
Dabur Ltd is an Indian multinational consumer goods company, founded by S. K. Burman and headquartered in Ghaziabad. It manufactures Ayurvedic products and fast-moving consumer goods. Dabur derives around 60% of its revenue from the consumer care business, 11% from the food business and remaining from the international business unit.
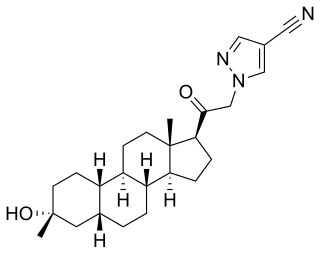
Neurosteroids, also known as neuroactive steroids, are endogenous or exogenous steroids that rapidly alter neuronal excitability through interaction with ligand-gated ion channels and other cell surface receptors. The term neurosteroid was coined by the French physiologist Étienne-Émile Baulieu and refers to steroids synthesized in the brain. The term, neuroactive steroid refers to steroids that can be synthesized in the brain, or are synthesized by an endocrine gland, that then reach the brain through the bloodstream and have effects on brain function. The term neuroactive steroids was first coined in 1992 by Steven Paul and Robert Purdy. In addition to their actions on neuronal membrane receptors, some of these steroids may also exert effects on gene expression via nuclear steroid hormone receptors. Neurosteroids have a wide range of potential clinical applications from sedation to treatment of epilepsy and traumatic brain injury. Ganaxolone, a synthetic analog of the endogenous neurosteroid allopregnanolone, is under investigation for the treatment of epilepsy.

The metabotropic glutamate receptors, or mGluRs, are a type of glutamate receptor that are active through an indirect metabotropic process. They are members of the group C family of G-protein-coupled receptors, or GPCRs. Like all glutamate receptors, mGluRs bind with glutamate, an amino acid that functions as an excitatory neurotransmitter.
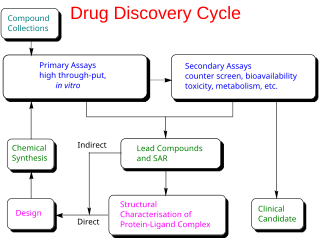
Drug development is the process of bringing a new pharmaceutical drug to the market once a lead compound has been identified through the process of drug discovery. It includes preclinical research on microorganisms and animals, filing for regulatory status, such as via the United States Food and Drug Administration for an investigational new drug to initiate clinical trials on humans, and may include the step of obtaining regulatory approval with a new drug application to market the drug. The entire process—from concept through preclinical testing in the laboratory to clinical trial development, including Phase I–III trials—to approved vaccine or drug typically takes more than a decade.

The C3a receptor also known as complement component 3a receptor 1 (C3AR1) is a G protein-coupled receptor protein involved in the complement system.

Drug delivery refers to approaches, formulations, manufacturing techniques, storage systems, and technologies involved in transporting a pharmaceutical compound to its target site to achieve a desired therapeutic effect. Principles related to drug preparation, route of administration, site-specific targeting, metabolism, and toxicity are used to optimize efficacy and safety, and to improve patient convenience and compliance. Drug delivery is aimed at altering a drug's pharmacokinetics and specificity by formulating it with different excipients, drug carriers, and medical devices. There is additional emphasis on increasing the bioavailability and duration of action of a drug to improve therapeutic outcomes. Some research has also been focused on improving safety for the person administering the medication. For example, several types of microneedle patches have been developed for administering vaccines and other medications to reduce the risk of needlestick injury.

Tinospora cordifolia is a herbaceous vine of the family Menispermaceae indigenous to tropical regions of the Indian subcontinent. It has been used in Ayurveda to treat various disorders.

Metabotropic glutamate receptor 5 is an excitatory Gq-coupled G protein-coupled receptor predominantly expressed on the postsynaptic sites of neurons. In humans, it is encoded by the GRM5 gene.

Adenosine kinase is an enzyme that catalyzes the transfer of gamma-phosphate from Adenosine triphosphate (ATP) to adenosine (Ado) leading to formation of Adenosine monophosphate (AMP). In addition to its well-studied role in controlling the cellular concentration of Ado, AdK also plays an important role in the maintenance of methylation reactions. All S-adenosylmethionine-dependent transmethylation reactions in cells lead to production of S-adenosylhomocysteine (SAH), which is cleaved by SAH hydrolase into Ado and homocysteine. The failure to efficiently remove these end products can result in buildup of SAH, which is a potent inhibitor of all transmethylation reactions. The disruption of AdK gene (-/-) in mice causes neonatal hepatic steatosis, a fatal condition characterized by rapid microvesicular fat infiltration, leading to early postnatal death. The liver was the main organ affected in these animals and in it the levels of adenine nucleotides were decreased, while those of SAH were elevated. Recently, missense mutations in the AdK gene in humans which result in AdK deficiency have also been shown to cause hypermethioninemia, encephalopathy and abnormal liver function.

Sulfotransferase 1A1 is an enzyme that in humans is encoded by the SULT1A1 gene.

PHD finger protein 8 is a protein that in humans is encoded by the PHF8 gene.

Lipid nanoparticles (LNPs) are very small spherical particles composed of lipids. They are a novel pharmaceutical drug delivery system, and a novel pharmaceutical formulation. Using LNPs for drug delivery was first approved in 2018 for the siRNA drug Onpattro. LNPs became more widely known in late 2020, as some COVID-19 vaccines that use RNA vaccine technology coat the fragile mRNA strands with PEGylated lipid nanoparticles as their delivery vehicle.

CSIR Institute of Genomics and Integrative Biology (CSIR-IGIB) is a scientific research institute devoted primarily to biological research. It is a part of Council of Scientific and Industrial Research (CSIR), India.

Candocuronium iodide is an aminosteroid neuromuscular-blocking drug. Its use within anesthesia for endotracheal intubation and for providing skeletal muscle relaxation during surgery or mechanical ventilation was briefly evaluated in clinical studies in India, though further development was discontinued due to attendant cardiovascular effects, primarily tachycardia that was about the same as the clinically established pancuronium bromide. Candocuronium demonstrated a short duration in the body, but a rapid onset of action. It had little to no ganglion blocking activity, with a greater potency than pancuronium.
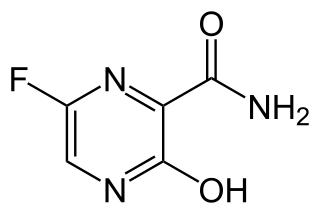
Favipiravir, sold under the brand name Avigan among others, is an antiviral medication used to treat influenza in Japan. It is also being studied to treat a number of other viral infections, including SARS-CoV-2. Like the experimental antiviral drugs T-1105 and T-1106, it is a pyrazinecarboxamide derivative.
Virander Singh Chauhan is an Indian scientist and a Rhodes Scholar working in the fields of genetic engineering and biotechnology. He is known for his contributions to the development of a recombinant vaccine for malaria. and for synthetic structural peptides with biological functions. He was honored by the Government of India in 2012 with the fourth highest Indian civilian award of Padma Shri. He is the present Chancellor of the Gandhi Institute of Technology and Management.

Arketamine (developmental code names PCN-101, HR-071603), also known as (R)-ketamine or (R)-(−)-ketamine, is the (R)-(−) enantiomer of ketamine. Similarly to racemic ketamine and esketamine, the S(+) enantiomer of ketamine, arketamine is biologically active; however, it is less potent as an NMDA receptor antagonist and anesthetic and thus has never been approved or marketed for clinical use as an enantiopure drug. Arketamine is currently in clinical development as a novel antidepressant.

Brequinar (DuP-785) is a drug that acts as a potent and selective inhibitor of the enzyme dihydroorotate dehydrogenase. It blocks synthesis of pyrimidine based nucleotides in the body and so inhibits cell growth. Brequinar was invented by DuPont Pharmaceuticals in the 1980s. In 2001, Bristol-Myers Squibb acquired DuPont, and in 2017, Clear Creek Bio acquired the rights to brequinar from BMS.

An mRNAvaccine is a type of vaccine that uses a copy of a molecule called messenger RNA (mRNA) to produce an immune response. The vaccine delivers molecules of antigen-encoding mRNA into cells, which use the designed mRNA as a blueprint to build foreign protein that would normally be produced by a pathogen or by a cancer cell. These protein molecules stimulate an adaptive immune response that teaches the body to identify and destroy the corresponding pathogen or cancer cells. The mRNA is delivered by a co-formulation of the RNA encapsulated in lipid nanoparticles that protect the RNA strands and help their absorption into the cells.

Toreforant (JNJ-38518168) is an orally-dosed selective antagonist of the histamine H4 receptor that has been studied for various health conditions. It is the successor of a number of H4-selective compounds developed by Johnson & Johnson. Phase IIa clinical trials completed as recently as November 2018 continue to suggest that toreforant is safe.



















