Related Research Articles

The cytoskeleton is a complex, dynamic network of interlinking protein filaments present in the cytoplasm of all cells, including those of bacteria and archaea. In eukaryotes, it extends from the cell nucleus to the cell membrane and is composed of similar proteins in the various organisms. It is composed of three main components: microfilaments, intermediate filaments, and microtubules, and these are all capable of rapid growth or disassembly depending on the cell's requirements.

Microvilli are microscopic cellular membrane protrusions that increase the surface area for diffusion and minimize any increase in volume, and are involved in a wide variety of functions, including absorption, secretion, cellular adhesion, and mechanotransduction.

Microfilaments, also called actin filaments, are protein filaments in the cytoplasm of eukaryotic cells that form part of the cytoskeleton. They are primarily composed of polymers of actin, but are modified by and interact with numerous other proteins in the cell. Microfilaments are usually about 7 nm in diameter and made up of two strands of actin. Microfilament functions include cytokinesis, amoeboid movement, cell motility, changes in cell shape, endocytosis and exocytosis, cell contractility, and mechanical stability. Microfilaments are flexible and relatively strong, resisting buckling by multi-piconewton compressive forces and filament fracture by nanonewton tensile forces. In inducing cell motility, one end of the actin filament elongates while the other end contracts, presumably by myosin II molecular motors. Additionally, they function as part of actomyosin-driven contractile molecular motors, wherein the thin filaments serve as tensile platforms for myosin's ATP-dependent pulling action in muscle contraction and pseudopod advancement. Microfilaments have a tough, flexible framework which helps the cell in movement.
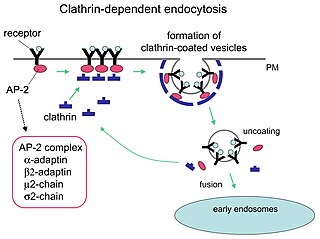
Receptor-mediated endocytosis (RME), also called clathrin-mediated endocytosis, is a process by which cells absorb metabolites, hormones, proteins – and in some cases viruses – by the inward budding of the plasma membrane (invagination). This process forms vesicles containing the absorbed substances and is strictly mediated by receptors on the surface of the cell. Only the receptor-specific substances can enter the cell through this process.
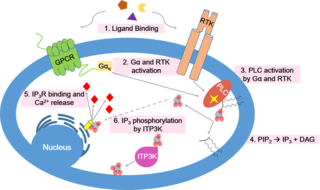
Calcium signaling is the use of calcium ions (Ca2+) to communicate and drive intracellular processes often as a step in signal transduction. Ca2+ is important for cellular signalling, for once it enters the cytosol of the cytoplasm it exerts allosteric regulatory effects on many enzymes and proteins. Ca2+ can act in signal transduction resulting from activation of ion channels or as a second messenger caused by indirect signal transduction pathways such as G protein-coupled receptors.
Cell migration is a central process in the development and maintenance of multicellular organisms. Tissue formation during embryonic development, wound healing and immune responses all require the orchestrated movement of cells in particular directions to specific locations. Cells often migrate in response to specific external signals, including chemical signals and mechanical signals. Errors during this process have serious consequences, including intellectual disability, vascular disease, tumor formation and metastasis. An understanding of the mechanism by which cells migrate may lead to the development of novel therapeutic strategies for controlling, for example, invasive tumour cells.
The cell cortex, also known as the actin cortex, cortical cytoskeleton or actomyosin cortex, is a specialized layer of cytoplasmic proteins on the inner face of the cell membrane. It functions as a modulator of membrane behavior and cell surface properties. In most eukaryotic cells lacking a cell wall, the cortex is an actin-rich network consisting of F-actin filaments, myosin motors, and actin-binding proteins. The actomyosin cortex is attached to the cell membrane via membrane-anchoring proteins called ERM proteins that plays a central role in cell shape control. The protein constituents of the cortex undergo rapid turnover, making the cortex both mechanically rigid and highly plastic, two properties essential to its function. In most cases, the cortex is in the range of 100 to 1000 nanometers thick.

Major sperm protein (MSP) is a nematode specific small protein of 126 amino acids with a molecular weight of 14 kDa. It is the key player in the motility machinery of nematodes that propels the crawling movement/motility of nematode sperm. It is the most abundant protein present in nematode sperm, comprising 15% of the total protein and more than 40% of the soluble protein. MSP is exclusively synthesized in spermatocytes of the nematodes. The MSP has two main functions in the reproduction of the helminthes: i) as cytosolic component it is responsible for the crawling movement of the mature sperm, and ii) once released, it acts as hormone on the female germ cells, where it triggers oocyte maturation and stimulates the oviduct wall to contract to bring the oocytes into position for fertilization. MSP has first been identified in Caenorhabditis elegans.
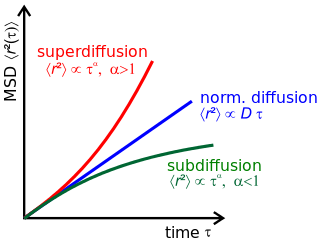
Anomalous diffusion is a diffusion process with a non-linear relationship between the mean squared displacement (MSD), , and time. This behavior is in stark contrast to Brownian motion, the typical diffusion process described by Einstein and Smoluchowski, where the MSD is linear in time.

In cell biology, a bleb is a bulge of the plasma membrane of a cell, characterized by a spherical, "blister-like", bulky morphology. It is characterized by the decoupling of the cytoskeleton from the plasma membrane, degrading the internal structure of the cell, allowing the flexibility required for the cell to separate into individual bulges or pockets of the intercellular matrix. Most commonly, blebs are seen in apoptosis but are also seen in other non-apoptotic functions, including apocrine secretion. Blebbing, or zeiosis, is the formation of blebs.

Erich Sackmann is a German experimental physicist and a pioneer of biophysics in Europe.
Single-particle tracking (SPT) is the observation of the motion of individual particles within a medium. The coordinates time series, which can be either in two dimensions (x, y) or in three dimensions (x, y, z), is referred to as a trajectory. The trajectory is typically analyzed using statistical methods to extract information about the underlying dynamics of the particle. These dynamics can reveal information about the type of transport being observed (e.g., thermal or active), the medium where the particle is moving, and interactions with other particles. In the case of random motion, trajectory analysis can be used to measure the diffusion coefficient.
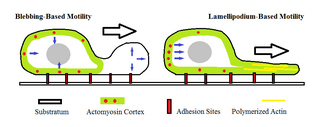
Amoeboid movement is the most typical mode of locomotion in adherent eukaryotic cells. It is a crawling-like type of movement accomplished by protrusion of cytoplasm of the cell involving the formation of pseudopodia ("false-feet") and posterior uropods. One or more pseudopodia may be produced at a time depending on the organism, but all amoeboid movement is characterized by the movement of organisms with an amorphous form that possess no set motility structures.

Eli Barkai is a professor of physics at Bar-Ilan University, located in Ramat-Gan, Israel.
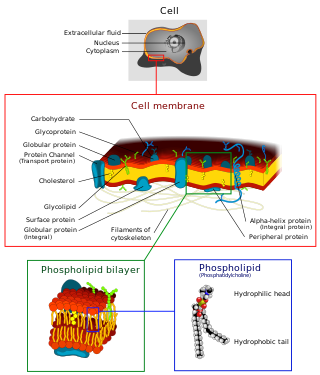
The cell membrane is a biological membrane that separates and protects the interior of a cell from the outside environment. The cell membrane consists of a lipid bilayer, made up of two layers of phospholipids with cholesterols interspersed between them, maintaining appropriate membrane fluidity at various temperatures. The membrane also contains membrane proteins, including integral proteins that span the membrane and serve as membrane transporters, and peripheral proteins that loosely attach to the outer (peripheral) side of the cell membrane, acting as enzymes to facilitate interaction with the cell's environment. Glycolipids embedded in the outer lipid layer serve a similar purpose. The cell membrane controls the movement of substances in and out of a cell, being selectively permeable to ions and organic molecules. In addition, cell membranes are involved in a variety of cellular processes such as cell adhesion, ion conductivity, and cell signalling and serve as the attachment surface for several extracellular structures, including the cell wall and the carbohydrate layer called the glycocalyx, as well as the intracellular network of protein fibers called the cytoskeleton. In the field of synthetic biology, cell membranes can be artificially reassembled.
The fences and pickets model of plasma membrane is a concept of cell membrane structure suggesting that the fluid plasma membrane is compartmentalized by actin-based membrane-skeleton "fences" and anchored transmembrane protein "pickets". This model differs from older cell membrane structure concepts such as the Singer-Nicolson fluid mosaic model and the Saffman-Delbrück two-dimensional continuum fluid model that view the membrane as more or less homogeneous. The fences and pickets model was proposed to explain observations of molecular traffic made due to recent advances in single molecule tracking techniques. However, it has been suggested that this model mischaracterizes protein islands, which explain these observations but need not have transmembrane proteins in a picket-like arrangement.
Hop diffusion - is a non-Brownian diffusion of proteins and lipid molecules within the plasma membrane. Hop diffusion occurs due to the discontinuity of the cell cytoplasmic membrane. According to the fences and pickets model, plasma membrane is compartmentalized by actin-based membrane-skeleton “fences”, that occur when cytoplasmic domains collide with the actin-based membrane skeleton; and anchored-transmembrane protein “pickets”. Due to these obstacles membrane proteins undergo temporary confinement within 30–700- nm compartments with infrequent intercompartmental hops. However, according to Kervin and Overduin's model, this diffusion pattern can be explained by the existence of protein islands, which may but need not depend on the cytoskeleton.
Kathryn Rachel Ayscough is a professor of molecular cell biology and head of the department of biomedical science at the University of Sheffield. She was awarded the 2002 Society for Experimental Biology President's Medal. Her research investigates the role of the actin cytoskeleton in membrane trafficking and cell organisation.
Ralf Metzler is a physicist that focuses on nonequilibrium statistical physics and anomalous stochastic processes, with applications to biological and soft matter systems. He currently is chair professor for theoretical physics at the University of Potsdam and is an Alexander von Humboldt Polish Honorary Research Fellow.

David G. Drubin is an American biologist, academic, and researcher. He is a Distinguished Professor of Cell and Developmental Biology at the University of California, Berkeley where he holds the Ernette Comby Chair in Microbiology.
References
- ↑ "Cees Dekker Lab – People" . Retrieved 27 March 2021.
- ↑ "How Nature Controls Traffic on the Surface of Cells". Physics central. 9 March 2017. Archived from the original on 2017-03-14. Retrieved 27 March 2021.
- ↑ "A traffic cop for the cell surface: Researchers illuminate a basic biological process". 27 February 2017. Archived from the original on 2017-02-27. Retrieved 27 March 2021.
- ↑ Weigel AV, Simon B, Tamkun MM, Krapf D (April 2011). "Ergodic and nonergodic processes coexist in the plasma membrane as observed by single-molecule tracking". Proceedings of the National Academy of Sciences of the United States of America. 108 (16): 6438–43. Bibcode:2011PNAS..108.6438W. doi: 10.1073/pnas.1016325108 . PMC 3081000 . PMID 21464280.
- ↑ Barkai E, Garini Y, Metzler R (2012-08-01). "Strange kinetics of single molecules in living cells". Physics Today. 65 (8): 29–35. Bibcode:2012PhT....65h..29B. doi:10.1063/PT.3.1677. ISSN 0031-9228.
- ↑ "Annual Index". Physics Today. 62 (12): 84. 2009. Bibcode:2009PhT....62l..84.. doi:10.1063/1.3273090. ISSN 0031-9228.
- ↑ Sadegh S, Higgins JL, Mannion PC, Tamkun MM, Krapf D (2017-03-09). "Plasma Membrane is Compartmentalized by a Self-Similar Cortical Actin Meshwork". Physical Review X. 7 (1): 011031. arXiv: 1702.03997 . Bibcode:2017PhRvX...7a1031S. doi:10.1103/PhysRevX.7.011031. PMC 5500227 . PMID 28690919.
- ↑ Gervasi MG, Xu X, Carbajal-Gonzalez B, Buffone MG, Visconti PE, Krapf D (June 2018). "The actin cytoskeleton of the mouse sperm flagellum is organized in a helical structure". Journal of Cell Science. 131 (11): jcs215897. doi:10.1242/jcs.215897. PMC 6031324 . PMID 29739876.
- ↑ Colorado State University. "2022 College Faculty and Staff Award Recipients".
- ↑ Colorado State University. "College Faculty and Staff Award Recipients". Archived from the original on 2021-03-26.
- ↑ "Premio Dr. Bernardo Houssay 2020".
- ↑ "Celebrate! Colorado State award winners for 2018". CSU Source. 11 April 2018. Archived from the original on 2020-08-14. Retrieved March 30, 2021.
- ↑ "2021 College Faculty and Staff Award Recipients". CSU Source. 24 March 2021. Archived from the original on 2020-08-05.
- ↑ "Hebrew University of Jerusalem, Office of the Rector, Rector's prize". Archived from the original on 2021-01-26.