Zirconium dioxide, sometimes known as zirconia, is a white crystalline oxide of zirconium. Its most naturally occurring form, with a monoclinic crystalline structure, is the mineral baddeleyite. A dopant stabilized cubic structured zirconia, cubic zirconia, is synthesized in various colours for use as a gemstone and a diamond simulant.

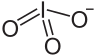
An iodate is the polyatomic anion with the formula IO−3. It is the most common form of iodine in nature, as it comprises the major iodine-containing ores. Iodate salts are often colorless. They are the salts of iodic acid.

Zirconium alloys are solid solutions of zirconium or other metals, a common subgroup having the trade mark Zircaloy. Zirconium has very low absorption cross-section of thermal neutrons, high hardness, ductility and corrosion resistance. One of the main uses of zirconium alloys is in nuclear technology, as cladding of fuel rods in nuclear reactors, especially water reactors. A typical composition of nuclear-grade zirconium alloys is more than 95 weight percent zirconium and less than 2% of tin, niobium, iron, chromium, nickel and other metals, which are added to improve mechanical properties and corrosion resistance.
Zirconium hydride describes an alloy made by combining zirconium and hydrogen. Hydrogen acts as a hardening agent, preventing dislocations in the zirconium atom crystal lattice from sliding past one another. Varying the amount of hydrogen and the form of its presence in the zirconium hydride controls qualities such as the hardness, ductility, and tensile strength of the resulting zirconium hydride. Zirconium hydride with increased hydrogen content can be made harder and stronger than zirconium, but such zirconium hydride is also less ductile than zirconium.

Zirconium carbide (ZrC) is an extremely hard refractory ceramic material, commercially used in tool bits for cutting tools. It is usually processed by sintering.

Zirconium(IV) chloride, also known as zirconium tetrachloride, is an inorganic compound frequently used as a precursor to other compounds of zirconium. This white high-melting solid hydrolyzes rapidly in humid air.

Silver iodate (AgIO3) is a light-sensitive, white crystal composed of silver, iodine and oxygen. Unlike most metal iodates, it is practically insoluble in water.

Sodium iodate (NaIO3) is the sodium salt of iodic acid. Sodium iodate is an oxidizing agent. It has several uses.

Zirconium(IV) fluoride describes members of a family inorganic compounds with the formula (ZrF4(H2O)x. All are colorless, diamagnetic solids. Anhydrous Zirconium(IV) fluoride' is a component of ZBLAN fluoride glass.

Lithium iodate (LiIO3) is a negative uniaxial crystal for nonlinear, acousto-optical and piezoelectric applications. It has been utilized for 347 nm ruby lasers.
Zirconium perchlorate is a molecular substance containing zirconium and perchlorate groups with formula Zr(ClO4)4. Zr(ClO4)4 is a volatile crystalline product. It can be formed by reacting zirconium tetrachloride with dry perchloric acid at liquid nitrogen temperatures. Zr(ClO4)4 sublimes slowly in a vacuum at 70°C showing that the molecule is covalently bound rather than being ionic. The reaction also forms some zirconyl perchlorate (or zirconium oxyperchlorate) ZrO(ClO4)2 as even apparently pure perchloric acid is in equilibrium with dichlorine heptoxide, hydronium ions and perchlorate ions. This side product can be minimised by adding more dichlorine heptoxide or doing the reaction as cold as possible.

Zirconium nitrate is a volatile anhydrous transition metal nitrate salt of zirconium with formula Zr(NO3)4. It has alternate names of zirconium tetranitrate, or zirconium(IV) nitrate.
The iodate fluorides are chemical compounds which contain both iodate and fluoride anions (IO3− and F−). In these compounds fluorine is not bound to iodine as it is in fluoroiodates.
Thulium(II) fluoride is one of the fluoride salts of the lanthanide metal thulium, with the chemical compound of TmF2. It can react with zirconium tetrafluoride at 900 °C to form TmZrF6, which has a hexagonal structure. In addition, low-temperature Mössbauer spectroscopy and some theoretical studies of thulium(II) fluoride have also been reported.
Lanthanum(III) iodate is an inorganic compound with the chemical formula La(IO3)3.
Praseodymium(III) iodate is an inorganic compound with the chemical formula Pr(IO3)3.
Erbium iodate is an inorganic compound with the chemical formula Er(IO3)3.
Ytterbium(III) iodate is an inorganic compound with the chemical formula Yb(IO3)3. Its dihydrate can be prepared by reacting ytterbium sulfate and iodic acid in water at 200 °C. It crystallizes in the P21/c space group, with unit cell parameters a=8.685, b=6.066, c=16.687 Å, β=115.01°.
Samarium iodate is an inorganic compound with the chemical formula Sm(IO3)3.
Europium(III) iodate is an inorganic compound with the chemical formula Eu(IO3)3. It can be produced by hydrothermal reaction of europium(III) nitrate or europium(III) oxide and iodic acid in water at 230 °C. It can be thermally decomposed as follows: