
Antidepressants are a class of medications used to treat major depressive disorder, anxiety disorders, chronic pain, and addiction.

Sertraline, sold under the brand name Zoloft among others, is an antidepressant of the selective serotonin reuptake inhibitor (SSRI) class. The efficacy of sertraline for depression is similar to that of other antidepressants, and the differences are mostly confined to side effects. Sertraline is better tolerated than the older tricyclic antidepressants. Sertraline is effective for panic disorder, social anxiety disorder, generalized anxiety disorder (GAD), and obsessive–compulsive disorder (OCD). Although approved for post-traumatic stress disorder (PTSD), sertraline leads to only modest improvement in this condition. Sertraline also alleviates the symptoms of premenstrual dysphoric disorder (PMDD) and can be used in sub-therapeutic doses or intermittently for its treatment.

Venlafaxine, sold under the brand name Effexor among others, is an antidepressant medication of the serotonin-norepinephrine reuptake inhibitor (SNRI) class. It is used to treat major depressive disorder, generalized anxiety disorder, panic disorder, and social anxiety disorder. Studies have shown that Venlafaxine improves quality of life. It may also be used for chronic pain. It is taken by mouth. It is also available as the salt venlafaxine besylate in an extended-release formulation.

Duloxetine, sold under the brand name Cymbalta among others, is a medication used to treat major depressive disorder, generalized anxiety disorder, fibromyalgia, neuropathic pain and central sensitization. It is taken by mouth.

Serotonin–norepinephrine reuptake inhibitors (SNRIs) are a class of antidepressant medications used to treat major depressive disorder (MDD), anxiety disorders, social phobia, chronic neuropathic pain, fibromyalgia syndrome (FMS), and menopausal symptoms. Off-label uses include treatments for attention-deficit hyperactivity disorder (ADHD), obsessive–compulsive disorder (OCD), and migraine prevention. SNRIs are monoamine reuptake inhibitors; specifically, they inhibit the reuptake of serotonin and norepinephrine. These neurotransmitters are thought to play an important role in mood regulation. SNRIs can be contrasted with the selective serotonin reuptake inhibitors (SSRIs) and norepinephrine reuptake inhibitors (NRIs), which act upon single neurotransmitters.
A dopamine reuptake inhibitor (DRI) is a class of drug which acts as a reuptake inhibitor of the monoamine neurotransmitter dopamine by blocking the action of the dopamine transporter (DAT). Reuptake inhibition is achieved when extracellular dopamine not absorbed by the postsynaptic neuron is blocked from re-entering the presynaptic neuron. This results in increased extracellular concentrations of dopamine and increase in dopaminergic neurotransmission.

A norepinephrine reuptake inhibitor or noradrenaline reuptake inhibitor or adrenergic reuptake inhibitor (ARI), is a type of drug that acts as a reuptake inhibitor for the neurotransmitters norepinephrine (noradrenaline) and epinephrine (adrenaline) by blocking the action of the norepinephrine transporter (NET). This in turn leads to increased extracellular concentrations of norepinephrine and epinephrine and therefore can increase adrenergic neurotransmission.
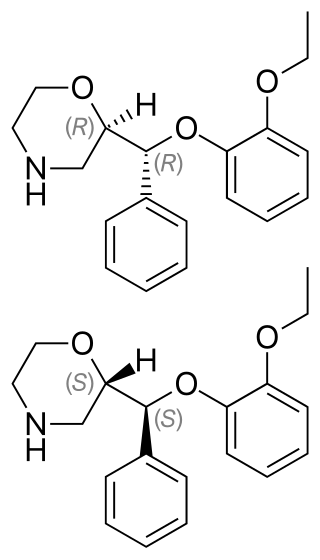
Reboxetine, sold under the brand name Edronax among others, is a drug of the norepinephrine reuptake inhibitor (NRI) class, marketed as an antidepressant by Pfizer for use in the treatment of major depression, although it has also been used off-label for panic disorder and attention deficit hyperactivity disorder (ADHD). It is approved for use in many countries worldwide, but has not been approved for use in the United States. Although its effectiveness as an antidepressant has been challenged in multiple published reports, its popularity has continued to increase.

The norepinephrine transporter (NET), also known as noradrenaline transporter (NAT), is a protein that in humans is encoded by the solute carrier family 6 member 2 (SLC6A2) gene.
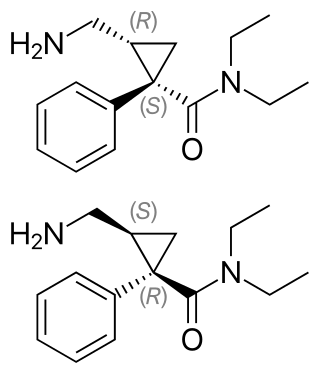
Milnacipran is a serotonin–norepinephrine reuptake inhibitor (SNRI) used in the clinical treatment of fibromyalgia. It is not approved for the clinical treatment of major depressive disorder in the US, but it is in other countries.
A serotonin–norepinephrine–dopamine reuptake inhibitor (SNDRI), also known as a triple reuptake inhibitor (TRI), is a type of drug that acts as a combined reuptake inhibitor of the monoamine neurotransmitters serotonin, norepinephrine, and dopamine. It does this by concomitantly inhibiting the serotonin transporter (SERT), norepinephrine transporter (NET), and dopamine transporter (DAT), respectively. Inhibition of the reuptake of these neurotransmitters increases their extracellular concentrations and, therefore, results in an increase in serotonergic, adrenergic, and dopaminergic neurotransmission. The naturally-occurring and potent SNDRI cocaine is widely used recreationally and often illegally for the euphoric effects it produces.

A serotonin reuptake inhibitor (SRI) is a type of drug which acts as a reuptake inhibitor of the neurotransmitter serotonin by blocking the action of the serotonin transporter (SERT). This in turn leads to increased extracellular concentrations of serotonin and, therefore, an increase in serotonergic neurotransmission. It is a type of monoamine reuptake inhibitor (MRI); other types of MRIs include dopamine reuptake inhibitors and norepinephrine reuptake inhibitors.

Nisoxetine, originally synthesized in the Lilly research laboratories during the early 1970s, is a potent and selective inhibitor for the reuptake of norepinephrine (noradrenaline) into synapses. It currently has no clinical applications in humans, although it was originally researched as an antidepressant. Nisoxetine is now widely used in scientific research as a standard selective norepinephrine reuptake inhibitor. It has been used to research obesity and energy balance, and exerts some local analgesia effects.

Talopram, also known as phthalapromine, is a selective norepinephrine reuptake inhibitor (NRI) which was researched for the management of depression in the 1960s and 1970s but was never commercialized. Along with talsupram, talopram is structurally related to the selective serotonin reuptake inhibitor (SSRI) citalopram, as well as to melitracen:
In 1971, the company hired Klaus Bøgesø as a medicinal chemist. Over the years Bøgesø turned out to have a Midas touch at the game of drug hunting, creating more molecules that made it to the market than almost any other medicinal chemist in the field. The challenge facing him in 1971 following his recruitment was to produce a selective norepinephrine reuptake inhibitor. Like other companies at the time, Lundbeck had little interest in an SSRI. Bøgesø began from an accident in the laboratory. Trying to create a derivative of their norepinephrine reuptake inhibiting antidepressant melitracen, Lundbeck chemists accidentally produced a new chemical — a phenylphthalene. Against all the odds, just like melitracen, this was also a selective norepinephrine reuptake inhibitor. Two potential antidepressants came out of this — talopram and tasulopram, which were pressed into clinical trials. Both however turned out to be energizing, and in a number of cases there were suicide attempts. The fact that there were suicide attempts appeared to confirm another proposal of Paul Kielholz, that activating antidepressants might lead to suicide. Lundbeck's experience suggested that norepinephrine reuptake inhibitors were likely to lead to just this problem. Lundbeck retreated, scared. If norepinephrine reuptake inhibitors were likely to trigger suicide, the greatest hazard of an antidepressant, then Kielholz's view suggested that an SSRI would be less likely to lead to suicide. Bøgesø's job was to see whether the new series of drugs could be converted into a series of SSRIs. Following a lead from Carlsson on how to do this, he converted talopram into citalopram, the most selective serotonin reuptake inhibitor to come to the market.

Selective serotonin reuptake inhibitors (SSRIs) are a class of drugs that are typically used as antidepressants in the treatment of major depressive disorder, anxiety disorders, and other psychological conditions.

Lortalamine (LM-1404) is an antidepressant which was synthesized in the early 1980s. It acts as a potent and highly selective norepinephrine reuptake inhibitor. Lortalamine was under development for clinical use but was shelved, likely due to the finding that it produced ocular toxicity in animals. It has been used to label the norepinephrine transporter in positron emission tomography studies.

A serotonin–dopamine reuptake inhibitor (SDRI) is a type of drug which acts as a reuptake inhibitor of the monoamine neurotransmitters serotonin and dopamine by blocking the actions of the serotonin transporter (SERT) and dopamine transporter (DAT), respectively. This in turn leads to increased extracellular concentrations of serotonin and dopamine, and, therefore, an increase in serotonergic and dopaminergic neurotransmission.
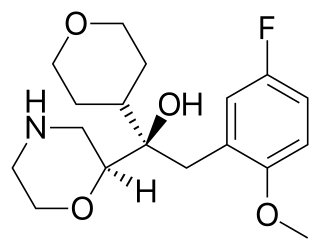
Edivoxetine is a drug which acts as a selective norepinephrine reuptake inhibitor and was under development by Eli Lilly for attention-deficit disorder (ADD) and as an antidepressant treatment. It was in phase III clinical trials in 2012 for major depressive disorder, but failed to get approval.
A monoamine reuptake inhibitor (MRI) is a drug that acts as a reuptake inhibitor of one or more of the three major monoamine neurotransmitters serotonin, norepinephrine, and dopamine by blocking the action of one or more of the respective monoamine transporters (MATs), which include the serotonin transporter (SERT), norepinephrine transporter (NET), and dopamine transporter (DAT). This in turn results in an increase in the synaptic concentrations of one or more of these neurotransmitters and therefore an increase in monoaminergic neurotransmission.
Selective norepinephrine reuptake inhibitors (sNRIs) are a class of drugs that have been marketed as antidepressants and are used for various mental disorders, mainly depression and attention-deficit hyperactivity disorder (ADHD). The norepinephrine transporter (NET) serves as the fundamental mechanism for the inactivation of noradrenergic signaling because of the NET termination in the reuptake of norepinephrine (NE). The selectivity and mechanism of action for the NRI drugs remain mostly unresolved and, to date, only a limited number of NRI-selective inhibitors are available. The first commercially available selective NRI was the drug reboxetine (Edronax), developed as a first-line therapy for major depressive disorder. Atomoxetine (Strattera) is another potent and selective NRI which is also effective and well tolerated for the treatment of ADHD in adults; it may also be a new treatment option for adults with ADHD, particularly for those patients at risk of substance abuse.













