Related Research Articles

Chlorophyll is any of several related green pigments found in cyanobacteria and in the chloroplasts of algae and plants. Its name is derived from the Greek words χλωρός, khloros and φύλλον, phyllon ("leaf"). Chlorophyll allow plants to absorb energy from light.

Photosynthesis is a process used by plants and other organisms to convert light energy into chemical energy that, through cellular respiration, can later be released to fuel the organism's activities. Some of this chemical energy is stored in carbohydrate molecules, such as sugars and starches, which are synthesized from carbon dioxide and water – hence the name photosynthesis, from the Greek phōs, "light", and synthesis, "putting together". Most plants, algae, and cyanobacteria perform photosynthesis; such organisms are called photoautotrophs. Photosynthesis is largely responsible for producing and maintaining the oxygen content of the Earth's atmosphere, and supplies most of the energy necessary for life on Earth.

A photosynthetic pigment is a pigment that is present in chloroplasts or photosynthetic bacteria and captures the light energy necessary for photosynthesis.
Phycobilins are light-capturing bilins found in cyanobacteria and in the chloroplasts of red algae, glaucophytes and some cryptomonads. Most of their molecules consist of a chromophore which makes them coloured. They are unique among the photosynthetic pigments in that they are bonded to certain water-soluble proteins, known as phycobiliproteins. Phycobiliproteins then pass the light energy to chlorophylls for photosynthesis.
Fucoxanthin is a xanthophyll, with formula C42H58O6. It is found as an accessory pigment in the chloroplasts of brown algae and most other heterokonts, giving them a brown or olive-green color. Fucoxanthin absorbs light primarily in the blue-green to yellow-green part of the visible spectrum, peaking at around 510-525 nm by various estimates and absorbing significantly in the range of 450 to 540 nm.
Phycoerythrin (PE) is a red protein-pigment complex from the light-harvesting phycobiliprotein family, present in cyanobacteria, red algae and cryptophytes, accessory to the main chlorophyll pigments responsible for photosynthesis.The red pigment is due to the prosthetic group, phycoerythrobilin, which gives phycoerythrin its red color.

Photosystems are functional and structural units of protein complexes involved in photosynthesis. Together they carry out the primary photochemistry of photosynthesis: the absorption of light and the transfer of energy and electrons. Photosystems are found in the thylakoid membranes of plants, algae, and cyanobacteria. These membranes are located inside the chloroplasts of plants and algae, and in the cytoplasmic membrane of photosynthetic bacteria. There are two kinds of photosystems: PSI and PSII.

Photosystem I is one of two photosystems in the photosynthetic light reactions of algae, plants, and cyanobacteria. Photosystem I is an integral membrane protein complex that uses light energy to catalyze the transfer of electrons across the thylakoid membrane from plastocyanin to ferredoxin. Ultimately, the electrons that are transferred by Photosystem I are used to produce the moderate-energy hydrogen carrier NADPH. The photon energy absorbed by Photosystem I also produces a proton-motive force that is used to generate ATP. PSI is composed of more than 110 cofactors, significantly more than Photosystem II.
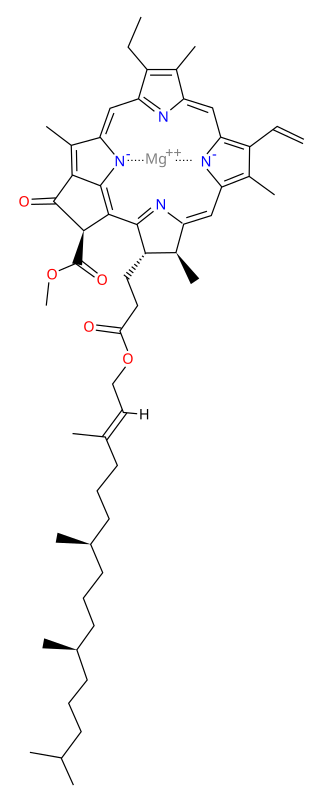
Chlorophyll a is a specific form of chlorophyll used in oxygenic photosynthesis. It absorbs most energy from wavelengths of violet-blue and orange-red light, and it is a poor absorber of green and near-green portions of the spectrum. Chlorophyll does not reflect light but chlorophyll-containing tissues appear green because green light is diffusively reflected by structures like cell walls. This photosynthetic pigment is essential for photosynthesis in eukaryotes, cyanobacteria and prochlorophytes because of its role as primary electron donor in the electron transport chain. Chlorophyll a also transfers resonance energy in the antenna complex, ending in the reaction center where specific chlorophylls P680 and P700 are located.

Phycobilisomes are light harvesting antennae of photosystem II in cyanobacteria, red algae and glaucophytes. It was lost in the plastids of green algae / plants (chloroplasts).
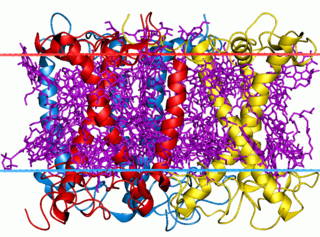
The light-harvesting complex is an array of protein and chlorophyll molecules embedded in the thylakoid membrane of plants and cyanobacteria, which transfer light energy to one chlorophyll a molecule at the reaction center of a photosystem.
Photodissociation, photolysis, photodecomposition, or photofragmentation is a chemical reaction in which molecules of a chemical compound are broken down by photons. It is defined as the interaction of one or more photons with one target molecule.
A photosynthetic reaction center is a complex of several proteins, pigments and other co-factors that together execute the primary energy conversion reactions of photosynthesis. Molecular excitations, either originating directly from sunlight or transferred as excitation energy via light-harvesting antenna systems, give rise to electron transfer reactions along the path of a series of protein-bound co-factors. These co-factors are light-absorbing molecules (also named chromophores or pigments) such as chlorophyll and pheophytin, as well as quinones. The energy of the photon is used to excite an electron of a pigment. The free energy created is then used, via a chain of nearby electron acceptors, for a transfer of hydrogen atoms (as protons and electrons) from H2O or hydrogen sulfide towards carbon dioxide, eventually producing glucose. These electron transfer steps ultimately result in the conversion of the energy of photons to chemical energy.
A light-harvesting complex consists of a number of chromophores which are complex subunit proteins that may be part of a larger super complex of a photosystem, the functional unit in photosynthesis. It is used by plants and photosynthetic bacteria to collect more of the incoming light than would be captured by the photosynthetic reaction center alone. The light which is captured by the chromophores is capable of exciting molecules from their ground state to a higher energy state, known as the excited state. This excited state does not last very long and is known to be short-lived.
Photoprotection is the biochemical process that helps organisms cope with molecular damage caused by sunlight. Plants and other oxygenic phototrophs have developed a suite of photoprotective mechanisms to prevent photoinhibition and oxidative stress caused by excess or fluctuating light conditions. Humans and other animals have also developed photoprotective mechanisms to avoid UV photodamage to the skin, prevent DNA damage, and minimize the downstream effects of oxidative stress.

Biological pigments, also known simply as pigments or biochromes, are substances produced by living organisms that have a color resulting from selective color absorption. Biological pigments include plant pigments and flower pigments. Many biological structures, such as skin, eyes, feathers, fur and hair contain pigments such as melanin in specialized cells called chromatophores. In some species, pigments accrue over very long periods during an individual's lifespan.
Non-photochemical quenching (NPQ) is a mechanism employed by plants and algae to protect themselves from the adverse effects of high light intensity. It involves the quenching of singlet excited state chlorophylls (Chl) via enhanced internal conversion to the ground state, thus harmlessly dissipating excess excitation energy as heat through molecular vibrations. NPQ occurs in almost all photosynthetic eukaryotes, and helps to regulate and protect photosynthesis in environments where light energy absorption exceeds the capacity for light utilization in photosynthesis.

Light-dependent reactions is jargon for certain photochemical reactions that are involved in photosynthesis, the main process by which plants acquire energy. There are two light dependent reactions, the first occurs at photosystem II (PSII) and the second occurs at photosystem I (PSI),

Bacterial anoxygenic photosynthesis differs from the better known oxygenic photosynthesis in plants by the reductant used and the byproduct generated.

Biliproteins are pigment protein compounds that are located in photosynthesising organisms such as algae and certain insects. They refer to any protein that contains a bilin chromophore. In plants and algae, the main function of biliproteins is to make the process of light accumulation required for photosynthesis more efficient; while in insects they play a role in growth and development. Some of their properties: including light-receptivity, light-harvesting and fluorescence have made them suitable for applications in bioimaging and as indicators; while other properties such as anti-oxidation, anti-aging and anti-inflammation in phycobiliproteins have given them potential for use in medicine, cosmetics and food technology. While research on biliproteins dates back as far as 1950, it was hindered due to issues regarding biliprotein structure, lack of methods available for isolating individual biliprotein components, as well as limited information on lyase reactions . Research on biliproteins has also been primarily focused on phycobiliproteins; but advances in technology and methodology, along with the discovery of different types of lyases, has renewed interest in biliprotein research, allowing new opportunities for investigating biliprotein processes such as assembly/disassembly and protein folding.
References
- ↑ McElroy, J Scot; Kopsell, Dean (2009). "Physiological role of carotenoids and other antioxidants in plants and application to turfgrass stress management". Zealand Journal of Crop and Horticultural Science. 37 (4): 327–33. doi: 10.1080/01140671.2009.9687587 .
- ↑ Falkowski, Paul; Raven, John (October 31, 2013). "2". Aquatic Photosynthesis (2 ed.). Princeton University Press. p. 80. ISBN 978-1400849727 . Retrieved June 8, 2018.
- ↑ Hanelt, Dieter; Wiencke, Christian; Bischof, Kai (November 30, 2003). "18". In Larkum, Anthony; Douglas, Susan; Raven, John (eds.). Photosynthesis in Algae. Advances in Photosynthesis and Respiration. Vol. 14. Springer Science & Business Media. p. 417. doi:10.1007/978-94-007-1038-2. ISBN 978-0792363330. ISSN 1572-0233. S2CID 45648608 . Retrieved June 8, 2018.
- ↑ M. Stomp; J. Huisman; L.J. Stal; H.C. Matthijs (August 2007). "Colorful niches of phototrophic microorganisms shaped by vibrations of the water molecule". ISME J. 1 (4): 271–282. doi: 10.1038/ismej.2007.59 . PMID 18043638.