| |
 | |
| Names | |
|---|---|
| Preferred IUPAC name 4,7-Dihydro-1H-tricyclopenta[def,jkl,pqr]triphenylene | |
| Identifiers | |
3D model (JSmol) | |
| ChemSpider | |
PubChem CID | |
| UNII | |
CompTox Dashboard (EPA) | |
| |
| |
| Properties | |
| C21H12 | |
| Molar mass | 264.32 |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |
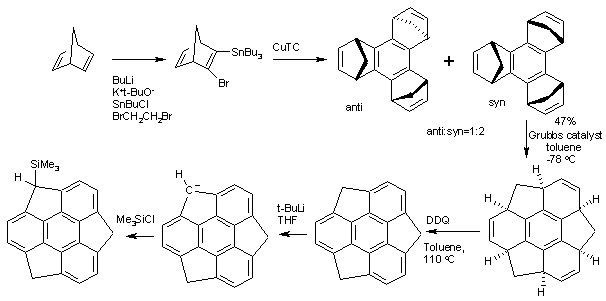
Sumanene is a polycyclic aromatic hydrocarbon and of scientific interest because the molecule can be considered a fragment of buckminsterfullerene. Due to this connection and also its bowl shape, Sumanene is also known as a buckybowl.Suman means "flower" in both Hindi and Sanskrit. [1] The core of the arene is a benzene ring and the periphery consists of alternating benzene rings (3) and cyclopentadiene rings (3). Unlike fullerene, sumanene has benzyl positions which are available for organic reactions.