Related Research Articles

The nitrogen cycle is the biogeochemical cycle by which nitrogen is converted into multiple chemical forms as it circulates among atmosperic, terrestrial, and marine ecosystems. The conversion of nitrogen can be carried out through both biological and physical processes. Important processes in the nitrogen cycle include fixation, ammonification, nitrification, and denitrification. The majority of Earth's atmosphere (78%) is atmospheric nitrogen, making it the largest source of nitrogen. However, atmospheric nitrogen has limited availability for biological use, leading to a scarcity of usable nitrogen in many types of ecosystems.
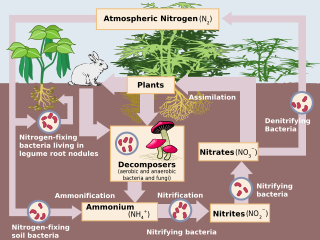
Nitrification is the biological oxidation of ammonia to nitrite followed by the oxidation of the nitrite to nitrate occurring through separate organisms or direct ammonia oxidation to nitrate in comammox bacteria. The transformation of ammonia to nitrite is usually the rate limiting step of nitrification. Nitrification is an important step in the nitrogen cycle in soil. Nitrification is an aerobic process performed by small groups of autotrophic bacteria and archaea.

Denitrification is a microbially facilitated process where nitrate (NO3−) is reduced and ultimately produces molecular nitrogen (N2) through a series of intermediate gaseous nitrogen oxide products. Facultative anaerobic bacteria perform denitrification as a type of respiration that reduces oxidized forms of nitrogen in response to the oxidation of an electron donor such as organic matter. The preferred nitrogen electron acceptors in order of most to least thermodynamically favorable include nitrate (NO3−), nitrite (NO2−), nitric oxide (NO), nitrous oxide (N2O) finally resulting in the production of dinitrogen (N2) completing the nitrogen cycle. Denitrifying microbes require a very low oxygen concentration of less than 10%, as well as organic C for energy. Since denitrification can remove NO3−, reducing its leaching to groundwater, it can be strategically used to treat sewage or animal residues of high nitrogen content. Denitrification can leak N2O, which is an ozone-depleting substance and a greenhouse gas that can have a considerable influence on global warming.

Anammox, an abbreviation for anaerobic ammonium oxidation, is a globally important microbial process of the nitrogen cycle that takes place in many natural environments. The bacteria mediating this process were identified in 1999, and were a great surprise for the scientific community. In the anammox reaction, nitrite and ammonium ions are converted directly into diatomic nitrogen and water.
Isotopic labeling is a technique used to track the passage of an isotope through a reaction, metabolic pathway, or cell. The reactant is 'labeled' by replacing specific atoms by their isotope. The reactant is then allowed to undergo the reaction. The position of the isotopes in the products is measured to determine the sequence the isotopic atom followed in the reaction or the cell's metabolic pathway. The nuclides used in isotopic labeling may be stable nuclides or radionuclides. In the latter case, the labeling is called radiolabeling.

Nitrosomonas is a genus of Gram-negative bacteria, belonging to the Betaproteobacteria. It is one of the five genera of ammonia-oxidizing bacteria and, as an obligate chemolithoautotroph, uses ammonia as an energy source and as a carbon source in presence of oxygen. Nitrosomonas are important in the global biogeochemical nitrogen cycle, since they increase the bioavailability of nitrogen to plants and in the denitrification, which is important for the release of nitrous oxide, a powerful greenhouse gas. This microbe is photophobic, and usually generate a biofilm matrix, or form clumps with other microbes, to avoid light. Nitrosomonas can be divided into six lineages: the first one includes the species Nitrosomonas europea, Nitrosomonas eutropha, Nitrosomonas halophila, and Nitrosomonas mobilis. The second lineage presents the species Nitrosomonas communis, N. sp. I and N. sp. II, meanwhile the third lineage includes only Nitrosomonas nitrosa. The fourth lineage includes the species Nitrosomonas ureae and Nitrosomonas oligotropha and the fifth and sixth lineages include the species Nitrosomonas marina, N. sp. III, Nitrosomonas estuarii and Nitrosomonas cryotolerans.
Isotopologues are molecules that differ only in their isotopic composition. They have the same chemical formula and bonding arrangement of atoms, but at least one atom has a different number of neutrons than the parent.
In atmospheric chemistry, NOx is the term for the nitrogen oxides that are most relevant for air pollution, namely nitric oxide (NO) and nitrogen dioxide. These gases contribute to the formation of smog and acid rain, as well as affecting tropospheric ozone.
Denitrifying bacteria are a diverse group of bacteria that encompass many different phyla. This group of bacteria, together with denitrifying fungi and archaea, is capable of performing denitrification as part of the nitrogen cycle. Denitrification is performed by a variety of denitrifying bacteria that are widely distributed in soils and sediments and that use oxidized nitrogen compounds in absence of oxygen as a terminal electron acceptor. They metabolise nitrogenous compounds using various enzymes, turning nitrogen oxides back to nitrogen gas or nitrous oxide.

Soil biology is the study of microbial and faunal activity and ecology in soil. Soil life, soil biota, soil fauna, or edaphon is a collective term that encompasses all organisms that spend a significant portion of their life cycle within a soil profile, or at the soil-litter interface. These organisms include earthworms, nematodes, protozoa, fungi, bacteria, different arthropods, as well as some reptiles, and species of burrowing mammals like gophers, moles and prairie dogs. Soil biology plays a vital role in determining many soil characteristics. The decomposition of organic matter by soil organisms has an immense influence on soil fertility, plant growth, soil structure, and carbon storage. As a relatively new science, much remains unknown about soil biology and its effect on soil ecosystems.
Paracoccus denitrificans, is a coccoid bacterium known for its nitrate reducing properties, its ability to replicate under conditions of hypergravity and for being a relative of the eukaryotic mitochondrion.
Simultaneous nitrification–denitrification (SNdN) is a wastewater treatment process. Microbial simultaneous nitrification-denitrification is the conversion of the ammonium ion to nitrogen gas in a single bioreactor. The process is dependent on floc characteristics, reaction kinetics, mass loading of readily biodegradable chemical oxygen demand, rbCOD, and the dissolved oxygen, DO, concentration

Human impact on the nitrogen cycle is diverse. Agricultural and industrial nitrogen (N) inputs to the environment currently exceed inputs from natural N fixation. As a consequence of anthropogenic inputs, the global nitrogen cycle (Fig. 1) has been significantly altered over the past century. Global atmospheric nitrous oxide (N2O) mole fractions have increased from a pre-industrial value of ~270 nmol/mol to ~319 nmol/mol in 2005. Human activities account for over one-third of N2O emissions, most of which are due to the agricultural sector. This article is intended to give a brief review of the history of anthropogenic N inputs, and reported impacts of nitrogen inputs on selected terrestrial and aquatic ecosystems.
Aerobic denitrification or co-respiration the simultaneous use of both oxygen (O2) and nitrate (NO3−) as oxidizing agents, performed by various genera of microorganisms. This process differs from anaerobic denitrification not only in its insensitivity to the presence of oxygen, but also in that it has a higher potential to create the harmful byproduct nitrous oxide.
Mary K. Firestone is a professor of soil microbiology in the Department of Environmental Studies, Policy, and Management at the University of California, Berkeley and a member of the National Academy of Sciences. Her laboratory's research focuses on the ecology of microbes in various soils, and their contribution to the carbon cycle and nitrogen cycle in particular.

Urine patches in cattle pastures generate large concentrations of the greenhouse gas nitrous oxide through nitrification and denitrification processes in urine-contaminated soils. Over the past few decades, the cattle population has increased more rapidly than the human population. Between the years 2000 and 2050, the cattle population is expected to increase from 1.5 billion to 2.6 billion. When large populations of cattle are packed into pastures, excessive amounts of urine soak into soils. This increases the rate at which nitrification and denitrification occur and produce nitrous oxide. Currently, nitrous oxide is one of the single most important ozone-depleting emissions and is expected to remain the largest throughout the 21st century.
Dissimilatory nitrate reduction to ammonium (DNRA), also known as nitrate/nitrite ammonification, is the result of anaerobic respiration by chemoorganoheterotrophic microbes using nitrate (NO3−) as an electron acceptor for respiration. In anaerobic conditions microbes which undertake DNRA oxidise organic matter and use nitrate (rather than oxygen) as an electron acceptor, reducing it to nitrite, then ammonium (NO3−→NO2−→NH4+).
Wendy Yang is an associate professor of Plant Biology and Geology at the University of Illinois Urbana-Champaign where she works on soil biogeochemistry and ecosystem ecology.
Bess Ward is an American oceanographer, biogeochemist, microbiologist, and William J. Sinclair Professor of Geosciences at Princeton University.
Seventeen elements or nutrients are essential for plant growth and reproduction. They are carbon (C), hydrogen (H), oxygen (O), nitrogen (N), phosphorus (P), potassium (K), sulfur (S), calcium (Ca), magnesium (Mg), iron (Fe), boron (B), manganese (Mn), copper (Cu), zinc (Zn), molybdenum (Mo), nickel (Ni) and chlorine (Cl). Nutrients required for plants to complete their life cycle are considered essential nutrients. Nutrients that enhance the growth of plants but are not necessary to complete the plant's life cycle are considered non-essential. With the exception of carbon, hydrogen and oxygen, which are supplied by carbon dioxide and water, and nitrogen, provided through nitrogen fixation, the nutrients derive originally from the mineral component of the soil. The Law of the Minimum expresses that when the available form of a nutrient is not in enough proportion in the soil solution, then other nutrients cannot be taken up at an optimum rate by a plant. A particular nutrient ratio of the soil solution is thus mandatory for optimizing plant growth, a value which might differ from nutrient ratios calculated from plant composition.
References
- ↑ Hart, Stephen C.; Myrold, David D. (1996). "15N Tracer Studies of Soil Nitrogen Transformations". Mass Spectrometry of Soils.
- ↑ Bateman, E. J.; Baggs, E. M. (2005-03-23). "Contributions of nitrification and denitrification to N2O emissions from soils at different water-filled pore space". Biology and Fertility of Soils. 41 (6): 379–388. doi:10.1007/s00374-005-0858-3. ISSN 0178-2762. S2CID 34419659.
- ↑ Müller, Christoph; Laughlin, Ronnie J.; Spott, Oliver; Rütting, Tobias (2014-05-01). "Quantification of N2O emission pathways via a 15N tracing model". Soil Biology and Biochemistry. 72: 44–54. doi:10.1016/j.soilbio.2014.01.013.
- ↑ Köster, Jan Reent; Cárdenas, Laura; Senbayram, Mehmet; Bol, Roland; Well, Reinhard; Butler, Mark; Mühling, Karl Hermann; Dittert, Klaus (2011-08-01). "Rapid shift from denitrification to nitrification in soil after biogas residue application as indicated by nitrous oxide isotopomers". Soil Biology and Biochemistry. 43 (8): 1671–1677. doi:10.1016/j.soilbio.2011.04.004.
- ↑ Baggs, E. M. (2008-06-15). "A review of stable isotope techniques for N2O source partitioning in soils: recent progress, remaining challenges and future considerations". Rapid Communications in Mass Spectrometry. 22 (11): 1664–1672. Bibcode:2008RCMS...22.1664B. doi: 10.1002/rcm.3456 . ISSN 1097-0231. PMID 18435506.
- ↑ Rütting, T.; Müller, C. (2008-04-01). "Process-specific analysis of nitrite dynamics in a permanent grassland soil by using a Monte Carlo sampling technique". European Journal of Soil Science. 59 (2): 208–215. doi:10.1111/j.1365-2389.2007.00976.x. ISSN 1365-2389.
- ↑ Müller, Christoph; Laughlin, Ronnie J.; Spott, Oliver; Rütting, Tobias (2014-05-01). "Quantification of N2O emission pathways via a 15N tracing model". Soil Biology and Biochemistry. 72: 44–54. doi:10.1016/j.soilbio.2014.01.013.