
The thymus is a specialized primary lymphoid organ of the immune system. Within the thymus, thymus cell lymphocytes or T cells mature. T cells are critical to the adaptive immune system, where the body adapts to specific foreign invaders. The thymus is located in the upper front part of the chest, in the anterior superior mediastinum, behind the sternum, and in front of the heart. It is made up of two lobes, each consisting of a central medulla and an outer cortex, surrounded by a capsule.
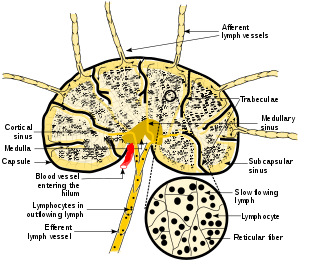
A lymph node, or lymph gland, is a kidney-shaped organ of the lymphatic system and the adaptive immune system. A large number of lymph nodes are linked throughout the body by the lymphatic vessels. They are major sites of lymphocytes that include B and T cells. Lymph nodes are important for the proper functioning of the immune system, acting as filters for foreign particles including cancer cells, but have no detoxification function.

T cells are one of the important types of white blood cells of the immune system and play a central role in the adaptive immune response. T cells can be distinguished from other lymphocytes by the presence of a T-cell receptor (TCR) on their cell surface.
In immunology, central tolerance is the process of eliminating any developing T or B lymphocytes that are autoreactive, i.e. reactive to the body itself. Through elimination of autoreactive lymphocytes, tolerance ensures that the immune system does not attack self peptides. Lymphocyte maturation occurs in primary lymphoid organs such as the bone marrow and the thymus. In mammals, B cells mature in the bone marrow and T cells mature in the thymus.
Gut-associated lymphoid tissue (GALT) is a component of the mucosa-associated lymphoid tissue (MALT) which works in the immune system to protect the body from invasion in the gut.
A thymocyte is an immune cell present in the thymus, before it undergoes transformation into a T cell. Thymocytes are produced as stem cells in the bone marrow and reach the thymus via the blood.
Lymphopoiesis (lĭm'fō-poi-ē'sĭs) is the generation of lymphocytes, one of the five types of white blood cells (WBCs). It is more formally known as lymphoid hematopoiesis.

The autoimmune regulator (AIRE) is a protein that in humans is encoded by the AIRE gene. It is a 13kb gene on chromosome 21q22.3 that has 545 amino acids. AIRE is a transcription factor expressed in the medulla of the thymus. It is part of the mechanism which eliminates self-reactive T cells that would cause autoimmune disease. It exposes T cells to normal, healthy proteins from all parts of the body, and T cells that react to those proteins are destroyed.

Chemokine ligand 21 (CCL21) is a small cytokine belonging to the CC chemokine family. This chemokine is also known as 6Ckine, exodus-2, and secondary lymphoid-tissue chemokine (SLC). CCL21 elicits its effects by binding to a cell surface chemokine receptor known as CCR7. The main function of CCL21 is to guide CCR7 expressing leukocytes to the secondary lymphoid organs, such as lymph nodes and Peyer´s patches.

C-C chemokine receptor type 7 is a protein that in humans is encoded by the CCR7 gene. Two ligands have been identified for this receptor: the chemokines ligand 19 (CCL19/ELC) and ligand 21 (CCL21). The ligands have similar affinity for the receptor, though CCL19 has been shown to induce internalisation of CCR7 and desensitisation of the cell to CCL19/CCL21 signals. CCR7 is a transmembrane protein with 7 transmembrane domains, which is coupled with heterotrimeric G proteins, which transduce the signal downstream through various signalling cascades. The main function of the receptor is to guide immune cells to immune organs by detecting specific chemokines, which these tissues secrete.

Thymic stromal lymphopoietin (TSLP) is a protein belonging to the cytokine family. It is known to play an important role in the maturation of T cell populations through activation of antigen-presenting cells.
In immunology, clonal deletion is the removal through apoptosis of B cells and T cells that have expressed receptors for self before developing into fully immunocompetent lymphocytes. This prevents recognition and destruction of self host cells, making it a type of negative selection or central tolerance. Central tolerance prevents B and T lymphocytes from reacting to self. Thus, clonal deletion can help protect individuals against autoimmunity. Clonal deletion is thought to be the most common type of negative selection. It is one method of immune tolerance.
Thymic nurse cells (TNCs) are large epithelial cells found in the cortex of the thymus and also in cortico-medullary junction. They have their own nucleus and are known to internalize thymocytes through extensions of plasma membrane. The cell surfaces of TNCs and their cytoplasmic vacuoles express MHC Class I and MHC Class II antigens. The interaction of these antigens with the developing thymocytes determines whether the thymocytes undergo positive or negative selection.
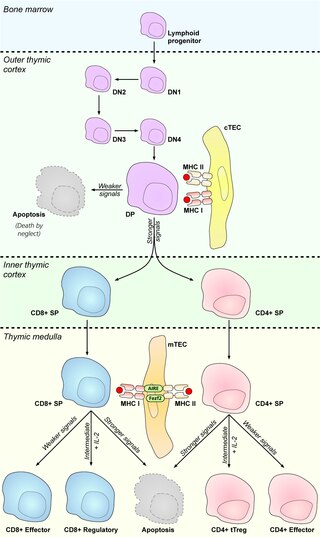
Medullary thymic epithelial cells (mTECs) represent a unique stromal cell population of the thymus which plays an essential role in the establishment of central tolerance. Therefore, mTECs rank among cells relevant for the development of functional mammal immune system.
Antigen transfer in the thymus is the transmission of self-antigens between thymic antigen-presenting cells which contributes to the establishment of T cell central tolerance.

Cortical thymic epithelial cells (cTECs) form unique parenchyma cell population of the thymus which critically contribute to the development of T cells.
Thymic epithelial cells (TECs) are specialized cells with high degree of anatomic, phenotypic and functional heterogeneity that are located in the outer layer (epithelium) of the thymic stroma. The thymus, as a primary lymphoid organ, mediates T cell development and maturation. The thymic microenvironment is established by TEC network filled with thymocytes in different developing stages. TECs and thymocytes are the most important components in the thymus, that are necessary for production of functionally competent T lymphocytes and self tolerance. Dysfunction of TECs causes several immunodeficiencies and autoimmune diseases.
Promiscuous gene expression (PGE), formerly referred to as ectopic expression, is a process specific to the thymus that plays a pivotal role in the establishment of central tolerance. This phenomenon enables generation of self-antigens, so called tissue-restricted antigens (TRAs), which are in the body expressed only by one or few specific tissues. These antigens are represented for example by insulin from the pancreas or defensins from the gastrointestinal tract. Antigen-presenting cells (APCs) of the thymus, namely medullary thymic epithelial cells (mTECs), dendritic cells (DCs) and B cells are capable to present peptides derived from TRAs to developing T cells and hereby test, whether their T cell receptors (TCRs) engage self entities and therefore their occurrence in the body can potentially lead to the development of autoimmune disease. In that case, thymic APCs either induce apoptosis in these autoreactive T cells or they deviate them to become T regulatory cells, which suppress self-reactive T cells in the body that escaped negative selection in the thymus. Thus, PGE is crucial for tissues protection against autoimmunity.
Thymus stromal cells are subsets of specialized cells located in different areas of the thymus. They include all non-T-lineage cells, such as thymic epithelial cells (TECs), endothelial cells, mesenchymal cells, dendritic cells, and B lymphocytes, and provide signals essential for thymocyte development and the homeostasis of the thymic stroma.
Thymic mimetic cells are a heterogeneous population of cells located in the thymus that exhibit phenotypes of a wide variety of differentiated peripheral cells. They arise from medullary thymic epithelial cells (mTECs) and also function in negative selection of self-reactive T cells.








