Antidepressants are a class of medications used to treat major depressive disorder, anxiety disorders, chronic pain, and addiction.
An anxiolytic is a medication or other intervention that reduces anxiety. This effect is in contrast to anxiogenic agents which increase anxiety. Anxiolytic medications are used for the treatment of anxiety disorders and their related psychological and physical symptoms.
A psychiatric or psychotropic medication is a psychoactive drug taken to exert an effect on the chemical makeup of the brain and nervous system. Thus, these medications are used to treat mental illnesses. These medications are typically made of synthetic chemical compounds and are usually prescribed in psychiatric settings, potentially involuntarily during commitment. Since the mid-20th century, such medications have been leading treatments for a broad range of mental disorders and have decreased the need for long-term hospitalization, thereby lowering the cost of mental health care. The recidivism or rehospitalization of the mentally ill is at a high rate in many countries, and the reasons for the relapses are under research.

Paroxetine, sold under the brand name Paxil among others, is an antidepressant of the selective serotonin reuptake inhibitor (SSRI) class. It is used to treat major depressive disorder, obsessive–compulsive disorder (OCD), panic disorder, social anxiety disorder, post-traumatic stress disorder (PTSD), generalized anxiety disorder, and premenstrual dysphoric disorder. It has also been used in the treatment of premature ejaculation and hot flashes due to menopause. It is taken orally.

Sertraline, sold under the brand name Zoloft among others, is an antidepressant of the selective serotonin reuptake inhibitor (SSRI) class. The effectiveness of sertraline for depression is similar to that of other antidepressants such as Fluoxetine or Paroxetine. Sertraline is better tolerated than the older tricyclic antidepressants. Sertraline is effective for panic disorder, social anxiety disorder, generalized anxiety disorder, and obsessive–compulsive disorder (OCD). Although approved for post-traumatic stress disorder (PTSD), findings have shown sertraline leads to only modest improvements in symptoms. Sertraline also alleviates the symptoms of premenstrual dysphoric disorder and can be used in sub-therapeutic doses or intermittently for its treatment.

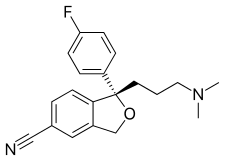
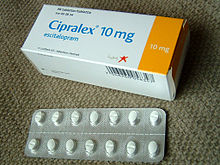
Citalopram, sold under the brand name Celexa among others, is an antidepressant of the selective serotonin reuptake inhibitor (SSRI) class. It is used to treat major depressive disorder, obsessive compulsive disorder, panic disorder, and social phobia. The antidepressant effects may take one to four weeks to occur. It is typically taken orally. In some European countries, it is sometimes given intravenously to initiate treatment, before switching to the oral route of administration for continuation of treatment. It has also been used intravenously in other parts of the world in some other circumstances.

Fluvoxamine, sold under the brand name Luvox among others, is an antidepressant of the selective serotonin reuptake inhibitor (SSRI) class. It is primarily used to treat major depressive disorder and, perhaps more-especially, obsessive–compulsive disorder (OCD), but is also used to treat anxiety disorders such as panic disorder, social anxiety disorder, and post-traumatic stress disorder.
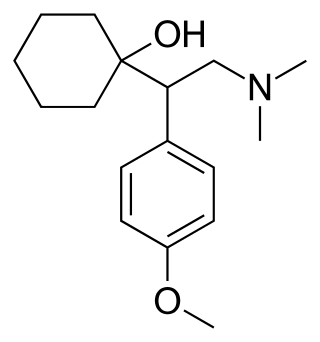
Venlafaxine, sold under the brand name Effexor among others, is an antidepressant medication of the serotonin–norepinephrine reuptake inhibitor (SNRI) class. It is used to treat major depressive disorder, generalized anxiety disorder, panic disorder, and social anxiety disorder. Studies have shown that venlafaxine improves post-traumatic stress disorder (PTSD). It may also be used for chronic neuropathic pain. It is taken orally. It is also available as the salt venlafaxine besylate in an extended-release formulation.

Duloxetine, sold under the brand name Cymbalta among others, is a medication used to treat major depressive disorder, generalized anxiety disorder, obsessive-compulsive disorder, fibromyalgia, neuropathic pain and central sensitization. It is taken by mouth.

Serotonin–norepinephrine reuptake inhibitors (SNRIs) are a class of antidepressant medications used to treat major depressive disorder (MDD), anxiety disorders, social phobia, chronic neuropathic pain, fibromyalgia syndrome (FMS), and menopausal symptoms. Off-label uses include treatments for attention-deficit hyperactivity disorder (ADHD), and obsessive–compulsive disorder (OCD). SNRIs are monoamine reuptake inhibitors; specifically, they inhibit the reuptake of serotonin and norepinephrine. These neurotransmitters are thought to play an important role in mood regulation. SNRIs can be contrasted with the selective serotonin reuptake inhibitors (SSRIs) and norepinephrine reuptake inhibitors (NRIs), which act upon single neurotransmitters.

Clomipramine, sold under the brand name Anafranil among others, is a tricyclic antidepressant (TCA). It is used in the treatment of various conditions, most notably obsessive–compulsive disorder but also many other disorders, including hyperacusis, panic disorder, major depressive disorder, trichotillomania, body dysmorphic disorder and chronic pain. It has also been notably used to treat premature ejaculation and the cataplexy associated with narcolepsy.
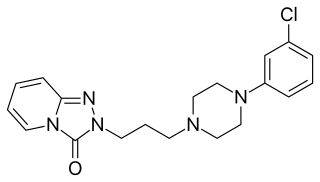
Trazodone, sold under many brand names, is an antidepressant medication, used to treat major depressive disorder, anxiety disorders, and insomnia. It is a phenylpiperazine compound of the serotonin antagonist and reuptake inhibitor (SARI) class. The medication is taken orally.
Fluoxetine, sold under the brand name Prozac, among others, is an antidepressant of the selective serotonin reuptake inhibitor (SSRI) class. It is used for the treatment of major depressive disorder, obsessive–compulsive disorder (OCD), anxiety, bulimia nervosa, panic disorder, and premenstrual dysphoric disorder. It is also approved for treatment of major depressive disorder in adolescents and children 8 years of age and over. It has also been used to treat premature ejaculation. Fluoxetine is taken by mouth.
The number of new psychiatric drugs, and especially antidepressants on the market in Japan, is significantly less than Western countries.

Vortioxetine, sold under the brand name Trintellix and Brintellix among others, is an antidepressant of the serotonin modulator and stimulator (SMS) class. Its effectiveness is viewed as similar to that of other antidepressants. It is taken orally.

Selective serotonin reuptake inhibitors (SSRIs) are a class of drugs that are typically used as antidepressants in the treatment of major depressive disorder, anxiety disorders, and other psychological conditions.

Brexpiprazole, sold under the brand name Rexulti among others, is an atypical antipsychotic medication used for the treatment of major depressive disorder, schizophrenia, and agitation associated with dementia due to Alzheimer's disease.
QT prolongation is a measure of delayed ventricular repolarisation, which means the heart muscle takes longer than normal to recharge between beats. It is an electrical disturbance which can be seen on an electrocardiogram (ECG). Excessive QT prolongation can trigger tachycardias such as torsades de pointes (TdP). QT prolongation is an established side effect of antiarrhythmics, but can also be caused by a wide range of non-cardiac medicines, including antibiotics, antidepressants, antihistamines, opioids, and complementary medicines. On an ECG, the QT interval represents the summation of action potentials in cardiac muscle cells, which can be caused by an increase in inward current through sodium or calcium channels, or a decrease in outward current through potassium channels. By binding to and inhibiting the “rapid” delayed rectifier potassium current protein, certain drugs are able to decrease the outward flow of potassium ions and extend the length of phase 3 myocardial repolarization, resulting in QT prolongation.
Selective serotonin reuptake inhibitors, or serotonin-specific re-uptake inhibitor (SSRIs), are a class of chemical compounds that have application as antidepressants and in the treatment of depression and other psychiatric disorders. SSRIs are therapeutically useful in the treatment of panic disorder (PD), posttraumatic stress disorder (PTSD), social anxiety disorder, obsessive-compulsive disorder (OCD), premenstrual dysphoric disorder (PMDD), and anorexia. There is also clinical evidence of the value of SSRIs in the treatment of the symptoms of schizophrenia and their ability to prevent cardiovascular diseases.
Selective norepinephrine reuptake inhibitors (sNRIs) are a class of drugs that have been marketed as antidepressants and are used for various mental disorders, mainly depression and attention deficit hyperactivity disorder (ADHD). The norepinephrine transporter (NET) serves as the fundamental mechanism for the inactivation of noradrenergic signaling because of the NET termination in the reuptake of norepinephrine (NE). The selectivity and mechanism of action for the NRI drugs remain mostly unresolved and, to date, only a limited number of NRI-selective inhibitors are available. The first commercially available selective NRI was the drug reboxetine (Edronax), developed as a first-line therapy for major depressive disorder. Atomoxetine (Strattera) is another potent and selective NRI which is also effective and well tolerated for the treatment of ADHD in adults; it may also be a new treatment option for adults with ADHD, particularly for those patients at risk of substance abuse.