
Turmeric or Curcuma longa, is a flowering plant in the ginger family Zingiberaceae. It is a perennial, rhizomatous, herbaceous plant native to the Indian subcontinent and Southeast Asia that requires temperatures between 20 and 30 °C and high annual rainfall to thrive. Plants are gathered each year for their rhizomes, some for propagation in the following season and some for consumption.

Curcumin is a bright yellow chemical produced by plants of the Curcuma longa species. It is the principal curcuminoid of turmeric, a member of the ginger family, Zingiberaceae. It is sold as a herbal supplement, cosmetics ingredient, food flavoring, and food coloring.

Cyclodextrins are a family of cyclic oligosaccharides, consisting of a macrocyclic ring of glucose subunits joined by α-1,4 glycosidic bonds. Cyclodextrins are produced from starch by enzymatic conversion. They are used in food, pharmaceutical, drug delivery, and chemical industries, as well as agriculture and environmental engineering.

Rosocyanine and rubrocurcumin are two red colored materials, which are formed by the reaction between curcumin and borates.

Pinosylvin is an organic compound with the formula C6H5CH=CHC6H3(OH)2. A white solid, it is related to trans-stilbene, but with two hydroxy groups on one of the phenyl substituents. It is very soluble in many organic solvents, such as acetone.

The adenosine A2B receptor, also known as ADORA2B, is a G-protein coupled adenosine receptor, and also denotes the human adenosine A2b receptor gene which encodes it.

ADX-47273 is a research pharmaceutical developed by Addex Therapeutics which acts as a positive allosteric modulator (PAM) selective for the metabotropic glutamate receptor subtype mGluR5. It has nootropic and antipsychotic effects in animal studies, and has been used as a lead compound to develop improved derivatives.

Arylcyclohexylamines, also known as arylcyclohexamines or arylcyclohexanamines, are a chemical class of pharmaceutical, designer, and experimental drugs.

DS-1 is a drug from the imidazopyridine family, which is the first drug developed that acts as a GABAA receptor positive allosteric modulator (PAM) selective for the α4β3δ subtype, which is not targeted by other GABAA receptor PAMs such as the benzodiazepines or other nonbenzodiazepine drugs. Novel selective drugs such as DS-1 are useful in the study of this receptor subtype.
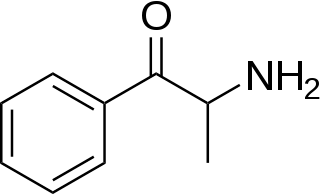
Substituted cathinones, which include some stimulants and entactogens, are derivatives of cathinone. They feature a phenethylamine core with an alkyl group attached to the alpha carbon, and a ketone group attached to the beta carbon, along with additional substitutions. Cathinone occurs naturally in the plant khat whose leaves are chewed as a recreational drug.

Rhodanine is a 5-membered heterocyclic organic compound possessing a thiazolidine core. It was discovered in 1877 by Marceli Nencki who named it "Rhodaninsaure" in reference to its synthesis from ammonium rhodanide and chloroacetic acid in water.
Desmethoxycurcumin is a curcuminoid found in turmeric. Commercial grade curcumin contains a mixture of curcuminoids.

The diarylheptanoids are polyphenols and a relatively small class of plant secondary metabolites. Diarylheptanoids consist of two aromatic rings joined by a seven carbons chain (heptane) and having various substituents. They can be classified into linear (curcuminoids) and cyclic diarylheptanoids. The best known member is curcumin, which is isolated from turmeric and is known as food coloring E100. Some other Curcuma species, such as Curcuma comosa also produce diarylheptanoids.
1,7-Bis(4-hydroxyphenyl)-1,4,6-heptatrien-3-one is a natural product, a curcuminoid antioxidant found in turmeric and torch ginger.
Curcumin synthase categorizes three enzyme isoforms, type III polyketide synthases (PKSs) present in the leaves and rhizome of the turmeric plant that synthesize curcumin. CURS1-3 are responsible for the hydrolysis of feruloyldiketide-CoA, previously produced in the curcuminoid pathway, and a decarboxylative condensation reaction that together comprise one of the final steps in the synthesis pathway for curcumin, demethoxycurcumin, and bisdemethoxycurcumin, the compounds that give turmeric both its distinctive yellow color, and traditional medical benefits. CURS should not be confused with Curcuminoid Synthase (CUS), which catalyzes the one-pot synthesis of bisdemethoxycurcumin in Oryza sativa.

The ProTide technology is a prodrug approach used in molecular biology and drug design. It is designed to deliver nucleotide analogues into the cell. This technology was invented by Professor Chris McGuigan from the School of Pharmacy and Pharmaceutical Sciences at Cardiff University in the early 1990s. ProTides form a critical part of antiviral drugs such as sofosbuvir, tenofovir alafenamide, and remdesivir.
Pan-assay interference compounds (PAINS) are chemical compounds that often give false positive results in high-throughput screens. PAINS tend to react nonspecifically with numerous biological targets rather than specifically affecting one desired target. A number of disruptive functional groups are shared by many PAINS.

The substituted benzofurans are a class of chemical compounds based on the heterocyclyc and polycyclic compound benzofuran. Many medicines use the benzofuran core as a scaffold, but most commonly the term is used to refer to the simpler compounds in this class which include numerous psychoactive drugs, including stimulants, psychedelics and empathogens. In general, these compounds have a benzofuran core to which a 2-aminoethyl group is attached, and combined with a range of other substituents. Some psychoactive derivatives from this family have been sold under the name Benzofury.

para-Aminoblebbistatin is a water-soluble, non-fluorescent, photostable myosin II inhibitor, developed from blebbistatin. Among the several blebbistatin derivatives it is one of the most promising for research applications. Furthermore, it has a favourable overall profile considering inhibitory properties and ADME calculations.

EF-24 is a compound that is a synthetic analogue of curcumin, a bioactive phytochemical from turmeric. Curcumin has antioxidant, antibiotic, anti-inflammatory and anti-cancer properties in vitro but has low potency and very poor bioavailability when taken orally, resulting in limited efficacy. EF-24 was developed to try to improve upon these properties, and has been found to be around 10x more potent than curcumin and with much higher systemic bioavailability. It has never been developed for medical use, though research continues to investigate whether it may be useful as an adjuvant treatment for some cancers alongside conventional chemotherapy drugs.


















