Related Research Articles
Unconventional superconductors are materials that display superconductivity which does not conform to either the conventional BCS theory or Nikolay Bogolyubov's theory or its extensions.

High-temperature superconductors are defined as materials that behave as superconductors at temperatures above 77 K, the boiling point of liquid nitrogen. The adjective "high temperature" is only in respect to previously known superconductors, which function at even colder temperatures close to absolute zero. In absolute terms, these "high temperatures" are still far below ambient, and therefore require cooling. The first high-temperature superconductor was discovered in 1986, by IBM researchers Bednorz and Müller, who were awarded the Nobel Prize in Physics in 1987 "for their important break-through in the discovery of superconductivity in ceramic materials". Most high-Tc materials are type-II superconductors.

A perovskite is any material with a crystal structure following the formula ABX3, which was first discovered as the mineral called perovskite, which consists of calcium titanium oxide (CaTiO3). The mineral was first discovered in the Ural mountains of Russia by Gustav Rose in 1839 and named after Russian mineralogist L. A. Perovski (1792–1856). 'A' and 'B' are two positively charged ions (i.e. cations), often of very different sizes, and X is a negatively charged ion (an anion, frequently oxide) that bonds to both cations. The 'A' atoms are generally larger than the 'B' atoms. The ideal cubic structure has the B cation in 6-fold coordination, surrounded by an octahedron of anions, and the A cation in 12-fold cuboctahedral coordination. Additional perovskite forms may exist where either/both the A and B sites have a configuration of A1x-1A2x and/or B1y-1B2y and the X may deviate from the ideal coordination configuration as ions within the A and B sites undergo changes in their oxidation states.

Yttrium barium copper oxide (YBCO) is a family of crystalline chemical compounds that display high-temperature superconductivity; it includes the first material ever discovered to become superconducting above the boiling point of liquid nitrogen at about 93 K.

Bismuth(III) oxide is perhaps the most industrially important compound of bismuth. It is also a common starting point for bismuth chemistry. It is found naturally as the mineral bismite (monoclinic) and sphaerobismoite, but it is usually obtained as a by-product of the smelting of copper and lead ores. Dibismuth trioxide is commonly used to produce the "Dragon's eggs" effect in fireworks, as a replacement of red lead.
Multiferroics are defined as materials that exhibit more than one of the primary ferroic properties in the same phase:

Heusler compounds are magnetic intermetallics with face-centered cubic crystal structure and a composition of XYZ (half-Heuslers) or X2YZ (full-Heuslers), where X and Y are transition metals and Z is in the p-block. The term derives from the name of German mining engineer and chemist Friedrich Heusler, who studied such a compound (Cu2MnAl) in 1903. Many of these compounds exhibit properties relevant to spintronics, such as magnetoresistance, variations of the Hall effect, ferro-, antiferro-, and ferrimagnetism, half- and semimetallicity, semiconductivity with spin filter ability, superconductivity, topological band structure and are actively studied as Thermoelectric materials. Their magnetism results from a double-exchange mechanism between neighboring magnetic ions. Manganese, which sits at the body centers of the cubic structure, was the magnetic ion in the first Heusler compound discovered. (See the Bethe–Slater curve for details of why this happens.)

Bismuth strontium calcium copper oxide (BSCCO, pronounced bisko), is a type of cuprate superconductor having the generalized chemical formula Bi2Sr2Can−1CunO2n+4+x, with n = 2 being the most commonly studied compound (though n = 1 and n = 3 have also received significant attention). Discovered as a general class in 1988, BSCCO was the first high-temperature superconductor which did not contain a rare-earth element.

In chemistry, non-stoichiometric compounds are chemical compounds, almost always solid inorganic compounds, having elemental composition whose proportions cannot be represented by a ratio of small natural numbers ; most often, in such materials, some small percentage of atoms are missing or too many atoms are packed into an otherwise perfect lattice work.

Bismuth germanium oxide or bismuth germanate is an inorganic chemical compound of bismuth, germanium and oxygen. Most commonly the term refers to the compound with chemical formula Bi4Ge3O12 (BGO), with the cubic evlitine crystal structure, used as a scintillator. (The term may also refer to a different compound with formula Bi12GeO20, an electro-optical material with sillenite structure, and Bi2Ge3O9.)
Bismuth ferrite (BiFeO3, also commonly referred to as BFO in materials science) is an inorganic chemical compound with perovskite structure and one of the most promising multiferroic materials. The room-temperature phase of BiFeO3 is classed as rhombohedral belonging to the space group R3c. It is synthesized in bulk and thin film form and both its antiferromagnetic (G type ordering) Néel temperature (approximately 653 K) and ferroelectric Curie temperature are well above room temperature (approximately 1100K). Ferroelectric polarization occurs along the pseudocubic direction () with a magnitude of 90–95 μC/cm2.
Cuprate superconductors are a family of high-temperature superconducting materials made of layers of copper oxides (CuO2) alternating with layers of other metal oxides, which act as charge reservoirs. At ambient pressure, cuprate superconductors are the highest temperature superconductors known. However, the mechanism by which superconductivity occurs is still not understood.
Superstripes is a generic name for a phase with spatial broken symmetry that favors the onset of superconducting or superfluid quantum order. This scenario emerged in the 1990s when non-homogeneous metallic heterostructures at the atomic limit with a broken spatial symmetry have been found to favor superconductivity. Before a broken spatial symmetry was expected to compete and suppress the superconducting order. The driving mechanism for the amplification of the superconductivity critical temperature in superstripes matter has been proposed to be the shape resonance in the energy gap parameters ∆n that is a type of Fano resonance for coexisting condensates.
Lanthanum manganite is an inorganic compound with the formula LaMnO3, often abbreviated as LMO. Lanthanum manganite is formed in the perovskite structure, consisting of oxygen octahedra with a central Mn atom. The cubic perovskite structure is distorted into an orthorhombic structure by a strong Jahn–Teller distortion of the oxygen octahedra.
Helium is the smallest and the lightest noble gas and one of the most unreactive elements, so it was commonly considered that helium compounds cannot exist at all, or at least under normal conditions. Helium's first ionization energy of 24.57 eV is the highest of any element. Helium has a complete shell of electrons, and in this form the atom does not readily accept any extra electrons nor join with anything to make covalent compounds. The electron affinity is 0.080 eV, which is very close to zero. The helium atom is small with the radius of the outer electron shell at 0.29 Å. Helium is a very hard atom with a Pearson hardness of 12.3 eV. It has the lowest polarizability of any kind of atom, however, very weak van der Waals forces exist between helium and other atoms. This force may exceed repulsive forces, so at extremely low temperatures helium may form van der Waals molecules. Helium has the lowest boiling point of any known substance.
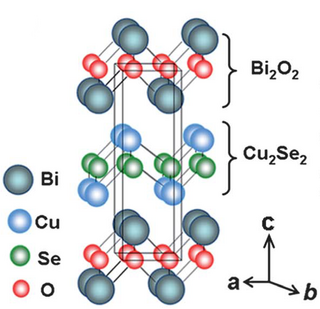
Oxyselenides are a group of chemical compounds that contain oxygen and selenium atoms. Oxyselenides can form a wide range of structures in compounds containing various transition metals, and thus can exhibit a wide range of properties. Most importantly, oxyselenides have a wide range of thermal conductivity, which can be controlled with changes in temperature in order to adjust their thermoelectric performance. Current research on oxyselenides indicates their potential for significant application in electronic materials.

Sillénite or sillenite is a mineral with the chemical formula Bi12SiO20. It is named after the Swedish chemist Lars Gunnar Sillén, who mostly studied bismuth-oxygen compounds. It is found in Australia, Europe, China, Japan, Mexico and Mozambique, typically in association with bismutite.
Nickel forms a series of mixed oxide compounds which are commonly called nickelates. A nickelate is an anion containing nickel or a salt containing a nickelate anion, or a double compound containing nickel bound to oxygen and other elements. Nickel can be in different or even mixed oxidation states, ranging from +1, +2, +3 to +4. The anions can contain a single nickel ion, or multiple to form a cluster ion. The solid mixed oxide compounds are often ceramics, but can also be metallic. They have a variety of electrical and magnetic properties. Rare-earth elements form a range of perovskite nickelates, in which the properties vary systematically as the rare-earth element changes. Fine tuning of properties is achievable with mixtures of elements, applying stress or pressure, or varying the physical form.
Oxyphosphides are chemical compounds formally containing the group PO, with one phosphorus and one oxygen atom. The phosphorus and oxygen are not bound together as in phosphates or phosphine oxides, instead they are bound separately to the cations (metals), and could be considered as a mixed phosphide-oxide compound. So a compound with OmPn requires cations to balance a negative charge of 2m+3n. The cations will have charges of +2 or +3. The trications are often rare earth elements or actinides. They are in the category of oxy-pnictide compounds.
Mohindar Singh Seehra is an Indian-American Physicist, academic and researcher. He is Eberly Distinguished Professor Emeritus at West Virginia University (WVU).
References
- ↑ Owens, Frank J.; Poole, Charles P. Jr. (2008-04-11). The Physics and Chemistry of Nanosolids. John Wiley & Sons. ISBN 9780470067406.
- ↑ "Crystalography". April 2014.
- ↑ Meek, KM; Knupp, C (2015). "Corneal structure and transparency". Prog Retin Eye Res. 49: 1–16. doi:10.1016/j.preteyeres.2015.07.001. PMC 4655862 . PMID 26145225.
- ↑ Becerro, Ana I., et al. "The transition from short-range to long-range ordering of oxygen vacancies in CaFe_x Ti_1-x O 3− x/2 perovskites." Physical Chemistry Chemical Physics 2.17 (2000): 3933-3941.
- ↑ C. McCammon, A. Becerro, F. Langenhorst, R. Angel, S. Marion, and F. Seifert, Journal of Physics: Condensed Matter 12, 2969 (2000).
- ↑ C. Hou, A. Manthiram, L. Rabenberg, and J. Goodenough, Journal of Materials Research 5, 9 (1990).
- ↑ D. Werder, C. Chen, R. Cava, and B. Batlogg, Physical Review B 37, 2317 (1988) | https://doi.org/10.1103/PhysRevB.37.2317.
- ↑ Scurti, Craig A., et al. "Electron diffraction study of the sillenites Bi12SiO20, Bi25FeO39 and Bi25InO39: Evidence of short-range ordering of oxygen-vacancies in the trivalent sillenites." AIP Advances 4.8 (2014): 087125. | https://doi.org/10.1063/1.4893341