A geodesic polyarene in organic chemistry is a polycyclic aromatic hydrocarbon with curved convex or concave surfaces. [1] Examples include fullerenes, nanotubes, corannulenes, helicenes and sumanene. The molecular orbitals of the carbon atoms in these systems are to some extent pyramidalized resulting a different pi electron density on either side of the molecule with consequences for reactivity.
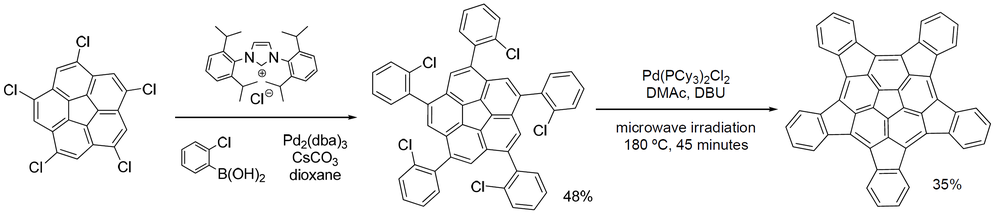
One member of this group of organic compounds, pentaindenocorannulene (depicted below), [2] [3] [4] can be considered a large fullerene fragment. The experimentally obtained curvature and degree of pyramidalizion (12.6° for the carbons of the pentagon at the center [5] ) are both actually larger than that of fullerene but according to its discoverers, the compound is relatively easy to synthesize starting from corannulene and a way is opened to produce larger such fragments by stitching.
The crystal structure of pentaindenocorannulene has been obtained. [4] An illustration of the crystal packing for pentaindenocorannulene is given below.
Another geodesic polyarene that has been synthesized is C50H10. [6] [4] C50H10 can be described as a short, rigid, structurally pure [5,5] carbon nanotube. The crystal structure of C50H10 has been obtained. The carbons of the pentagon at the center of C50H10 have a POAV angle of 12.3°, less than that of pentaindenocorannulene. The synthesis is as shown below. FVP stands for flash vacuum pyrolysis.
Some bowl-shaped molecules reported in the literature are in fact partially hydrogenated. A C56H40 hydrocarbon has been synthesized containing 54 of the 60 carbon atoms found in fullerene [7]