
Pseudoephedrine (PSE) is a sympathomimetic drug of the phenethylamine and amphetamine chemical classes. It may be used as a nasal/sinus decongestant, as a stimulant, or as a wakefulness-promoting agent in higher doses.

Over-the-counter (OTC) drugs are medicines sold directly to a consumer without a requirement for a prescription from a healthcare professional, as opposed to prescription drugs, which may be supplied only to consumers possessing a valid prescription. In many countries, OTC drugs are selected by a regulatory agency to ensure that they contain ingredients that are safe and effective when used without a physician's care. OTC drugs are usually regulated according to their active pharmaceutical ingredient (API) rather than final products. By regulating APIs instead of specific drug formulations, governments allow manufacturers the freedom to formulate ingredients, or combinations of ingredients, into proprietary mixtures.

Temazepam is a medication of the benzodiazepine class which is generally used to treat severe or debilitating insomnia. It is taken by mouth. Temazepam is rapidly absorbed, and significant hypnotic effects begin in less than 30 minutes and can last for up to eight hours. Many studies, some going as far back as the early 1980s out of Australia and the United Kingdom, both of which have had serious temazepam abuse epidemics and related mortality, have all mostly corroborated each other and proven that the potential for abuse and physical dependence is very high, even in comparison to many other benzodiazepines. As a result, prescriptions for hypnotics such as temazepam have seen a dramatic decrease since 2010, while anxiolytics such as alprazolam (Xanax), clonazepam, and lorazepam (Ativan) have increased or remained stable. Temazepam and similar hypnotics, such as triazolam (Halcion) are generally reserved for severe and debilitating insomnia. They have largely been replaced by z-drugs and atypical antidepressants as first line treatment for insomnia.

Alprazolam, sold under the brand name Xanax, among others, is a fast-acting, potent tranquilizer of medium duration in the triazolobenzodiazepine (TBZD) class, which are benzodiazepines (BZDs) fused with a triazole ring. It is most commonly used in management of anxiety disorders, specifically panic disorder or generalized anxiety disorder (GAD). Other uses include the treatment of chemotherapy-induced nausea, together with other treatments. GAD improvement occurs generally within a week. Alprazolam is generally taken by mouth.

Naproxen, sold under the brand name Aleve among others, is a nonsteroidal anti-inflammatory drug (NSAID) used to treat pain, menstrual cramps, inflammatory diseases such as rheumatoid arthritis, gout and fever. It is taken orally. It is available in immediate and delayed release formulations. Onset of effects is within an hour and lasts for up to twelve hours.

Benadryl is a brand of various antihistamine medications used to stop allergies, whose content varies in different countries, but which includes some combination of diphenhydramine, acrivastine, and/or cetirizine.

Clonazepam, sold under the brand names Klonopin and Rivotril, is a medication used to prevent and treat seizures, panic disorder, anxiety disorders, and the movement disorder known as akathisia. It is a tranquilizer of the benzodiazepine class. It possesses anxiolytic, anticonvulsant, sedative, hypnotic, and skeletal muscle relaxant properties. It is typically taken by mouth. Effects begin within one hour and last between six and twelve hours.
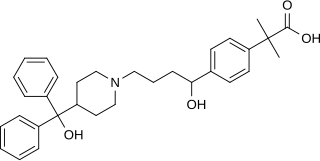
Fexofenadine, sold under the brand name Allegra among others, is an antihistamine pharmaceutical drug used in the treatment of allergy symptoms, such as hay fever and urticaria.

Dihydrocodeine is a semi-synthetic opioid analgesic prescribed for pain or severe dyspnea, or as an antitussive, either alone or compounded with paracetamol (acetaminophen) or aspirin. It was developed in Germany in 1908 and first marketed in 1911.

Excedrin is an over-the-counter headache pain reliever, typically in the form of tablets or caplets. It contains paracetamol (acetaminophen), aspirin, and caffeine. It was manufactured by Bristol-Myers Squibb until it was purchased by Novartis in July, 2005 along with other products from BMS's over-the-counter business. As of March 2015, GSK holds majority ownership of Excedrin through a joint venture transaction with Novartis. On 18 July 2022, GSK spun off its consumer healthcare business to Haleon.

Dextropropoxyphene is an analgesic in the opioid category, patented in 1955 and manufactured by Eli Lilly and Company. It is an optical isomer of levopropoxyphene. It is intended to treat mild pain and also has antitussive and local anaesthetic effects. The drug has been taken off the market in Europe and the US due to concerns of fatal overdoses and heart arrhythmias. It is still available in Australia, albeit with restrictions after an application by its manufacturer to review its proposed banning. Its onset of analgesia is said to be 20–30 minutes and peak effects are seen about 1.5–2.0 hours after oral administration.
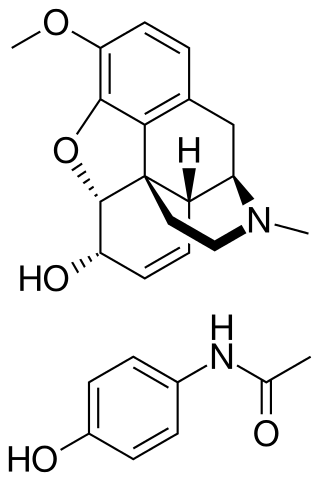
Codeine/paracetamol, also known as codeine/acetaminophen and co-codamol, is a compound analgesic consisting of a combination of codeine phosphate and paracetamol (acetaminophen). Codeine/paracetamol is used for the relief of mild to moderate pain when paracetamol or non-steroidal anti-inflammatory drug (NSAIDs) such as ibuprofen, aspirin or naproxen alone do not sufficiently relieve symptoms.

Co-codaprin (BAN) is a compound analgesic, a combination of codeine phosphate with aspirin.

Ranitidine, sold under the brand name Zantac among others, is a medication used to decrease stomach acid production. It is commonly used in treatment of peptic ulcer disease, gastroesophageal reflux disease, and Zollinger–Ellison syndrome. It can be given by mouth, injection into a muscle, or injection into a vein. In September 2019, the probable carcinogen N-nitrosodimethylamine (NDMA) was discovered in ranitidine products from a number of manufacturers, resulting in recalls. In April 2020, ranitidine was withdrawn from the United States market and suspended in the European Union and Australia due to these concerns. In 2022, these concerns were confirmed in a nationwide population study "ranitidine increased the risk of liver", lung, gastric and pancreatic cancer by 22%,17%, 26% and 35%, respectively. It increased overall cancer risk 10%, p < 0.001.

Doxylamine, sold under the brand name Unisom among others, is an antihistamine medication which is used in the treatment of insomnia and allergies. In combination with pyridoxine (vitamin B6), it is also used to treat morning sickness in pregnant women. Doxylamine is available over-the-counter, and is used in nighttime cold medicines, such as NyQuil, as well as in pain medications containing acetaminophen and codeine, to help with sleep. The medication is taken by mouth.
Actifed is a registered trademark for a combination antihistamine and nasal decongestant medication used for cold and allergy symptoms. Actifed was developed in 1958 by Burroughs Wellcome & Company, later Haleon.

Corvalol is a tranquilizer based on the herb valerian root, as well peppermint oil Mentha piperita and hop extract Humulus lupulus and the barbiturate phenobarbital, popular in Eastern Europe and the former Soviet Union as a heart medication. It is available as a transparent liquid with a characteristic strong aroma, and as white bi-concave scored tablets. While not available for sale in the Western countries, Corvalol is sometimes brought over from Eastern Europe for self-administration to other countries of residence. Corvalol contains documented amounts of psychoactive chemicals, and may interact with other prescription medications that a person is taking.
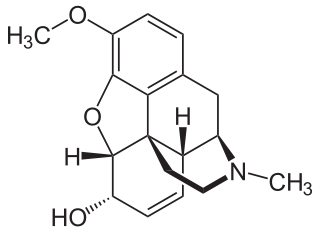
Codeine is an opiate and prodrug of morphine mainly used to treat pain, coughing, and diarrhea. It is also commonly used as a recreational drug. It is found naturally in the sap of the opium poppy, Papaver somniferum. It is typically used to treat mild to moderate degrees of pain. Greater benefit may occur when combined with paracetamol (acetaminophen) or a nonsteroidal anti-inflammatory drug (NSAID) such as aspirin or ibuprofen. Evidence does not support its use for acute cough suppression in children or adults. In Europe, it is not recommended as a cough medicine in those under 12 years of age. It is generally taken by mouth. It typically starts working after half an hour, with maximum effect at two hours. Its effects last for about four to six hours. Codeine exhibits abuse potential similar to other opioid medications.

Naltrexone/bupropion, sold under the brand name Contrave among others, is a fixed-dose combination medication for the management of chronic obesity in adults in combination with a reduced-calorie diet and increased physical activity. It contains naltrexone, an opioid antagonist, and bupropion, an aminoketone antidepressant. It is taken by mouth. Both medications have individually shown some evidence of effectiveness in weight loss, and the combination has been shown to have some synergistic effects on weight.

The Poison Prevention Packaging Act of 1970 (PPPA); was signed into law by U.S. President Richard Nixon on December 30, 1970. It was enacted by the 91st United States Congress. This law required the use of child-resistant packaging for prescription drugs, over-the-counter (OTC) drugs, household chemicals, and other hazardous materials that could be considered dangerous for children.


















