Related Research Articles
omega-Grammotoxin SIA (ω-grammotoxin SIA) is a protein toxin that inhibits P, Q, and N voltage-gated calcium channels (Ca2+ channels) in neurons.

Agitoxin is a toxin found in the venom of the scorpion Leiurus quinquestriatus hebraeus. Other toxins found in this species include charybdotoxin (CTX). CTX is a close homologue of Agitoxin.

Grammotoxin is a toxin in the venom of the tarantula Grammostola spatulata. It is a protein toxin that inhibits P-, Q- and N-type voltage-gated calcium channels in neurons. Grammotoxin is also known as omega-grammotoxin SIA.

Heteropodatoxins are peptide toxins from the venom of the giant crab spider Heteropoda venatoria, which block Kv4.2 voltage-gated potassium channels.
Stromatoxin is a spider toxin that blocks certain delayed-rectifier and A-type voltage-gated potassium channels.

Potassium voltage-gated channel, Shab-related subfamily, member 1, also known as KCNB1 or Kv2.1, is a protein that, in humans, is encoded by the KCNB1 gene.

Spider toxins are a family of proteins produced by spiders which function as neurotoxins. The mechanism of many spider toxins is through blockage of calcium channels.
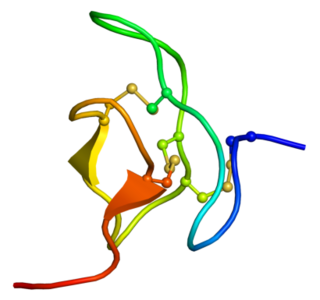
Phrixotoxins are peptide toxins derived from the venom of the Chilean copper tarantula Phrixotrichus auratus, also named Paraphysa scrofa. Phrixotoxin-1 and -2 block A-type voltage-gated potassium channels; phrixotoxin-3 blocks voltage-gated sodium channels. Similar toxins are found in other species, for instance the Chilean rose tarantula.
BeKm-1 is a toxin from the Central Asian scorpion Buthus eupeus. BeKm-1 acts by selectively inhibiting the human Ether-à-go-go Related Gene (hERG) channels, which are voltage gated potassium ion channels.
Jingzhaotoxin proteins are part of a venom secreted by Chilobrachys jingzhao, the Chinese tarantula. and act as neurotoxins. There are several subtypes of jingzhaotoxin, which differ in terms of channel selectivity and modification characteristics. All subspecies act as gating modifiers of sodium channels and/or, to a lesser extent, potassium channels.

Heteroscodratoxin-1 is a neurotoxin produced by the venom glands of Heteroscodra maculata that shifts the activation threshold of voltage-gated potassium channels and the inactivation of Nav1.1 sodium channels to more positive potentials.

Vanillotoxins are neurotoxins found in the venom of the tarantula Psalmopoeus cambridgei. They act as agonists for the transient receptor potential cation channel subfamily V member 1 (TRPV1), activating the pain sensory system. VaTx1 and 2 also act as antagonists for the Kv2-type voltage-gated potassium channel (Kv2), inducing paralytic behavior in small animals.

Guangxitoxin, also known as GxTX, is a peptide toxin found in the venom of the tarantula Plesiophrictus guangxiensis. It primarily inhibits outward voltage-gated Kv2.1 potassium channel currents, which are prominently expressed in pancreatic β-cells, thus increasing insulin secretion.
Theraphosa leblondi toxin (TLTx) is a toxin occurring in three different forms (subtypes) that are purified and sequenced from the venom of the giant tarantula Theraphosa blondi. This toxin selectively inhibits Kv4.2 voltage-gated potassium channels by acting as a gating modifier.
Centruroides suffusus suffusus toxin II (CssII) is a scorpion β-toxin from the venom of the scorpion Centruroides suffusus suffusus. CssII primarily affects voltage-gated sodium channels by causing a hyperpolarizing shift of voltage dependence, a reduction in peak transient current, and the occurrence of resurgent currents.
HgeTx1 (systematic name: α-KTx 6.14) is a toxin produced by the Mexican scorpion Hoffmanihadrurus gertschi that is a reversible blocker of the Shaker B K+-channel, a type of voltage-gated potassium channels.
LmαTX5 is an α-scorpion toxin which inhibits the fast inactivation of voltage-gated sodium channels. It has been identified through transcriptome analysis of the venom gland of Lychas mucronatus, also known as the Chinese swimming scorpion – a scorpion species which is widely distributed in Southeast Asia.
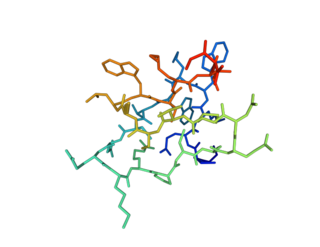
GiTx1 (β/κ-theraphotoxin-Gi1a) is a peptide toxin present in the venom of Grammostola iheringi. It reduces both inward and outward currents by blocking voltage-gated sodium and potassium channels, respectively.
Protoxin-I, also known as ProTx-I, or Beta/omega-theraphotoxin-Tp1a, is a 35-amino-acid peptide neurotoxin extracted from the venom of the tarantula Thrixopelma pruriens. Protoxin-I belongs to the inhibitory cystine knot (ICK) family of peptide toxins, which have been known to potently inhibit voltage-gated ion channels. Protoxin-I selectively blocks low voltage threshold T-type calcium channels, voltage gated sodium channels and the nociceptor cation channel TRPA1. Due to its unique ability to bind to TRPA1, Protoxin-I has been implicated as a valuable pharmacological reagent with potential applications in clinical contexts with regards to pain and inflammation
Grammostola iheringi also known as the Entre Rios tarantula, it was first described by Keyserling in 1891. They are found in Brazil, and is considered the biggest tarantula in the Grammostola genus.
References
- ↑ http://supfam.org/SUPERFAMILY/cgi-bin/search.cgi?search_field=hanatoxin, consulted 10th Oct. 2012.
- 1 2 Swartz, K.J.; MacKinnon, R. (1995). "An Inhibitor of the Kv2.1 Potassium Channel Isolated from the Venom of a Chilean Tarantula". Neuron. 15 (4): 941–949. doi: 10.1016/0896-6273(95)90184-1 . PMID 7576642. S2CID 11188679.
- 1 2 Swartz, K.J.; MacKinnon, R. (1997). "Hanatoxin modifies the gating of a voltage-dependent K+ channel through multiple binding sites". Neuron. 18 (4): 665–673. doi: 10.1016/s0896-6273(00)80306-2 . PMID 9136774. S2CID 17929074.
- ↑ Takahashi, H. e.a. (2000). "Solution structure of hanatoxin1, a gating modifier of voltage-dependent K+ channels: common surface features of gating modifier toxins". Journal of Molecular Biology. 297 (3): 771–780. doi:10.1006/jmbi.2000.3609. PMID 10731427.
- 1 2 Lee, C. e.a. (2004). "Solution Structure and Functional Characterization of SGTx1, a Modifier of Kv2.1 Channel Gating". Biochemistry. 43 (4): 890–897. doi:10.1021/bi0353373. PMID 14744131.
- ↑ Li-Smerin, Y.; Swartz, K.J. (1998). "Gating modifier toxins reveal a conserved structural motif in voltage-gated Ca2+ and K+ channels". Proceedings of the National Academy of Sciences. 95 (15): 8585–8589. Bibcode:1998PNAS...95.8585L. doi: 10.1073/pnas.95.15.8585 . PMC 21119 . PMID 9671721.
- ↑ Gonzalez, C. e.a. (2000). "Modulation of the Shaker K(+) channel gating kinetics by the S3-S4 linker". Journal of General Physiology. 115 (2): 193–208. doi:10.1085/jgp.115.2.193. PMC 2217197 . PMID 10653896.
- ↑ Li-Smerin, Y.; Swartz, K.J. (2001). "Helical structure of the COOH terminus of S3 and its contribution to the gating modifier toxin receptor in voltage-gated ion channels". Journal of General Physiology. 117 (3): 205–218. doi:10.1085/jgp.117.3.205. PMC 2225613 . PMID 11222625.
- ↑ Huang, P.; Shiau, Y.; Lou, K. (2007). "The interaction of spider gating modifier peptides with voltage-gated potassium channels". Toxicon. 49 (2): 285–292. Bibcode:2007Txcn...49..285H. doi:10.1016/j.toxicon.2006.09.015. PMID 17113615.
- ↑ http://www.t3db.org/toxins, consulted 9th Oct. 2012.
- ↑ Escoubas, P.; Rash, L. (2004). "Review Tarantulas: eight-legged pharmacists and combinatorial chemists". Toxicon. 43 (5): 555–574. doi:10.1016/j.toxicon.2004.02.007. PMID 15066413.
- ↑ Tintinalli, J. e.a. (2004). Tintinalli’s Emergency Medicine: A comprehensive Study Guide, 7e. New York, NY McGraw-Hill, Chapter 50, 205.
- ↑ Escoubas, P.; Bosmans, F. (2007). "Spider peptide toxins as leads for drug development". Natural Product Reports. 2 (6): 1–13. doi:10.1517/17460441.2.6.823. PMID 23489000. S2CID 22614679.
- ↑ Herrington, J. e.a. (2006). "Blockers of the Delayed-Rectifier Potassium Current in Pancreatic β-Cells Enhance Glucose-Dependent Insulin Secretion". Diabetes. 55 (4): 1034–1042. doi: 10.2337/diabetes.55.04.06.db05-0788 . PMID 16567526.