Related Research Articles

A conotoxin is one of a group of neurotoxic peptides isolated from the venom of the marine cone snail, genus Conus.

Delta atracotoxin is a low-molecular-weight neurotoxic polypeptide found in the venom of the Sydney funnel-web spider.

Grammotoxin is a toxin in the venom of the tarantula Grammostola spatulata. It is a protein toxin that inhibits P-, Q- and N-type voltage-gated calcium channels in neurons. Grammotoxin is also known as omega-grammotoxin SIA.

Heteropodatoxins are peptide toxins from the venom of the giant crab spider Heteropoda venatoria, which block Kv4.2 voltage-gated potassium channels.

Insect toxins are various protein toxins produced by insect species. Assassin bugs, sometimes known as kissing bugs, are one of the largest and morphologically diverse families of true bugs feeding on crickets, caterpillars and other insects. Some assassin bug species are bloodsucking parasites of mammals, even feed on humans. They can be commonly found throughout most of the world and their size varies from a few millimeters to as much as 3 or 4 centimeters. The toxic saliva of the predatory assassin bugs contains a complex mixture of small and large peptides for diverse uses such as immobilizing and pre-digesting their prey, as well as defense against competitors and predators. Assassin bug toxins are small peptides with disulfide connectivity that target ion channels. They are relatively homologous to the calcium channel blockers omega-conotoxins from marine cone snails and belong to the four-loop cysteine scaffold structural class.
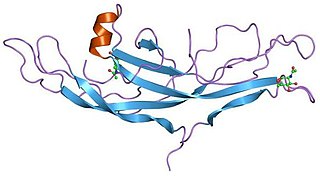
A cystine knot is a protein structural motif containing three disulfide bridges. The sections of polypeptide that occur between two of them form a loop through which a third disulfide bond passes, forming a rotaxane substructure. The cystine knot motif stabilizes protein structure and is conserved in proteins across various species. There are three types of cystine knot, which differ in the topology of the disulfide bonds:

delta-Palutoxins (δ-palutoxins) consist of a homologous group of four insect-specific toxins from the venom of the spider Pireneitega luctuosa. They show a high toxicity against Spodoptera litura larvae by inhibiting sodium channels, leading to strong paralytic activity and eventually to the death of the insect.
Theraphosa leblondi toxin (TLTx) is a toxin occurring in three different forms (subtypes) that are purified and sequenced from the venom of the giant tarantula Theraphosa blondi. This toxin selectively inhibits Kv4.2 voltage-gated potassium channels by acting as a gating modifier.
Huwentoxins (HWTX) are a group of neurotoxic peptides found in the venom of the Chinese bird spider Haplopelma schmidti. The species was formerly known as Haplopelma huwenum, Ornithoctonus huwena and Selenocosmia huwena. While structural similarity can be found among several of these toxins, HWTX as a group possess high functional diversity.
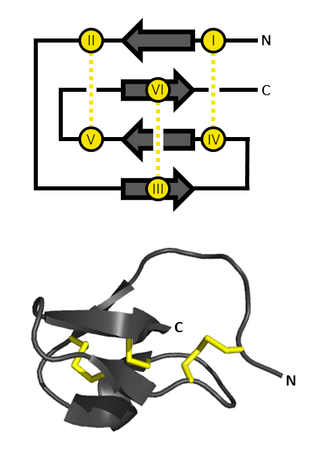
An inhibitor cystine knot is a protein structural motif containing three disulfide bridges. Knottins are one of three folds in the cystine knot motif; the other closely related knots are the growth factor cystine knot (GFCK) and the cyclic cystine knot. Types include a) cyclic mobius, b) cyclic bracelet, c) acyclic inhibitor knottins. Cystine knot motifs are found frequently in nature in a plethora of plants, animals, and fungi and serve diverse functions from appetite suppression to anti-fungal activity.
GTx1-15 is a toxin from the Chilean tarantula venom that acts as both a voltage-gated calcium channel blocker and a voltage-gated sodium channel blocker.
Protoxin-II, also known as ProTx-II, PT-II or β/ω-TRTX-Tp2a, is a neurotoxin that inhibits certain voltage-gated calcium and voltage-gated sodium channels. This toxin is a 30-residue disulfide-rich peptide that has unusually high affinity and selectivity toward the human Nav1.7. channel.
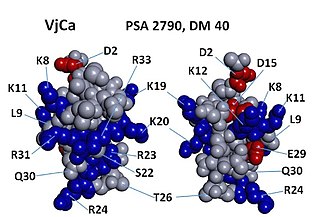
Vejocalcin is a toxin from the venom of the Mexican scorpion Vaejovis mexicanus. Vejocalcin is a member of the calcin family of toxins. It acts as a cell-penetrating peptide (CPP); it binds with high affinity and specificity to skeletal ryanodine receptor 1 (RYR1) of the sarcoplasmic reticulum, thereby triggering calcium release from intracellular Ca2+
stores.
Intrepicalcin (ViCaTx1) is a short peptide toxin found in the venom of scorpion Vaejovis intrepidus. It is one of a group of short, basic peptides called calcins, which bind to ryanodine receptors (RyRs) and thereby trigger calcium release from the sarcoplasmic reticulum.
μ-THTX-Cl6a, also known as Cl6a, is a 33-residue peptide toxin extracted from the venom of the spider Cyriopagopus longipes. The toxin acts as an inhibitor of the tetrodotoxin-sensitive (TTX-S) voltage-gated sodium channel (NaV1.7), thereby causing sustained reduction of NaV1.7 currents.
GiTx1 (β/κ-theraphotoxin-Gi1a) is a peptide toxin present in the venom of Grammostola iheringi. It reduces both inward and outward currents by blocking voltage-gated sodium and potassium channels, respectively.
Protoxin-I, also known as ProTx-I, or Beta/omega-theraphotoxin-Tp1a, is a 35-amino-acid peptide neurotoxin extracted from the venom of the tarantula Thrixopelma pruriens. Protoxin-I belongs to the inhibitory cystine knot (ICK) family of peptide toxins, which have been known to potently inhibit voltage-gated ion channels. Protoxin-I selectively blocks low voltage threshold T-type calcium channels, voltage gated sodium channels and the nociceptor cation channel TRPA1. Due to its unique ability to bind to TRPA1, Protoxin-I has been implicated as a valuable pharmacological reagent with potential applications in clinical contexts with regards to pain and inflammation
Cl6b (μ-THTX-Cl6b) is a peptide toxin from the venom of the spider Cyriopagopus longipes. It acts as a sodium channel blocker: Cl6b significantly and persistently reduces currents through the tetrodotoxin-sensitive sodium channels NaV1.2-1.4, NaV1.6, and NaV1.7.

Grammostola mechanotoxin #4, also known as M-theraphotoxin-Gr1a (M-TRTX-Gr1a), is a neurotoxin isolated from the venom of the spider Chilean rose tarantula Grammostola spatulate. This amphiphilic peptide, which consists of 35 amino acids, belongs to the inhibitory cysteine knot (ICK) peptide family. It reduces mechanical sensation by inhibiting mechanosensitive channels (MSCs).
Phlotoxin is a neurotoxin from the venom of the tarantula Phlogiellus that targets mostly voltage-sensitive sodium channels and mainly Nav1.7. The only non-sodium voltage-sensitive channel that is inhibited by Phlotoxin is Kv3.4. Nav1.4 and Nav1.6 seem to be Phlotoxin-1-sensitive to some extent as well.
References
- ↑ "Toxin Ptu1 - Peirates turpis (Assassin bug)". www.uniprot.org. UniProt . Retrieved 2019-10-26.
- ↑ Walker, Andrew A.; Weirauch, Christiane; Fry, Bryan G.; King, Glenn F. (February 2016). "Venoms of Heteropteran Insects: A Treasure Trove of Diverse Pharmacological Toolkits". Toxins. 8 (2). MDPI: 43. doi: 10.3390/toxins8020043 . PMC 4773796 . PMID 26907342.
- 1 2 3 4 5 Corzo, G.; Adachi-Akahane, S.; Nagao, T.; Kusui, Y.; Nakajima, T. (2001-06-22). "Novel peptides from assassin bugs (Hemiptera: Reduviidae): isolation, chemical and biological characterization". FEBS Letters. 499 (3): 256–261. doi:10.1016/s0014-5793(01)02558-3. ISSN 0014-5793. PMID 11423127. S2CID 45675280.
- 1 2 3 4 5 6 7 8 Bernard, C.; Corzo, G.; Mosbah, A.; Nakajima, T.; Darbon, H. (2001-10-30). "Solution structure of Ptu1, a toxin from the assassin bug Peirates turpis that blocks the voltage-sensitive calcium channel N-type". Biochemistry. 40 (43): 12795–12800. doi:10.1021/bi015537j. ISSN 0006-2960. PMID 11669615.