two α-helices from amino acid position 13–20 and 24–35 separated by a linker region.
Dipeptidyl peptidase-4, also known as adenosine deaminase complexing protein 2 or CD26 is a protein that, in humans, is encoded by the DPP4 gene. DPP4 is related to FAP, DPP8, and DPP9. The enzyme was discovered in 1966 by Hopsu-Havu and Glenner, and as a result of various studies on chemism, was called dipeptidyl peptidase IV [DP IV].

Saxagliptin, sold under the brand name Onglyza, is an oral hypoglycemic of the dipeptidyl peptidase-4 (DPP-4) inhibitor class. Early development was solely by Bristol-Myers Squibb; in 2007 AstraZeneca joined with Bristol-Myers Squibb to co-develop the final compound and collaborate on the marketing of the drug.

The glucagon-like peptide-1 receptor (GLP1R) is a G protein-coupled receptor (GPCR) found on beta cells of the pancreas and on neurons of the brain. It is involved in the control of blood sugar level by enhancing insulin secretion. In humans it is synthesised by the gene GLP1R, which is present on chromosome 6. It is a member of the glucagon receptor family of GPCRs. GLP1R is composed of two domains, one extracellular (ECD) that binds the C-terminal helix of GLP-1, and one transmembrane (TMD) domain that binds the N-terminal region of GLP-1. In the TMD domain there is a fulcrum of polar residues that regulates the biased signaling of the receptor while the transmembrane helical boundaries and extracellular surface are a trigger for biased agonism.

Linagliptin, sold under the brand name Tradjenta among others, is a medication used to treat type 2 diabetes in conjunction with exercise and diet. It is generally less preferred than metformin and sulfonylureas as an initial treatment. It is taken by mouth.
Dipeptidyl peptidase-4 inhibitors are enzyme inhibitors that inhibit the enzyme dipeptidyl peptidase-4 (DPP-4). They are used in the treatment of type 2 diabetes mellitus. Inhibition of the DPP-4 enzyme prolongs and enhances the activity of incretins that play an important role in insulin secretion and blood glucose control regulation. Type 2 diabetes mellitus is a chronic metabolic disease that results from inability of the β-cells in the pancreas to secrete sufficient amounts of insulin to meet the body's needs. Insulin resistance and increased hepatic glucose production can also play a role by increasing the body's demand for insulin. Current treatments, other than insulin supplementation, are sometimes not sufficient to achieve control and may cause undesirable side effects, such as weight gain and hypoglycemia. In recent years, new drugs have been developed, based on continuing research into the mechanism of insulin production and regulation of the metabolism of sugar in the body. The enzyme DPP-4 has been found to play a significant role.
Albiglutide is a glucagon-like peptide-1 agonist drug marketed by GlaxoSmithKline (GSK) for treatment of type 2 diabetes. As of 2017 it is unclear if it affects a person's risk of death. GSK has announced that it intends to withdraw the drug from the worldwide market by July 2018 for economic reasons.
Glucagon-like peptide-1 (GLP-1) receptor agonists, also known as GLP-1 analogs, are a class of drugs that reduce blood sugar and energy intake by activating the GLP-1 receptor. They mimic the actions of the endogenous incretin hormone GLP-1 that is released by the gut after eating.

Gemigliptin (rINN), sold under the brand name Zemiglo, is an oral anti-hyperglycemic agent of the dipeptidyl peptidase-4 inhibitor class of drugs. Glucose lowering effects of DPP-4 inhibitors are mainly mediated by GLP-1 and gastric inhibitory polypeptide (GIP) incretin hormones which are inactivated by DPP-4.

Teneligliptin is a pharmaceutical drug for the treatment of type 2 diabetes mellitus. It belongs to the class of anti-diabetic drugs known as dipeptidyl peptidase-4 inhibitors or "gliptins".

Dulaglutide, sold under the brand name Trulicity among others, is a medication used for the treatment of type 2 diabetes in combination with diet and exercise. It is also approved in the United States for the reduction of major adverse cardiovascular events in adults with type 2 diabetes who have established cardiovascular disease or multiple cardiovascular risk factors. It is a once-weekly injection.

Trelagliptin is a pharmaceutical drug used for the treatment of type 2 diabetes.
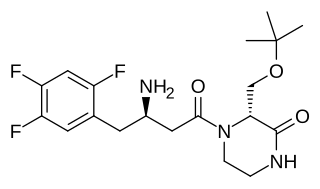
Evogliptin is an antidiabetic drug in the dipeptidyl peptidase-4 (DPP-4) inhibitor or "gliptin" class of drugs. It was developed by the South Korean pharmaceutical company Dong-A ST and is approved for use in South Korea and Russia. In a meta-analysis involving data from 6 randomized controlled trials, Dutta et. al. demonstrated the good glycaemic efficacy and safety of this medicine as compared to other DPP4 inhibitors like sitagliptin and linagliptin.

Gosogliptin is a drug for the treatment of type II diabetes. It is in the class of dipeptidyl peptidase-4 (DPP-4) inhibitors. It was discovered and developed through Phase 1 and Phase 2 by Pfizer. The crystal structure of DPP-4 in complex with gosogliptin is available. Its metabolism, excretion and pharmacokinetics in rat, dog and human have been described. A cost efficient route has been published. Other studies including Phase 3 studies were conducted in Russia. It is approved for use in Russia.














