Related Research Articles

Within a nervous system, a neuron, neurone, or nerve cell is an electrically excitable cell that fires electric signals called action potentials across a neural network. Neurons communicate with other cells via synapses, which are specialized connections that commonly use minute amounts of chemical neurotransmitters to pass the electric signal from the presynaptic neuron to the target cell through the synaptic gap.
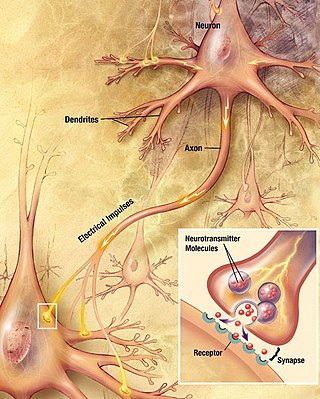
Chemical synapses are biological junctions through which neurons' signals can be sent to each other and to non-neuronal cells such as those in muscles or glands. Chemical synapses allow neurons to form circuits within the central nervous system. They are crucial to the biological computations that underlie perception and thought. They allow the nervous system to connect to and control other systems of the body.
The development of the nervous system, or neural development (neurodevelopment), refers to the processes that generate, shape, and reshape the nervous system of animals, from the earliest stages of embryonic development to adulthood. The field of neural development draws on both neuroscience and developmental biology to describe and provide insight into the cellular and molecular mechanisms by which complex nervous systems develop, from nematodes and fruit flies to mammals.
An inhibitory postsynaptic potential (IPSP) is a kind of synaptic potential that makes a postsynaptic neuron less likely to generate an action potential. The opposite of an inhibitory postsynaptic potential is an excitatory postsynaptic potential (EPSP), which is a synaptic potential that makes a postsynaptic neuron more likely to generate an action potential. IPSPs can take place at all chemical synapses, which use the secretion of neurotransmitters to create cell-to-cell signalling. EPSPs and IPSPs compete with each other at numerous synapses of a neuron. This determines whether an action potential occurring at the presynaptic terminal produces an action potential at the postsynaptic membrane. Some common neurotransmitters involved in IPSPs are GABA and glycine.

Interneurons are neurons that connect to brain regions, i.e. not direct motor neurons or sensory neurons. Interneurons are the central nodes of neural circuits, enabling communication between sensory or motor neurons and the central nervous system (CNS). They play vital roles in reflexes, neuronal oscillations, and neurogenesis in the adult mammalian brain.
In the anatomy of the eye, amacrine cells are interneurons in the retina. They are named from Greek a– 'non', makr– 'long', and in– 'fiber', because of their short neuronal processes. Amacrine cells are inhibitory neurons, and they project their dendritic arbors onto the inner plexiform layer (IPL), they interact with retinal ganglion cells, and bipolar cells or both of these.

Basket cells are inhibitory GABAergic interneurons of the brain, found throughout different regions of the cortex and cerebellum.

Stellate cells are neurons in the central nervous system, named for their star-like shape formed by dendritic processes radiating from the cell body. Many stellate cells are GABAergic and are located in the molecular layer of the cerebellum. Stellate cells are derived from dividing progenitor cells in the white matter of postnatal cerebellum. Dendritic trees can vary between neurons. There are two types of dendritic trees in the cerebral cortex, which include pyramidal cells, which are pyramid shaped and stellate cells which are star shaped. Dendrites can also aid neuron classification. Dendrites with spines are classified as spiny, those without spines are classified as aspinous. Stellate cells can be spiny or aspinous, while pyramidal cells are always spiny. Most common stellate cells are the inhibitory interneurons found within the upper half of the molecular layer in the cerebellum. Cerebellar stellate cells synapse onto the dendritic trees of Purkinje cells and send inhibitory signals. Stellate neurons are sometimes found in other locations in the central nervous system; cortical spiny stellate cells are found in layer IVC of the primary visual cortex. In the somatosensory barrel cortex of mice and rats, glutamatergic (excitatory) spiny stellate cells are organized in the barrels of layer 4. They receive excitatory synaptic fibres from the thalamus and process feed forward excitation to 2/3 layer of the primary visual cortex to pyramidal cells. Cortical spiny stellate cells have a 'regular' firing pattern. Stellate cells are chromophobes, that is cells that does not stain readily, and thus appears relatively pale under the microscope.

In neuroscience, Golgi cells are the most abundant inhibitory interneurons found within the granular layer of the cerebellum. Golgi cells can be found in the granular layer at various layers. The Golgi cell is essential for controlling the activity of the granular layer. They were first identified as inhibitory in 1964. It was also the first example of an inhibitory feedback network in which the inhibitory interneuron was identified anatomically. Golgi cells produce a wide lateral inhibition that reaches beyond the afferent synaptic field and inhibit granule cells via feedforward and feedback inhibitory loops. These cells synapse onto the dendrite of granule cells and unipolar brush cells. They receive excitatory input from mossy fibres, also synapsing on granule cells, and parallel fibers, which are long granule cell axons. Thereby this circuitry allows for feed-forward and feed-back inhibition of granule cells.

Neurotransmission is the process by which signaling molecules called neurotransmitters are released by the axon terminal of a neuron, and bind to and react with the receptors on the dendrites of another neuron a short distance away. A similar process occurs in retrograde neurotransmission, where the dendrites of the postsynaptic neuron release retrograde neurotransmitters that signal through receptors that are located on the axon terminal of the presynaptic neuron, mainly at GABAergic and glutamatergic synapses.
Depolarization-induced suppression of inhibition is the classical and original electrophysiological example of endocannabinoid function in the central nervous system. Prior to the demonstration that depolarization-induced suppression of inhibition was dependent on the cannabinoid CB1 receptor function, there was no way of producing an in vitro endocannabinoid mediated effect.

In the hippocampus, the mossy fiber pathway consists of unmyelinated axons projecting from granule cells in the dentate gyrus that terminate on modulatory hilar mossy cells and in Cornu Ammonis area 3 (CA3), a region involved in encoding short-term memory. These axons were first described as mossy fibers by Santiago Ramón y Cajal as they displayed varicosities along their lengths that gave them a mossy appearance. The axons that make up the pathway emerge from the basal portions of the granule cells and pass through the hilus of the dentate gyrus before entering the stratum lucidum of CA3. Granule cell synapses tend to be glutamatergic, though immunohistological data has indicated that some synapses contain neuropeptidergic elements including opiate peptides such as dynorphin and enkephalin. There is also evidence for co-localization of both GABAergic and glutamatergic neurotransmitters within mossy fiber terminals. GABAergic and glutamatergic co-localization in mossy fiber boutons has been observed primarily in the developing hippocampus, but in adulthood, evidence suggests that mossy fiber synapses may alternate which neurotransmitter is released through activity-dependent regulation.

Chandelier neurons or chandelier cells are a subset of GABAergic cortical interneurons. They are described as parvalbumin-containing and fast-spiking to distinguish them from other subtypes of GABAergic neurons, although more recent work has suggested that only a subset of chandelier cells test positive for parvalbumin by immunostaining. The name comes from the specific shape of their axon arbors, with the terminals forming distinct arrays called "cartridges". The cartridges are immunoreactive to an isoform of the GABA membrane transporter, GAT-1, and this serves as their identifying feature. GAT-1 is involved in the process of GABA reuptake into nerve terminals, thus helping to terminate its synaptic activity. Chandelier neurons synapse exclusively to the axon initial segment of pyramidal neurons, near the site where action potential is generated. It is believed that they provide inhibitory input to the pyramidal neurons, but there is data showing that in some circumstances the GABA from chandelier neurons could be excitatory.
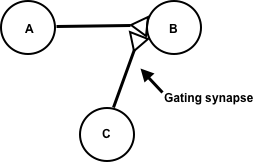
Synaptic gating is the ability of neural circuits to gate inputs by either suppressing or facilitating specific synaptic activity. Selective inhibition of certain synapses has been studied thoroughly, and recent studies have supported the existence of permissively gated synaptic transmission. In general, synaptic gating involves a mechanism of central control over neuronal output. It includes a sort of gatekeeper neuron, which has the ability to influence transmission of information to selected targets independently of the parts of the synapse upon which it exerts its action.

Cannabinoid receptor 1 (CB1), is a G protein-coupled cannabinoid receptor that in humans is encoded by the CNR1 gene. The human CB1 receptor is expressed in the peripheral nervous system and central nervous system. It is activated by endocannabinoids, a group of retrograde neurotransmitters that include anandamide and 2-arachidonoylglycerol (2-AG); plant phytocannabinoids, such as docosatetraenoylethanolamide found in wild daga, the compound THC which is an active constituent of the psychoactive drug cannabis; and synthetic analogs of THC. CB1 is antagonized by the phytocannabinoid tetrahydrocannabivarin (THCV).

The ganglionic eminence (GE) is a transitory structure in the development of the nervous system that guides cell and axon migration. It is present in the embryonic and fetal stages of neural development found between the thalamus and caudate nucleus.

The name granule cell has been used for a number of different types of neurons whose only common feature is that they all have very small cell bodies. Granule cells are found within the granular layer of the cerebellum, the dentate gyrus of the hippocampus, the superficial layer of the dorsal cochlear nucleus, the olfactory bulb, and the cerebral cortex.
An autapse is a chemical or electrical synapse from a neuron onto itself. It can also be described as a synapse formed by the axon of a neuron on its own dendrites, in vivo or in vitro.
An axo-axonic synapse is a type of synapse, formed by one neuron projecting its axon terminals onto another neuron's axon.
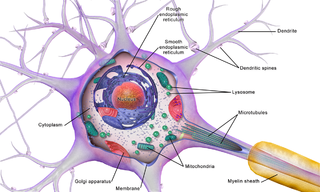
Brain cells make up the functional tissue of the brain. The rest of the brain tissue is structural or connective called the stroma which includes blood vessels. The two main types of cells in the brain are neurons, also known as nerve cells, and glial cells, also known as neuroglia.
References
- ↑ Bezaire, Marianne J.; Soltesz, Ivan (September 2013). "Quantitative assessment of CA1 local circuits: knowledge base for interneuron-pyramidal cell connectivity". Hippocampus. 23 (9): 751–785. doi:10.1002/hipo.22141. ISSN 1098-1063. PMC 3775914 . PMID 23674373.
- 1 2 3 4 Chris J. McBain; Overstreet-Wadiche, Linda (August 2015). "Neurogliaform cells in cortical circuits". Nature Reviews Neuroscience. 16 (8): 458–468. doi:10.1038/nrn3969. ISSN 1471-0048. PMC 5207343 . PMID 26189693.
- ↑ Jiang, Xiaolong; Wang, Guangfu; Lee, Alice J.; Stornetta, Ruth L.; Zhu, J. Julius (February 2013). "The organization of two novel cortical interneuronal circuits". Nature Neuroscience. 16 (2): 210–218. doi:10.1038/nn.3305. ISSN 1097-6256. PMC 3589105 . PMID 23313910.
- ↑ Muñoz-Manchado, A. B.; Foldi, C.; Szydlowski, S.; Sjulson, L.; Farries, M.; Wilson, C.; Silberberg, G.; Hjerling-Leffler, J. (2016-01-01). "Novel Striatal GABAergic Interneuron Populations Labeled in the 5HT3aEGFP Mouse". Cerebral Cortex. 26 (1): 96–105. doi:10.1093/cercor/bhu179. ISSN 1047-3211. PMC 4677971 . PMID 25146369.
- ↑ Oláh, Szabolcs; Füle, Miklós; Komlósi, Gergely; Varga, Csaba; Báldi, Rita; Barzó, Pál; Tamás, Gábor (2009-10-29). "Regulation of cortical microcircuits by unitary GABA-mediated volume transmission". Nature. 461 (7268): 1278–1281. Bibcode:2009Natur.461.1278O. doi:10.1038/nature08503. ISSN 1476-4687. PMC 2771344 . PMID 19865171.