Related Research Articles
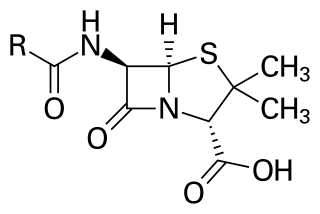
Penicillins are a group of β-lactam antibiotics originally obtained from Penicillium moulds, principally P. chrysogenum and P. rubens. Most penicillins in clinical use are synthesised by P. chrysogenum using deep tank fermentation and then purified. A number of natural penicillins have been discovered, but only two purified compounds are in clinical use: penicillin G and penicillin V. Penicillins were among the first medications to be effective against many bacterial infections caused by staphylococci and streptococci. They are still widely used today for different bacterial infections, though many types of bacteria have developed resistance following extensive use.
Peptidoglycan or murein is a unique large macromolecule, a polysaccharide, consisting of sugars and amino acids that forms a mesh-like layer (sacculus) that surrounds the bacterial cytoplasmic membrane. The sugar component consists of alternating residues of β-(1,4) linked N-acetylglucosamine (NAG) and N-acetylmuramic acid (NAM). Attached to the N-acetylmuramic acid is an oligopeptide chain made of three to five amino acids. The peptide chain can be cross-linked to the peptide chain of another strand forming the 3D mesh-like layer. Peptidoglycan serves a structural role in the bacterial cell wall, giving structural strength, as well as counteracting the osmotic pressure of the cytoplasm. This repetitive linking results in a dense peptidoglycan layer which is critical for maintaining cell form and withstanding high osmotic pressures, and it is regularly replaced by peptidoglycan production. Peptidoglycan hydrolysis and synthesis are two processes that must occur in order for cells to grow and multiply, a technique carried out in three stages: clipping of current material, insertion of new material, and re-crosslinking of existing material to new material.
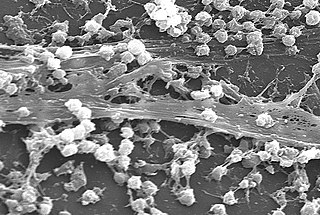
A biofilm is a syntrophic community of microorganisms in which cells stick to each other and often also to a surface. These adherent cells become embedded within a slimy extracellular matrix that is composed of extracellular polymeric substances (EPSs). The cells within the biofilm produce the EPS components, which are typically a polymeric combination of extracellular polysaccharides, proteins, lipids and DNA. Because they have a three-dimensional structure and represent a community lifestyle for microorganisms, they have been metaphorically described as "cities for microbes".
Lantibiotics are a class of polycyclic peptide antibiotics that contain the characteristic thioether amino acids lanthionine or methyllanthionine, as well as the unsaturated amino acids dehydroalanine, and 2-aminoisobutyric acid. They belong to ribosomally synthesized and post-translationally modified peptides.
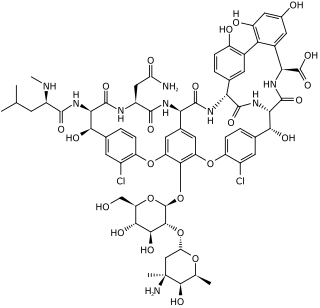
Glycopeptide antibiotics are a class of drugs of microbial origin that are composed of glycosylated cyclic or polycyclic nonribosomal peptides. Significant glycopeptide antibiotics include the anti-infective antibiotics vancomycin, teicoplanin, telavancin, ramoplanin and decaplanin, corbomycin, complestatin and the antitumor antibiotic bleomycin. Vancomycin is used if infection with methicillin-resistant Staphylococcus aureus (MRSA) is suspected.

Defensins are small cysteine-rich cationic proteins across cellular life, including vertebrate and invertebrate animals, plants, and fungi. They are host defense peptides, with members displaying either direct antimicrobial activity, immune signaling activities, or both. They are variously active against bacteria, fungi and many enveloped and nonenveloped viruses. They are typically 18-45 amino acids in length, with three or four highly conserved disulphide bonds.

Antimicrobial peptides (AMPs), also called host defence peptides (HDPs) are part of the innate immune response found among all classes of life. Fundamental differences exist between prokaryotic and eukaryotic cells that may represent targets for antimicrobial peptides. These peptides are potent, broad spectrum antimicrobials which demonstrate potential as novel therapeutic agents. Antimicrobial peptides have been demonstrated to kill Gram negative and Gram positive bacteria, enveloped viruses, fungi and even transformed or cancerous cells. Unlike the majority of conventional antibiotics it appears that antimicrobial peptides frequently destabilize biological membranes, can form transmembrane channels, and may also have the ability to enhance immunity by functioning as immunomodulators.
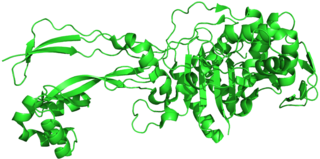
Penicillin-binding proteins (PBPs) are a group of proteins that are characterized by their affinity for and binding of penicillin. They are a normal constituent of many bacteria; the name just reflects the way by which the protein was discovered. All β-lactam antibiotics bind to PBPs, which are essential for bacterial cell wall synthesis. PBPs are members of a subgroup of enzymes called transpeptidases. Specifically, PBPs are DD-transpeptidases.
Cathelicidin antimicrobial peptide (CAMP) is an antimicrobial peptide encoded in the human by the CAMP gene. The active form is LL-37. In humans, CAMP encodes the peptide precursor CAP-18, which is processed by proteinase 3-mediated extracellular cleavage into the active form LL-37.

Beta-defensin 2 (BD-2) also known as skin-antimicrobial peptide 1 (SAP1) is a peptide that in humans is encoded by the DEFB4 gene.

Alpha defensins are a family of mammalian defensin peptides of the alpha subfamily. They are also known as cryptdins and are produced within the small bowel. Cryptdin is a portmanteau of crypt and defensin.

Polypeptide antibiotics are a chemically diverse class of anti-infective and antitumor antibiotics containing non-protein polypeptide chains. Examples of this class include actinomycin, bacitracin, colistin, and polymyxin B. Actinomycin-D has found use in cancer chemotherapy. Most other polypeptide antibiotics are too toxic for systemic administration, but can safely be administered topically to the skin as an antiseptic for shallow cuts and abrasions.
Protegrins are small peptides containing 16-18 amino acid residues. Protegrins were first discovered in porcine leukocytes and were found to have antimicrobial activity against bacteria, fungi, and some enveloped viruses. The amino acid composition of protegrins contains six positively charged arginine residues and four cysteine residues. Their secondary structure is classified as cysteine-rich β-sheet antimicrobial peptides, AMPs, that display limited sequence similarity to certain defensins and tachyplesins. In solution, the peptides fold to form an anti-parallel β-strand with the structure stabilized by two cysteine bridges formed among the four cysteine residues. Recent studies suggest that protegrins can bind to lipopolysaccharide, a property that may help them to insert into the membranes of gram-negative bacteria and permeabilize them.
Sortases are membrane anchored enzyme that sort these surface proteins onto the bacterial cell surface and anchor them to the peptidoglycan. There are different types of sortases and each catalyse the anchoring of different proteins to cell walls.
Brilacidin, an investigational new drug, is a polymer-based antibiotic currently in human clinical trials, and represents a new class of antibiotics called host defense protein mimetics, or HDP-mimetics, which are non-peptide synthetic small molecules modeled after host defense peptides (HDPs). HDPs, also called antimicrobial peptides, some of which are defensins, are part of the innate immune response and are common to most higher forms of life. As brilacidin is modeled after a defensin, it is also called a defensin mimetic.
Teixobactin is a peptide-like secondary metabolite of some species of bacteria, that kills some gram-positive bacteria. It appears to belong to a new class of antibiotics, and harms bacteria by binding to lipid II and lipid III, important precursor molecules for forming the cell wall.
Eleftheria terrae is a recently discovered Gram-negative bacterium. E. terrae is a temporary name for the organism, as it was only discovered in 2014 and is still undergoing scientific study. It was found to produce a previously unknown antibiotic named teixobactin. The discovery of E. terrae could represent a new age of antibiotics, as teixobactin is the first new antibiotic discovered since the synthetic era of the 1980s. Prior research has indicated that other uncultivable bacteria like E. terrae have potential in the development of new antimicrobial agents.

Lipid II is a precursor molecule in the synthesis of the cell wall of bacteria. It is a peptidoglycan, which is amphipathic and named for its bactoprenol hydrocarbon chain, which acts as a lipid anchor, embedding itself in the bacterial cell membrane. Lipid II must translocate across the cell membrane to deliver and incorporate its disaccharide-pentapeptide "building block" into the peptidoglycan mesh. Lipid II is the target of several antibiotics.
Copsin is a fungal defensin that acts as an antimicrobial polypeptide secreted from the inky cap mushroom, first reported at the end of 2014. The fungal defensin acts against gram positive bacteria.
Heike Brötz-Oesterhelt is a German microbiologist. She is a full professor and holds the Chair of the Department for Microbial Bioactive Compounds at the Interfaculty Institute for Microbiology and Infection Medicine, University of Tübingen, Germany.
References
- ↑ Mygind, Per H.; Fischer, Rikke L.; Schnorr, Kirk M.; Hansen, Mogens T.; Sönksen, Carsten P.; Ludvigsen, Svend; Raventós, Dorotea; Buskov, Steen; Christensen, Bjarke; De Maria, Leonardo; Taboureau, Olivier (2005). "Plectasin is a peptide antibiotic with therapeutic potential from a saprophytic fungus". Nature. 437 (7061): 975–980. Bibcode:2005Natur.437..975M. doi:10.1038/nature04051. ISSN 1476-4687. PMID 16222292. S2CID 4423851.
- 1 2 "Novozymes reveals knowledge on new antibiotic against resistant bacteria". Novozymes. 2010-05-28. Retrieved 2010-05-28.
- ↑ "Plectasin NZ2114 - Novel Microbial Agent". Drug Development Technology. Retrieved 2010-05-28.
- ↑ Kristensen, HH; et al. (2010). "Plectasin, a Fungal Defensin, Targets the Bacterial Cell Wall Precursor Lipid II". Science. 328 (5982): 1168–1172. Bibcode:2010Sci...328.1168S. doi:10.1126/science.1185723. hdl: 1874/202594 . PMID 20508130. S2CID 206524928.
- ↑ Jekhmane, S., Derks. M. (2024). "Host defence peptide plectasin targets bacterial cell wall precursor lipid II by a calcium-sensitive supramolecular mechanism". Nature Microbiology. doi:10.1038/s41564-024-01696-9. PMID 38783023.
{{cite journal}}: CS1 maint: multiple names: authors list (link) - ↑ Kristensen, HH; et al. (2009). "In Vivo Pharmacodynamic Characterization of a Novel Plectasin Antibiotic, NZ2114, in a Murine Infection Model". Antimicrob. Agents Chemother. 53 (7): 3003–3009. doi:10.1128/AAC.01584-08. PMC 2704636 . PMID 19414576.