Related Research Articles

Dentin or dentine is a calcified tissue of the body and, along with enamel, cementum, and pulp, is one of the four major components of teeth. It is usually covered by enamel on the crown and cementum on the root and surrounds the entire pulp. By volume, 45% of dentin consists of the mineral hydroxyapatite, 33% is organic material, and 22% is water. Yellow in appearance, it greatly affects the color of a tooth due to the translucency of enamel. Dentin, which is less mineralized and less brittle than enamel, is necessary for the support of enamel. Dentin rates approximately 3 on the Mohs scale of mineral hardness. There are two main characteristics which distinguish dentin from enamel: firstly, dentin forms throughout life; secondly, dentin is sensitive and can become hypersensitive to changes in temperature due to the sensory function of odontoblasts, especially when enamel recedes and dentin channels become exposed.

Endodontics is the dental specialty concerned with the study and treatment of the dental pulp.

The pulp is the connective tissue, nerves, blood vessels, and odontoblasts that comprise the innermost layer of a tooth. The pulp's activity and signalling processes regulate its behaviour.
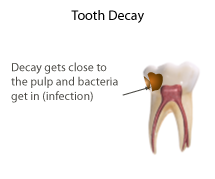
Pulpitis is inflammation of dental pulp tissue. The pulp contains the blood vessels, the nerves, and connective tissue inside a tooth and provides the tooth's blood and nutrients. Pulpitis is mainly caused by bacterial infection which itself is a secondary development of caries. It manifests itself in the form of a toothache.

Also known as a "bonderizer" bonding agents are resin materials used to make a dental composite filling material adhere to both dentin and enamel.

A temporary crown is a temporary (short-term) crown used in dentistry. Like other interim restorations, it serves until a final (definitive) restoration can be inserted. Usually the temporary crown is constructed from acrylic resins (monomethacrylate-based/polymethacrylate-based) or, chemical-cure/light cure composite (dimethacrylate-based), although alternative systems using aluminium crown forms are occasionally used. Temporary crowns function to protect the tooth, prevent teeth shifting, provide cosmetics, shape the gum tissue properly, and prevent sensitivity.

Dentinogenesis imperfecta (DI) is a genetic disorder of tooth development. It is inherited in an autosomal dominant pattern, as a result of mutations on chromosome 4q21, in the dentine sialophosphoprotein gene (DSPP). It is one of the most frequently occurring autosomal dominant features in humans. Dentinogenesis imperfecta affects an estimated 1 in 6,000-8,000 people.
Dentin hypersensitivity is dental pain which is sharp in character and of short duration, arising from exposed dentin surfaces in response to stimuli, typically thermal, evaporative, tactile, osmotic, chemical or electrical; and which cannot be ascribed to any other dental disease.

Dentin dysplasia (DD) is a rare genetic developmental disorder affecting dentine production of the teeth, commonly exhibiting an autosomal dominant inheritance that causes malformation of the root. It affects both primary and permanent dentitions in approximately 1 in every 100,000 patients. It is characterized by presence of normal enamel but atypical dentin with abnormal pulpal morphology. Witkop in 1972 classified DD into two types which are Type I (DD-1) is the radicular type, and type II (DD-2) is the coronal type. DD-1 has been further divided into 4 different subtypes (DD-1a,1b,1c,1d) based on the radiographic features.

Root canal treatment is a treatment sequence for the infected pulp of a tooth which is intended to result in the elimination of infection and the protection of the decontaminated tooth from future microbial invasion. Root canals, and their associated pulp chamber, are the physical hollows within a tooth that are naturally inhabited by nerve tissue, blood vessels and other cellular entities. Together, these items constitute the dental pulp.
In dentistry, the hydrodynamic or fluid movement theory is one of three main theories developed to explain dentine hypersensitivity, which is a sharp, transient pain arising from stimuli exposure. It states that different types of stimuli act on exposed dentine, causing increased fluid flow through the dentinal tubules. In response to this movement, mechanoreceptors on the pulp nerves trigger the acute, temporary pain of dentine hypersensitivity.
Mineral trioxide aggregate (MTA) was developed for use as a dental root repair material by Mahmoud Torabinejad. It is formulated from commercial Portland cement, combined with bismuth oxide powder for radio-opacity. MTA is used for creating apical plugs during apexification, repairing root perforations during root canal therapy, and treating internal root resorption. This can be used for root-end filling material and as pulp capping material. Originally, MTA was dark gray in color, but white versions have been on the market since 2002.
Pulpotomy is a minimally invasive procedure performed in children on a primary tooth with extensive caries but without evidence of root pathology. The minimally invasive endodontic techniques of vital pulp therapy (VPT) are based on improved understanding of the capacity of pulp (nerve) tissues to heal and regenerate plus the availability of advanced endodontic materials. During the caries removal, this results in a carious or mechanical pulp exposure (bleeding) from the cavity. During pulpotomy, the inflamed/diseased pulp tissue is removed from the coronal pulp chamber of the tooth leaving healthy pulp tissue which is dressed with a long-term clinically-successful medicament that maintains the survival of the pulp and promotes repair. There are various types of medicament placed above the vital pulp such as Buckley's Solution of formocresol, ferric sulfate, calcium hydroxide or mineral trioxide aggregate (MTA). MTA is a more recent material used for pulpotomies with a high rate of success, better than formocresol or ferric sulfate. It is also recommended to be the preferred pulpotomy agent in the future. After the coronal pulp chamber is filled, the tooth is restored with a filling material that seals the tooth from microleakage, such as a stainless steel crown which is the most effective long-term restoration. However, if there is sufficient remaining supporting tooth structure, other filling materials such as amalgam or composite resin can provide a functional alternative when the primary tooth has a life span of two years or less. The medium- to long-term treatment outcomes of pulpotomy in symptomatic permanent teeth with caries, especially in young people, indicate that pulpotomy can be a potential alternative to root canal therapy (RCT).

Resorption of the root of the tooth, or root resorption, is the progressive loss of dentin and cementum by the action of odontoclasts. Root resorption is a normal physiological process that occurs in the exfoliation of the primary dentition. However, pathological root resorption occurs in the permanent or secondary dentition and sometimes in the primary dentition.
Endodontic files and reamers are surgical instruments used by dentists when performing root canal treatment. These tools are used to clean and shape the root canal, with the concept being to perform complete chemomechanical debridement of the root canal to the length of the apical foramen. Preparing the canal in this way facilitates the chemical disinfection to a satisfactory length but also provides a shape conducive to obturation.

Pulp stones are nodular, calcified masses appearing in either or both the coronal and root portion of the pulp organ in teeth. Pulp stones are not painful unless they impinge on nerves.

Regenerative endodontic procedures is defined as biologically based procedures designed to replace damaged structures such as dentin, root structures, and cells of the pulp-dentin complex. This new treatment modality aims to promote normal function of the pulp. It has become an alternative to heal apical periodontitis. Regenerative endodontics is the extension of root canal therapy. Conventional root canal therapy cleans and fills the pulp chamber with biologically inert material after destruction of the pulp due to dental caries, congenital deformity or trauma. Regenerative endodontics instead seeks to replace live tissue in the pulp chamber. The ultimate goal of regenerative endodontic procedures is to regenerate the tissues and the normal function of the dentin-pulp complex.

Pulp capping is a technique used in dental restorations to prevent the dental pulp from necrosis, after being exposed, or nearly exposed during a cavity preparation, from a traumatic injury, or by a deep cavity that reaches the center of the tooth causing the pulp to die. When dental caries is removed from a tooth, all or most of the infected and softened enamel and dentin are removed. This can lead to the pulp of the tooth either being exposed or nearly exposed which causes pulpitis (inflammation). Pulpitis, in turn, can become irreversible, leading to pain and pulp necrosis, and necessitating either root canal treatment or extraction. The ultimate goal of pulp capping or stepwise caries removal is to protect a healthy dental pulp and avoid the need for root canal therapy.
Biofilling, also known as orthograde canal grafting technique or 4D sealing, is an endodontic root canal obturation technique with a Bioceramic material after root canal preparation and enlargement procedure.
Apexification is a method of dental treatment to induce a calcific barrier in a root with incomplete formation or open apex of a tooth with necrotic pulp. Pulpal involvement usually occurs as a consequence of trauma or caries involvement of young or immature permanent teeth. As a sequelae of untreated pulp involvement, loss of pulp vitality or necrotic pulp took place for the involved teeth.
References
- 1 2 Brännström, Martin; Johnson, Gunilla (April 1974). "Effects of various conditioners and cleaning agents on prepared dentin surfaces: A scanning electron microscopic investigation". The Journal of Prosthetic Dentistry. 31 (4): 422–430. doi:10.1016/0022-3913(74)90152-8. ISSN 0022-3913. PMID 4592745.
- ↑ Watts, D.; Silikas, N. (2005), "In Situ Photo-Polymerisation and Polymerisation-Shrinkage Phenomena", Dental Hard Tissues and Bonding, Springer Berlin Heidelberg, pp. 123–154, doi:10.1007/3-540-28559-8_6, ISBN 978-3-540-23408-1
- ↑ McComb, Dorothy; Smith, Dennis C. (July 1975). "A preliminary scanning electron microscopic study of root canals after endodontic procedures". Journal of Endodontics. 1 (7): 238–242. doi:10.1016/s0099-2399(75)80226-3. ISSN 0099-2399. PMID 1061799.
- 1 2 3 Mader, Carson L.; Baumgartner, J. Craig; Peters, Donald D. (October 1984). "Scanning electron microscopic investigation of the smeared layer on root canal walls". Journal of Endodontics. 10 (10): 477–483. doi:10.1016/s0099-2399(84)80204-6. ISSN 0099-2399. PMID 6593410.
- ↑ Şen, B. H.; Wesselink, P. R.; Türkün, M. (1995). "The smear layer: a phenomenon in root canal therapy". International Endodontic Journal. 28 (3): 141–148. doi:10.1111/j.1365-2591.1995.tb00289.x. ISSN 1365-2591. PMID 8626198.
- 1 2 Zargar, Nazanin; Dianat, Omid; Asnaashari, Mohammad; Ganjali, Mojtaba; Zadsirjan, Saeede (2015). "The Effect of Smear Layer on Antimicrobial Efficacy of Three Root Canal Irrigants". Iranian Endodontic Journal. 10 (3): 179–183. doi:10.7508/iej.2015.03.007. ISSN 1735-7497. PMC 4509126 . PMID 26213540.
- 1 2 3 4 Bindra. "A scanning electron microscope evaluation of smear layer removal from root canals prepared by manual or rotary instrumentation using three different irrigating systems: An in vitro study". www.endodontologyonweb.org. Retrieved 2020-03-03.
- ↑ Olgart L, Brännström M, Johnson G. Invasion of bacteria into dentinal tubules: Experiments in vivo and Invitro. Acta Odontologica Scandinavica 1974; 32:61-70.
- ↑ Pashley DH, Michelich V, Kehl T. Dentin permeability: effects of smear layer removal. J Prosthet Dent 1981; 46:531-7.
- ↑ Gettleman BH, Messer HH, ElDeeb ME. Adhesion of sealer cements to dentin with and without the smear layer. J Endod 1991; 17:15-20.
- 1 2 3 McCabe, J (2008). Applied Dental Materials 9th Edition. pp. Chapter 30.
- ↑ Goldberg, Fernando; Abramovich, Abraham (1977-03-01). "Analysis of the effect of EDTAC on the dentinal walls of the root canal". Journal of Endodontics. 3 (3): 101–105. doi:10.1016/S0099-2399(77)80203-3. ISSN 0099-2399. PMID 404380.
- ↑ Pashley, D. H. (1996). "Dynamics of the pulpo-dentin complex". Critical Reviews in Oral Biology and Medicine. 7 (2): 104–133. doi:10.1177/10454411960070020101. ISSN 1045-4411. PMID 8875027.
- ↑ Love, R. M.; Jenkinson, H. F. (2002). "Invasion of dentinal tubules by oral bacteria". Critical Reviews in Oral Biology and Medicine. 13 (2): 171–183. doi: 10.1177/154411130201300207 . ISSN 1045-4411. PMID 12097359.
- ↑ McComb, D.; Smith, D. C. (July 1975). "A preliminary scanning electron microscopic study of root canals after endodontic procedures". Journal of Endodontics. 1 (7): 238–242. doi:10.1016/S0099-2399(75)80226-3. ISSN 0099-2399. PMID 1061799.
- ↑ Saunders, W. P.; Saunders, E. M. (September 1992). "The effect of smear layer upon the coronal leakage of gutta-percha fillings and a glass ionomer sealer". International Endodontic Journal. 25 (5): 245–249. doi:10.1111/j.1365-2591.1992.tb01157.x. ISSN 0143-2885. PMID 1291521.
- ↑ Saunders, Saunders, WP, WE (1992). A Concise Guide to Endodontic Procedures. pp. Chapter 7.
- ↑ Haapasalo, M.; Shen, Y.; Wang, Z.; Gao, Y. (March 2014). "Irrigation in endodontics". British Dental Journal. 216 (6): 299–303. doi:10.1038/sj.bdj.2014.204. ISSN 1476-5373. PMID 24651335. S2CID 82082.
- 1 2 3 Chandler, Nicholas; Chellappa, Deepak (2019). "Lubrication during root canal treatment". Australian Endodontic Journal. 45 (1): 106–110. doi: 10.1111/aej.12282 . ISSN 1747-4477. PMID 30105836.
- ↑ Haapasalo, Markus; Qian, Wei; Shen, Ya (2012). "Irrigation: beyond the smear layer". Endodontic Topics. 27 (1): 35–53. doi:10.1111/etp.12030. ISSN 1601-1546.
- 1 2 3 4 Gokce, Kagan; Aykor, Arzu; Ersoy, Mustafa; Ozel, Emre; Soyman, Mubin (October 2008). "Effect of phosphoric acid etching and self-etching primer application methods on dentinal shear bond strength". The Journal of Adhesive Dentistry. 10 (5): 345–349. ISSN 1461-5185. PMID 19058679.
- ↑ Clark-Holke D, Drake D, Walton R, Rivera E, Guthmiller JM. Bacterial penetration through canals of endodontically treated teeth in the presence or absence of the smear layer. J Dent 2003; 31:275-281.
- ↑ Kokkas AB, Boutsioukis ACh, Vassiliadis LP, Stavrianos CK. The influence of the smear layer on dentinal tubule penetration depth by three different root canal sealers. J Endod 2004; 30:100-102.
- ↑ Çobankara FK, Adanir N, Sema Belli. Evaluation of the influence of smear layer on the apical and coronal sealing ability of two sealers. J Endod 2004; 30:406-409.
- ↑ Bertacci A, Baroni C, Breschi L, Venturi M, Prati C. The influence of smear layer in lateral channels filling. Clin Oral Investig 2007; 11:353-359.
- ↑ Yildirim T, Oruçoğlu H, Cobankara FK. Long-term evaluation of the influence of smear layer on the apical sealing ability of MTA. J Endod 2008; 34:1537-1540.
- ↑ Saleh IM, Ruyter IE, Haapasalo M, Ørstavik D. Bacterial penetration along different root canal filling materials in the presence or absence of smear layer. Int Endod J. 2008; 41:32-40.
- ↑ Fachin EV, Scarparo RK, Massoni LI. Influence of smear layer removal on the obturation of root canal ramifications. J Appl Oral Sci 2009; 17:240-243.