
A poison is any chemical substance that is harmful or lethal to living organisms. The term is used in a wide range of scientific fields and industries, where it is often specifically defined. It may also be applied colloquially or figuratively, with a broad sense.

Insecticides are pesticides used to kill insects. They include ovicides and larvicides used against insect eggs and larvae, respectively. Insecticides are used in agriculture, medicine, industry and by consumers. Insecticides are claimed to be a major factor behind the increase in the 20th-century's agricultural productivity. Nearly all insecticides have the potential to significantly alter ecosystems; many are toxic to humans and/or animals; some become concentrated as they spread along the food chain.

A pest is any organism harmful to humans or human concerns. The term is particularly used for creatures that damage crops, livestock, and forestry or cause a nuisance to people, especially in their homes. Humans have modified the environment for their own purposes and are intolerant of other creatures occupying the same space when their activities impact adversely on human objectives. Thus, an elephant is unobjectionable in its natural habitat but a pest when it tramples crops.

Bait is any appetizing substance used to attract prey when hunting or fishing, most commonly in the form of trapping, ambushing and angling.
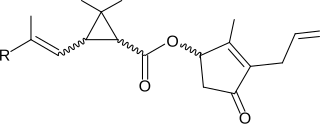
A pyrethroid is an organic compound similar to the natural pyrethrins, which are produced by the flowers of pyrethrums. Pyrethroids are used as commercial and household insecticides.

Molluscicides – also known as snail baits, snail pellets, or slug pellets – are pesticides against molluscs, which are usually used in agriculture or gardening, in order to control gastropod pests specifically slugs and snails which damage crops or other valued plants by feeding on them.

Pest control is the regulation or management of a species defined as a pest; such as any animal, plant or fungus that impacts adversely on human activities or environment. The human response depends on the importance of the damage done and will range from tolerance, through deterrence and management, to attempts to completely eradicate the pest. Pest control measures may be performed as part of an integrated pest management strategy.

Rodenticides are chemicals made and sold for the purpose of killing rodents. While commonly referred to as "rat poison", rodenticides are also used to kill mice, squirrels, woodchucks, chipmunks, porcupines, nutria, beavers, and voles. Despite the crucial roles that rodents play in nature, there are times when they need to be controlled.

Sodium fluoroacetate, also known as compound 1080, is an organofluorine chemical compound with the formula FCH2CO2Na. This colourless salt has a taste similar to that of sodium chloride and is used as a rodenticide.

Metaldehyde is an organic compound with the formula. It is used as a pesticide against slugs and snails. It is the cyclic tetramer of acetaldehyde.

Iron(III) phosphate, also ferric phosphate, is the inorganic compound with the formula FePO4. Several related materials are known, including four polymorphs of FePO4 and two polymorphs of the dihydrate FePO4·(H2O)2. These materials find few technical applications as well as occurring in the mineral kingdom.

The black slug, Arion ater, is a large terrestrial gastropod mollusk in the family Arionidae, the round back slugs. Many land slugs lack external shells, having a vestigial shell. Most slugs retain a remnant of their shell, which is usually internalized, unlike other terrestrial mollusks which have external shells. Without such shells, slugs produce mucus, that may also contain toxins—to deter predators. Terrestrial slugs produce two other forms of mucus that facilitate locomotion and prevent death from drying. Such mollusks are hermaphroditic. Slugs most often function as decomposers but are also often omnivores. Arion ater is one such slug, decomposing organic matter, preying on other organisms, and consuming vegetative matter including agricultural crops. Native to Europe, the black slug is an invasive species in Australia, Canada, and the United States.

Coumatetralyl is an anticoagulant of the 4-hydroxycoumarin vitamin K antagonist type used as a rodenticide.

A leaf miner is any one of numerous species of insects in which the larval stage lives in, and eats, the leaf tissue of plants. The vast majority of leaf-mining insects are moths (Lepidoptera), sawflies, and flies (Diptera). Some beetles also exhibit this behavior.

Spinosad is an insecticide based on chemical compounds found in the bacterial species Saccharopolyspora spinosa. The genus Saccharopolyspora was discovered in 1985 in isolates from crushed sugarcane. The bacteria produce yellowish-pink aerial hyphae, with bead-like chains of spores enclosed in a characteristic hairy sheath. This genus is defined as aerobic, Gram-positive, nonacid-fast actinomycetes with fragmenting substrate mycelium. S. spinosa was isolated from soil collected inside a nonoperational sugar mill rum still in the Virgin Islands. Spinosad is a mixture of chemical compounds in the spinosyn family that has a generalized structure consisting of a unique tetracyclic ring system attached to an amino sugar (D-forosamine) and a neutral sugar (tri-Ο-methyl-L-rhamnose). Spinosad is relatively nonpolar and not easily dissolved in water.
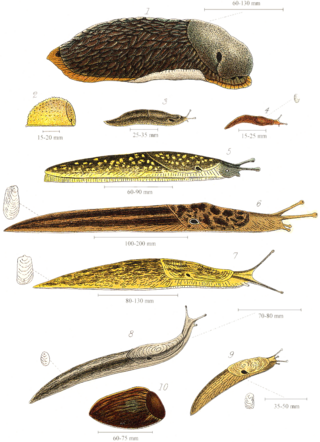
Slug, or land slug, is a common name for any apparently shell-less terrestrial gastropod mollusc. The word slug is also often used as part of the common name of any gastropod mollusc that has no shell, a very reduced shell, or only a small internal shell, particularly sea slugs and semi slugs.

Methiocarb is a carbamate pesticide which is used as an insecticide, bird repellent, acaricide and molluscicide since the 1960s. Methiocarb has contact and stomach action on mites and neurotoxic effects on molluscs. Seeds treated with methiocarb also affect birds. Other names for methiocarb are mesurol and mercaptodimethur.

Ethoprophos (or ethoprop) is an organophosphate ester with the formula C8H19O2PS2. It is a clear yellow to colourless liquid that has a characteristic mercaptan-like odour. It is used as an insecticide and nematicide and it is an acetylcholinesterase inhibitor.

4'-Aminopropiophenone is a chemical compound. It is highly toxic, and can cause injury or death upon physical contact or inhalation of fumes.

Ferric EDTA is the coordination complex formed from ferric ions and EDTA. EDTA has a high affinity for ferric ions. It is a yellow solid that gives yellowish aqueous solutions.





















