Related Research Articles
Ronald Mark Evans is an American Biologist, Professor and Head of the Salk’s Gene Expression Laboratory, and the March of Dimes Chair in Molecular and Developmental Biology at the Salk Institute for Biological Studies in La Jolla, California and a Howard Hughes Medical Institute Investigator. Dr. Ronald M. Evans is known for his original discoveries of nuclear hormone receptors (NR), a special class of transcriptional factor, and the elucidation of their universal mechanism of action, a process that governs how lipophilic hormones and drugs regulate virtually every developmental and metabolic pathway in animals and humans. Nowadays, NRs are among the most widely investigated group of pharmaceutical targets in the world, already yielding benefits in drug discovery for cancer, muscular dystrophies, osteoporosis, type II diabetes, obesity, and cardiovascular diseases. His current research focuses on the function of nuclear hormone signaling and their function in metabolism and cancer.

A selective progesterone receptor modulator (SPRM) is an agent that acts on the progesterone receptor (PR), the biological target of progestogens like progesterone. A characteristic that distinguishes such substances from full receptor agonists and full antagonists is that their action differs in different tissues, i.e. agonist in some tissues while antagonist in others. This mixed profile of action leads to stimulation or inhibition in tissue-specific manner, which further raises the possibility of dissociating undesirable adverse effects from the development of synthetic PR-modulator drug candidates.
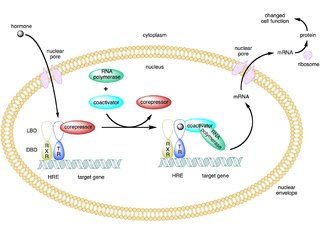
A coactivator is a type of transcriptional coregulator that binds to an activator to increase the rate of transcription of a gene or set of genes. The activator contains a DNA binding domain that binds either to a DNA promoter site or a specific DNA regulatory sequence called an enhancer. Binding of the activator-coactivator complex increases the speed of transcription by recruiting general transcription machinery to the promoter, therefore increasing gene expression. The use of activators and coactivators allows for highly specific expression of certain genes depending on cell type and developmental stage.

The vitamin D receptor (VDR also known as the calcitriol receptor) is a member of the nuclear receptor family of transcription factors. Calcitriol (the active form of vitamin D, 1,25-(OH)2vitamin D3) binds to VDR, which then forms a heterodimer with the retinoid-X receptor. The VDR heterodimer then enters the nucleus and binds to Vitamin D responsive elements (VDRE) in genomic DNA. VDR binding results in expression or transrepression of many specific gene products. VDR is also involved in microRNA-directed post transcriptional mechanisms. In humans, the vitamin D receptor is encoded by the VDR gene located on chromosome 12q13.11.

In the field of molecular biology, the pregnane X receptor (PXR), also known as the steroid and xenobiotic sensing nuclear receptor (SXR) or nuclear receptor subfamily 1, group I, member 2 (NR1I2) is a protein that in humans is encoded by the NR1I2 gene.
In the field of molecular biology, nuclear receptors are a class of proteins responsible for sensing steroids, thyroid hormones, vitamins, and certain other molecules. These intracellular receptors work with other proteins to regulate the expression of specific genes thereby controlling the development, homeostasis, and metabolism of the organism.

P300/CBP-associated factor (PCAF), also known as K(lysine) acetyltransferase 2B (KAT2B), is a human gene and transcriptional coactivator associated with p53.

TRAF6 is a TRAF human protein.
The nuclear receptor coactivator 1 (NCOA1) is a transcriptional coregulatory protein that contains several nuclear receptor interacting domains and an intrinsic histone acetyltransferase activity. NCOA1 is recruited to DNA promotion sites by ligand-activated nuclear receptors. NCOA1, in turn, acylates histones, which makes downstream DNA more accessible to transcription. Hence, NCOA1 assists nuclear receptors in the upregulation of DNA expression.

The nuclear receptor coactivator 3 also known as NCOA3 is a protein that, in humans, is encoded by the NCOA3 gene. NCOA3 is also frequently called 'amplified in breast 1' (AIB1), steroid receptor coactivator-3 (SRC-3), or thyroid hormone receptor activator molecule 1 (TRAM-1).

The nuclear receptor co-repressor 1 also known as thyroid-hormone- and retinoic-acid-receptor-associated co-repressor 1 (TRAC-1) is a protein that in humans is encoded by the NCOR1 gene.

The nuclear receptor co-repressor 2 (NCOR2) is a transcriptional coregulatory protein that contains several nuclear receptor-interacting domains. In addition, NCOR2 appears to recruit histone deacetylases to DNA promoter regions. Hence NCOR2 assists nuclear receptors in the down regulation of target gene expression. NCOR2 is also referred to as a silencing mediator for retinoid or thyroid-hormone receptors (SMRT) or T3 receptor-associating cofactor 1 (TRAC-1).

Retinoid X receptor alpha (RXR-alpha), also known as NR2B1 is a nuclear receptor that in humans is encoded by the RXRA gene.

Inhibitor of nuclear factor kappa-B kinase subunit alpha (IKK-α) also known as IKK1 or conserved helix-loop-helix ubiquitous kinase (CHUK) is a protein kinase that in humans is encoded by the CHUK gene. IKK-α is part of the IκB kinase complex that plays an important role in regulating the NF-κB transcription factor. However, IKK-α has many additional cellular targets, and is thought to function independently of the NF-κB pathway to regulate epidermal differentiation.

Retinoic acid receptor alpha (RAR-α), also known as NR1B1 is a nuclear receptor that in humans is encoded by the RARA gene.

Retinoid X receptor beta (RXR-beta), also known as NR2B2 is a nuclear receptor that in humans is encoded by the RXRB gene.
Peroxisome proliferator-activated receptor delta(PPAR-delta), or (PPAR-beta), also known as Nuclear hormone receptor 1(NUC1) is a nuclear receptor that in humans is encoded by the PPARD gene.

COUP-TF1 also known as NR2F1 is a protein that in humans is encoded by the NR2F1 gene. This protein is a member of nuclear hormone receptor family of steroid hormone receptors.

Mitogen-activated protein kinase kinase kinase 14 also known as NF-kappa-B-inducing kinase (NIK) is an enzyme that in humans is encoded by the MAP3K14 gene.
Nuclear receptor coregulators are a class of transcription coregulators that have been shown to be involved in any aspect of signaling by any member of the nuclear receptor superfamily. A comprehensive database of coregulators for nuclear receptors and other transcription factors was previously maintained at the Nuclear Receptor Signaling Atlas website which has since been replaced by the Signaling Pathways Project website.
References
- ↑ Margolis RN, Evans RM, O'Malley BW (October 2005). "The Nuclear Receptor Signaling Atlas: development of a functional atlas of nuclear receptors". Mol. Endocrinol. 19 (10): 2433–6. doi: 10.1210/me.2004-0461 . PMID 16051673.
- ↑ Lanz RB, Jericevic Z, Zuercher WJ, Watkins C, Steffen DL, Margolis R, McKenna NJ (January 2006). "Nuclear Receptor Signaling Atlas (): hyperlinking the nuclear receptor signaling community". Nucleic Acids Res. 34 (Database issue): D221–6. doi:10.1093/nar/gkj029. PMC 1347392 . PMID 16381851.
- ↑ Ochsner SA, Abraham D, Martin K, Ding W, McOwiti A, Kankanamge W, Wang Z, Andreano K, Hamilton RA, Chen Y, Hamilton A, Gantner ML, Dehart M, Qu S, Hilsenbeck SG, Becnel LB, Bridges D, Ma'ayan A, Huss JM, Stossi F, Foulds CE, Kralli A, McDonnell DP, McKenna NJ (October 2019). "The Signaling Pathways Project, an integrated 'omics knowledgebase for mammalian cellular signaling pathways". Scientific Data. 6 (1): 252. Bibcode:2019NatSD...6..252O. doi:10.1038/s41597-019-0193-4. PMC 6823428 . PMID 31672983.