
Phospholipids are a class of lipids whose molecule has a hydrophilic "head" containing a phosphate group and two hydrophobic "tails" derived from fatty acids, joined by an alcohol residue. Marine phospholipids typically have omega-3 fatty acids EPA and DHA integrated as part of the phospholipid molecule. The phosphate group can be modified with simple organic molecules such as choline, ethanolamine or serine.

The lipid bilayer is a thin polar membrane made of two layers of lipid molecules. These membranes are flat sheets that form a continuous barrier around all cells. The cell membranes of almost all organisms and many viruses are made of a lipid bilayer, as are the nuclear membrane surrounding the cell nucleus, and membranes of the membrane-bound organelles in the cell. The lipid bilayer is the barrier that keeps ions, proteins and other molecules where they are needed and prevents them from diffusing into areas where they should not be. Lipid bilayers are ideally suited to this role, even though they are only a few nanometers in width, because they are impermeable to most water-soluble (hydrophilic) molecules. Bilayers are particularly impermeable to ions, which allows cells to regulate salt concentrations and pH by transporting ions across their membranes using proteins called ion pumps.

A liposome is a small artificial vesicle, spherical in shape, having at least one lipid bilayer. Due to their hydrophobicity and/or hydrophilicity, biocompatibility, particle size and many other properties, liposomes can be used as drug delivery vehicles for administration of pharmaceutical drugs and nutrients, such as lipid nanoparticles in mRNA vaccines, and DNA vaccines. Liposomes can be prepared by disrupting biological membranes.
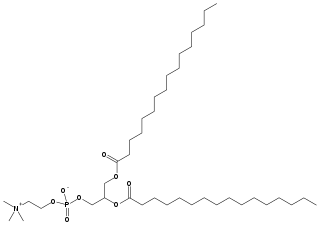
Dipalmitoylphosphatidylcholine (DPPC) is a phospholipid (and a lecithin) consisting of two C16 palmitic acid groups attached to a phosphatidylcholine head-group.
A lamella in biology refers to a thin layer, membrane or plate of tissue. This is a very broad definition, and can refer to many different structures. Any thin layer of organic tissue can be called a lamella and there is a wide array of functions an individual layer can serve. For example, an intercellular lipid lamella is formed when lamellar disks fuse to form a lamellar sheet. It is believed that these disks are formed from vesicles, giving the lamellar sheet a lipid bilayer that plays a role in water diffusion.

Cationic liposomes are spherical structures that contain positively charged lipids. Cationic liposomes can vary in size between 40 nm and 500 nm, and they can either have one lipid bilayer (monolamellar) or multiple lipid bilayers (multilamellar). The positive charge of the phospholipids allows cationic liposomes to form complexes with negatively charged nucleic acids through ionic interactions. Upon interacting with nucleic acids, cationic liposomes form clusters of aggregated vesicles. These interactions allow cationic liposomes to condense and encapsulate various therapeutic and diagnostic agents in their aqueous compartment or in their lipid bilayer. These cationic liposome-nucleic acid complexes are also referred to as lipoplexes. Due to the overall positive charge of cationic liposomes, they interact with negatively charged cell membranes more readily than classic liposomes. This positive charge can also create some issues in vivo, such as binding to plasma proteins in the bloodstream, which leads to opsonization. These issues can be reduced by optimizing the physical and chemical properties of cationic liposomes through their lipid composition. Cationic liposomes are increasingly being researched for use as delivery vectors in gene therapy due to their capability to efficiently transfect cells. A common application for cationic liposomes is cancer drug delivery.

Drug delivery refers to approaches, formulations, manufacturing techniques, storage systems, and technologies involved in transporting a pharmaceutical compound to its target site to achieve a desired therapeutic effect. Principles related to drug preparation, route of administration, site-specific targeting, metabolism, and toxicity are used to optimize efficacy and safety, and to improve patient convenience and compliance. Drug delivery is aimed at altering a drug's pharmacokinetics and specificity by formulating it with different excipients, drug carriers, and medical devices. There is additional emphasis on increasing the bioavailability and duration of action of a drug to improve therapeutic outcomes. Some research has also been focused on improving safety for the person administering the medication. For example, several types of microneedle patches have been developed for administering vaccines and other medications to reduce the risk of needlestick injury.
One property of a lipid bilayer is the relative mobility (fluidity) of the individual lipid molecules and how this mobility changes with temperature. This response is known as the phase behavior of the bilayer. Broadly, at a given temperature a lipid bilayer can exist in either a liquid or a solid phase. The solid phase is commonly referred to as a “gel” phase. All lipids have a characteristic temperature at which they undergo a transition (melt) from the gel to liquid phase. In both phases the lipid molecules are constrained to the two dimensional plane of the membrane, but in liquid phase bilayers the molecules diffuse freely within this plane. Thus, in a liquid bilayer a given lipid will rapidly exchange locations with its neighbor millions of times a second and will, through the process of a random walk, migrate over long distances.

In membrane biology, fusion is the process by which two initially distinct lipid bilayers merge their hydrophobic cores, resulting in one interconnected structure. If this fusion proceeds completely through both leaflets of both bilayers, an aqueous bridge is formed and the internal contents of the two structures can mix. Alternatively, if only one leaflet from each bilayer is involved in the fusion process, the bilayers are said to be hemifused. In hemifusion, the lipid constituents of the outer leaflet of the two bilayers can mix, but the inner leaflets remain distinct. The aqueous contents enclosed by each bilayer also remain separated.

Lipid nanoparticles (LNPs) are nanoparticles composed of lipids. They are a novel pharmaceutical drug delivery system, and a novel pharmaceutical formulation. LNPs as a drug delivery vehicle were first approved in 2018 for the siRNA drug Onpattro. LNPs became more widely known in late 2020, as some COVID-19 vaccines that use RNA vaccine technology coat the fragile mRNA strands with PEGylated lipid nanoparticles as their delivery vehicle.

Biophysical chemistry is a physical science that uses the concepts of physics and physical chemistry for the study of biological systems. The most common feature of the research in this subject is to seek an explanation of the various phenomena in biological systems in terms of either the molecules that make up the system or the supra-molecular structure of these systems. Apart from the biological applications, recent research showed progress in the medical field as well.
A vesosome is a multi-compartmental structure of lipidic nature used to deliver drugs. They can be considered multivesicular vesicles (MVV) and are, therefore, liposome-derived structures.
Nanoparticles for drug delivery to the brain is a method for transporting drug molecules across the blood–brain barrier (BBB) using nanoparticles. These drugs cross the BBB and deliver pharmaceuticals to the brain for therapeutic treatment of neurological disorders. These disorders include Parkinson's disease, Alzheimer's disease, schizophrenia, depression, and brain tumors. Part of the difficulty in finding cures for these central nervous system (CNS) disorders is that there is yet no truly efficient delivery method for drugs to cross the BBB. Antibiotics, antineoplastic agents, and a variety of CNS-active drugs, especially neuropeptides, are a few examples of molecules that cannot pass the BBB alone. With the aid of nanoparticle delivery systems, however, studies have shown that some drugs can now cross the BBB, and even exhibit lower toxicity and decrease adverse effects throughout the body. Toxicity is an important concept for pharmacology because high toxicity levels in the body could be detrimental to the patient by affecting other organs and disrupting their function. Further, the BBB is not the only physiological barrier for drug delivery to the brain. Other biological factors influence how drugs are transported throughout the body and how they target specific locations for action. Some of these pathophysiological factors include blood flow alterations, edema and increased intracranial pressure, metabolic perturbations, and altered gene expression and protein synthesis. Though there exist many obstacles that make developing a robust delivery system difficult, nanoparticles provide a promising mechanism for drug transport to the CNS.
Leaf Huang is a Fred Eshelman Distinguished Professor in the UNC Eshelman School of Pharmacy and a Professor UNC/NC State joint Biomedical Engineering department. He has authored and co-authored over 570 peer reviewed articles and as of 2017 carries an h-index of 138.
A unilamellar liposome is a spherical liposome, a vesicle, bounded by a single bilayer of an amphiphilic lipid or a mixture of such lipids, containing aqueous solution inside the chamber. Unilamellar liposomes are used to study biological systems and to mimic cell membranes, and are classified into three groups based on their size: small unilamellar liposomes/vesicles (SUVs) that with a size range of 20–100 nm, large unilamellar liposomes/vesicles (LUVs) with a size range of 100–1000 nm and giant unilamellar liposomes/vesicles (GUVs) with a size range of 1–200 μm. GUVs are mostly used as models for biological membranes in research work. Animal cells are 10–30 μm and plant cells are typically 10–100 μm. Even smaller cell organelles such as mitochondria are typically 1–2 μm. Therefore, a proper model should account for the size of the specimen being studied. In addition, the size of vesicles dictates their membrane curvature which is an important factor in studying fusion proteins. SUVs have a higher membrane curvature and vesicles with high membrane curvature can promote membrane fusion faster than vesicles with lower membrane curvature such as GUVs.

Lamellar phase refers generally to packing of polar-headed long chain nonpolar-tail molecules in an environment of bulk polar liquid, as sheets of bilayers separated by bulk liquid. In biophysics, polar lipids pack as a liquid crystalline bilayer, with hydrophobic fatty acyl long chains directed inwardly and polar headgroups of lipids aligned on the outside in contact with water, as a 2-dimensional flat sheet surface. Under transmission electron microscope (TEM), after staining with polar headgroup reactive chemical osmium tetroxide, lamellar lipid phase appears as two thin parallel dark staining lines/sheets, constituted by aligned polar headgroups of lipids. 'Sandwiched' between these two parallel lines, there exists one thicker line/sheet of non-staining closely packed layer of long lipid fatty acyl chains. This TEM-appearance became famous as Robertson's unit membrane - the basis of all biological membranes, and structure of lipid bilayer in unilamellar liposomes. In multilamellar liposomes, many such lipid bilayer sheets are layered concentrically with water layers in between.

Gerald Weissmann was an Austrian-born American physician/scientist, editor, and essayist. He was Professor Emeritus and Research Professor of Medicine (Rheumatology) at New York University School of Medicine. He was editor-in-chief (2006–16) of The FASEB Journal. At the time of his death he was its book review editor. In 1965, he was one of the discoverers of liposomes and is credited with coining that term.
Owais Mohammad is an Indian immunologist, nano-technologist and a professor at the interdisciplinary biotechnology unit of the Aligarh Muslim University. Known for his studies on nanotechnology-based vaccine and drug delivery, Owais is the author of two books, Trypanothione reductase: a potential anti-leishmanial drug target and Antimicrobial properties of clove oil: clove oils as antimicrobial agent. He has also co-edited two books, Modern Phytomedicine: Turning Medicinal Plants into Drugs and Combating Fungal Infections: Problems and Remedy, and has contributed chapters. His studies have also been documented by way of a number of articles and ResearchGate, an online repository of scientific articles has listed 60 of them. He is a recipient of the Rashtriya Gaurav Award of the India International Friendship Society. The Department of Biotechnology of the Government of India awarded him the National Bioscience Award for Career Development, one of the highest Indian science awards, for his contributions to biosciences in 2007. His work has been displayed on cover pages of FEMS Immunol. Med Microbiology for all the issues of Year 2006 and Molecular Medicine in May–June issue of Year 2007.

A liposome extruder is a device that prepares cell membranes, exosomes and also generates nanoscale liposome formulations. The liposome extruder employs the track-etched membrane to filter huge particles and achieve sterile filtration.
Immunoliposome therapy is a targeted drug delivery method that involves the use of liposomes coupled with monoclonal antibodies to deliver therapeutic agents to specific sites or tissues in the body. The antibody modified liposomes target tissue through cell-specific antibodies with the release of drugs contained within the assimilated liposomes. Immunoliposome aims to improve drug stability, personalize treatments, and increased drug efficacy. This form of therapy has been used to target specific cells, protecting the encapsulated drugs from degradation in order to enhance their stability, to facilitate sustained drug release and hence to advance current traditional cancer treatment.











