
A fullerene is an allotrope of carbon whose molecule consists of carbon atoms connected by single and double bonds so as to form a closed or partially closed mesh, with fused rings of five to seven atoms. The molecule may be a hollow sphere, ellipsoid, tube, or many other shapes and sizes. Graphene, which is a flat mesh of regular hexagonal rings, can be seen as an extreme member of the family.
Monosaccharides, also called simple sugars, are the simplest form of sugar and the most basic units of carbohydrates. They cannot be further hydrolyzed to simpler chemical compounds. The general formula is C
nH
2nO
n. They are usually colorless, water-soluble, and crystalline solids. Some monosaccharides have a sweet taste.
Cyclopropane is the cycloalkane molecule with the molecular formula C3H6, consisting of three carbon atoms linked to each other to form a ring, with each carbon atom bearing two hydrogen atoms resulting in D3h molecular symmetry. The small size of the ring creates substantial ring strain in the structure.
Octanitrocubane (molecular formula: C8(NO2)8) is a high explosive that, like TNT, is shock-insensitive (not readily detonated by shock). The octanitrocubane molecule has the same chemical structure as cubane (C8H8) except that each of the eight hydrogen atoms is replaced by a nitro group (NO2).
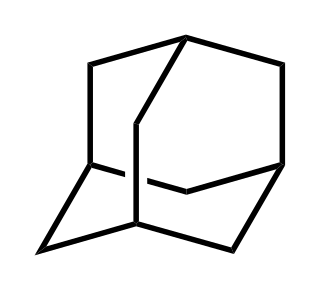
Adamantane is a colorless, crystalline chemical compound with a camphor-like odor. With a formula C10H16, it is a cycloalkane and also the simplest diamondoid. Adamantane molecules consists of three connected cyclohexane rings arranged in the "armchair" configuration. It is unique in that it is both rigid and virtually stress-free. Adamantane is the most stable among all the isomers with formula C10H16, which include the somewhat similar twistane. The spatial arrangement of carbon atoms in the adamantane molecule is the same as in the diamond crystal. This motivates the name adamantane, which is derived from the Greek adamantinos (relating to steel or diamond).
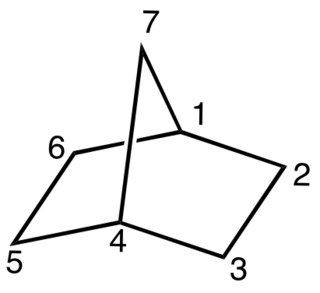
A bicyclic molecule is a molecule that features two joined rings. Bicyclic structures occur widely, for example in many biologically important molecules like α-thujene and camphor. A bicyclic compound can be carbocyclic, or heterocyclic, like DABCO. Moreover, the two rings can both be aliphatic, or can be aromatic, or a combination of aliphatic and aromatic.
Dodecahedrane is a chemical compound, a hydrocarbon with formula C
20H
20, whose carbon atoms are arranged as the vertices (corners) of a regular dodecahedron. Each carbon is bound to three neighbouring carbon atoms and to a hydrogen atom. This compound is one of the three possible Platonic hydrocarbons, the other two being cubane and tetrahedrane.

In organic chemistry, ring strain is a type of instability that exists when bonds in a molecule form angles that are abnormal. Strain is most commonly discussed for small rings such as cyclopropanes and cyclobutanes, whose internal angles are substantially smaller than the idealized value of approximately 109°. Because of their high strain, the heat of combustion for these small rings is elevated.

Prismane or 'Ladenburg benzene' is a polycyclic hydrocarbon with the formula C6H6. It is an isomer of benzene, specifically a valence isomer. Prismane is far less stable than benzene. The carbon (and hydrogen) atoms of the prismane molecule are arranged in the shape of a six-atom triangular prism—this compound is the parent and simplest member of the prismanes class of molecules. Albert Ladenburg proposed this structure for the compound now known as benzene. The compound was not synthesized until 1973.

Bredt's rule is an empirical observation in organic chemistry that states that a double bond cannot be placed at the bridgehead of a bridged ring system, unless the rings are large enough. The rule is named after Julius Bredt, who first discussed it in 1902 and codified it in 1924. It primarily relates to bridgeheads with carbon-carbon and carbon-nitrogen double bonds.

In a tetrahedral molecular geometry, a central atom is located at the center with four substituents that are located at the corners of a tetrahedron. The bond angles are cos−1(−⅓) = 109.4712206...° ≈ 109.5° when all four substituents are the same, as in methane (CH4) as well as its heavier analogues. Methane and other perfectly symmetrical tetrahedral molecules belong to point group Td, but most tetrahedral molecules have lower symmetry. Tetrahedral molecules can be chiral.

Dicarbon monoxide (C2O) is a molecule that contains two carbon atoms and one oxygen atom. It is a linear molecule that, because of its simplicity, is of interest in a variety of areas. It is, however, so extremely reactive that it is not encountered in everyday life. It is classified as a cumulene and an oxocarbon.

In the analysis of the molecular formula of organic molecules, the degree of unsaturation is a calculation that determines the total number of rings and π bonds. A formula is used in organic chemistry to help draw chemical structures. It does not give any information about those components individually—the specific number of rings, or of double bonds, or of triple bonds. The final structure is verified with use of NMR, mass spectrometry and IR spectroscopy, as well as qualitative inspection. It is based on comparing the actual molecular formula to what would be a possible formula if the structure were saturated—having no rings and containing only σ bonds—with all atoms having their standard valence.

A bridged compound is a chemical compound, generally an organic compound, but also possibly inorganic, that has two or more rings that contains a bridge—a single atom or an unbranched chain of atoms that connect two "bridgehead" atoms. The bridgehead atoms are defined as any atom that is not a hydrogen, and that is part of the skeletal framework of the molecule that is bonded to three or more other skeletal atoms. Clayden simplifies this description, saying that bridged bicyclic compounds "are just what the name implies—[molecules in which] a bridge of [an atom or of atoms] is thrown across from one side of the ring to the other," examples of which are shown at left and right, and below. The presence of the bridge connecting the bridgehead atoms, which are most often two non-adjacent atoms, distinguishes bridged compounds from fused ring compounds that have two rings linked by two adjacent atoms, and from spiro compounds that have two rings linked by a single atom.

[1.1.1]Propellane is an organic compound, the simplest member of the propellane family. It is a hydrocarbon with formula C5H6 or C2(CH2)3. The molecular structure consists of three rings of three carbon atoms each, sharing one C–C bond.

[2.2.2]Propellane, formally tricyclo[2.2.2.01,4]octane is an organic compound, a member of the propellane family. It is a hydrocarbon with formula C8H12, or C2(C2H4)3. Its molecule has three rings with four carbon atoms each, sharing one C–C bond.

Pagodane is an organic compound with formula C
20H
20 whose carbon skeleton was said to resemble a pagoda, hence the name. It is a polycyclic hydrocarbon whose molecule has the D2h point symmetry group. The compound is a highly crystalline solid that melts at 243 °C, is barely soluble in most organic solvents and moderately soluble in benzene and chloroform. It sublimes at low pressure.
The charge-shift bond has been proposed as a new class of chemical bond that sits alongside the three familiar families of covalent, ionic bonds, and metallic bonds where electrons are shared or transferred respectively. The charge shift bond derives its stability from the resonance of ionic forms rather than the covalent sharing of electrons which are often depicted as having electron density between the bonded atoms. A feature of the charge shift bond is that the predicted electron density between the bonded atoms is low. It has long been known from experiment that the accumulation of electronic charge between the bonded atoms is not necessarily a feature of covalent bonds. An example where charge shift bonding has been used to explain the low electron density found experimentally is in the central bond between the inverted tetrahedral carbons in [1.1.1]propellanes. Theoretical calculations on a range of molecules have indicated that a charge shift bond is present, a striking example being fluorine, F2, which is normally described as having a typical covalent bond.
![General structure of [m.n.o.p]paddlane General paddlane.svg](http://upload.wikimedia.org/wikipedia/commons/thumb/0/00/General_paddlane.svg/440px-General_paddlane.svg.png)


![[1.1.1.1]Paddlane 1111paddlane.svg](http://upload.wikimedia.org/wikipedia/commons/thumb/e/ec/1111paddlane.svg/260px-1111paddlane.svg.png)














