Chromatography is a laboratory technique for the separation of a mixture. The mixture is dissolved in a fluid called the mobile phase, which carries it through a system on which is fixed a material called the stationary phase. The different constituents of the mixture have different affinities for the stationary phase. The different molecules stay longer or shorter on the stationary phase, depending on their interactions with its surface sites. So, they travel at different apparent velocities in the mobile fluid, causing them to separate. The separation is based on the differential partitioning between the mobile and the stationary phases. Subtle differences in a compound's partition coefficient result in differential retention on the stationary phase and thus affect the separation.

Distillation, or classical distillation, is the process of separating the components or substances from a liquid mixture by using selective boiling and condensation. Dry distillation is the heating of solid materials to produce gaseous products. Dry distillation may involve chemical changes such as destructive distillation or cracking and is not discussed under this article. Distillation may result in essentially complete separation, or it may be a partial separation that increases the concentration of selected components in the mixture. In either case, the process exploits differences in the relative volatility of the mixture's components. In industrial applications, distillation is a unit operation of practically universal importance, but it is a physical separation process, not a chemical reaction.

Surfactants are compounds that lower the surface tension between two liquids, between a gas and a liquid, or between a liquid and a solid. Surfactants may act as detergents, wetting agents, emulsifiers, foaming agents, or dispersants.

Foam is an object formed by trapping pockets of gas in a liquid or solid.

A fractionating column or fractional column is an essential item used in the distillation of liquid mixtures to separate the mixture into its component parts, or fractions, based on the differences in volatilities. Fractionating columns are used in small scale laboratory distillations as well as large scale industrial distillations.

Froth flotation is a process for selectively separating hydrophobic materials from hydrophilic. This is used in mineral processing, paper recycling and waste-water treatment industries. Historically this was first used in the mining industry, where it was one of the great enabling technologies of the 20th century. It has been described as "the single most important operation used for the recovery and upgrading of sulfide ores". The development of froth flotation has improved the recovery of valuable minerals, such as copper- and lead-bearing minerals. Along with mechanized mining, it has allowed the economic recovery of valuable metals from much lower grade ore than previously.

In the field of extractive metallurgy, mineral processing, also known as ore dressing, is the process of separating commercially valuable minerals from their ores.

Continuous distillation, a form of distillation, is an ongoing separation in which a mixture is continuously fed into the process and separated fractions are removed continuously as output streams. Distillation is the separation or partial separation of a liquid feed mixture into components or fractions by selective boiling and condensation. The process produces at least two output fractions. These fractions include at least one volatile distillate fraction, which has boiled and been separately captured as a vapor condensed to a liquid, and practically always a bottoms fraction, which is the least volatile residue that has not been separately captured as a condensed vapor.
Dissolved air flotation (DAF) is a water treatment process that clarifies wastewaters by the removal of suspended matter such as oil or solids. The removal is achieved by dissolving air in the water or wastewater under pressure and then releasing the air at atmospheric pressure in a flotation tank basin. The released air forms tiny bubbles which adhere to the suspended matter causing the suspended matter to float to the surface of the water where it may then be removed by a skimming device.
Reversed-phase chromatography includes any chromatographic method that uses a hydrophobic stationary phase. RPC refers to liquid chromatography.
Groundwater remediation is the process that is used to treat polluted groundwater by removing the pollutants or converting them into harmless products. Groundwater is water present below the ground surface that saturates the pore space in the subsurface. Globally, between 25 per cent and 40 per cent of the world's drinking water is drawn from boreholes and dug wells. Groundwater is also used by farmers to irrigate crops and by industries to produce everyday goods. Most groundwater is clean, but groundwater can become polluted, or contaminated as a result of human activities or as a result of natural conditions.

A defoamer or an anti-foaming agent is a chemical additive that reduces and hinders the formation of foam in industrial process liquids. The terms anti-foam agent and defoamer are often used interchangeably. Strictly speaking, defoamers eliminate existing foam and anti-foamers prevent the formation of further foam. Commonly used agents are insoluble oils, polydimethylsiloxanes and other silicones, certain alcohols, stearates and glycols. The additive is used to prevent formation of foam or is added to break a foam already formed.
Deinking is the industrial process of removing printing ink from paperfibers of recycled paper to make deinked pulp.
Foam separation is a chemical process which falls into a category of separation techniques called "Adsorptive bubble separation methods". It is further divided into froth flotation and foam fractionation.

Sea foam, ocean foam, beach foam, or spume is a type of foam created by the agitation of seawater, particularly when it contains higher concentrations of dissolved organic matter derived from sources such as the offshore breakdown of algal blooms. These compounds can act as surfactants or foaming agents. As the seawater is churned by breaking waves in the surf zone adjacent to the shore, the surfactants under these turbulent conditions trap air, forming persistent bubbles that stick to each other through surface tension. Sea foam is a global phenomenon and it varies depending on location and the potential influence of the surrounding marine, freshwater, and/or terrestrial environments. Due to its low density and persistence, foam can be blown by strong on-shore winds from the beach face inland. Sea foam can also occur after a ship sinks completely
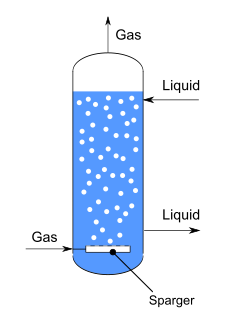
A bubble column reactor is an apparatus used to generate and control gas-liquid chemical reactions. It consists of a vertically-arranged cylindrical column filled with liquid, at the bottom of which gas is inserted.
Adsorption of polyelectrolytes on solid substrates is a surface phenomenon where long-chained polymer molecules with charged groups bind to a surface that is charged in the opposite polarity. On the molecular level, the polymers do not actually bond to the surface, but tend to "stick" to the surface via intermolecular forces and the charges created by the dissociation of various side groups of the polymer. Because the polymer molecules are so long, they have a large amount of surface area with which to contact the surface and thus do not desorb as small molecules are likely to do. This means that adsorbed layers of polyelectrolytes form a very durable coating. Due to this important characteristic of polyelectrolyte layers they are used extensively in industry as flocculants, for solubilization, as supersorbers, antistatic agents, as oil recovery aids, as gelling aids in nutrition, additives in concrete, or for blood compatibility enhancement to name a few.
A separation process is a method that converts a mixture or solution of chemical substances into two or more distinct product mixtures. At least one of results of the separation is enriched in one or more of the source mixture's constituents. In some cases, a separation may fully divide the mixture into pure constituents. Separations exploit differences in chemical properties or physical properties between the constituents of a mixture.
Continuous foam separation is a chemical process closely related to foam fractionation in which foam is used to separate components of a solution when they differ in surface activity. In any solution, surface active components tend to adsorb to gas-liquid interfaces while surface inactive components stay within the bulk solution. When a solution is foamed, the most surface active components collect in the foam and the foam can be easily extracted. This process is commonly used in large-scale projects such as water waste treatment due to a continuous gas flow in the solution.

The Jameson Cell is a high-intensity froth flotation cell that was invented by Laureate Professor Graeme Jameson of the University of Newcastle (Australia) and developed in conjunction with Mount Isa Mines Limited.