In organic chemistry, an amide, also known as an organic amide or a carboxamide, is a compound with the general formula R−C(=O)−NR′R″, where R, R', and R″ represent any group, typically organyl groups or hydrogen atoms. The amide group is called a peptide bond when it is part of the main chain of a protein, and an isopeptide bond when it occurs in a side chain, as in asparagine and glutamine. It can be viewed as a derivative of a carboxylic acid with the hydroxyl group replaced by an amine group ; or, equivalently, an acyl (alkanoyl) group joined to an amine group.
Chymotrypsin (EC 3.4.21.1, chymotrypsins A and B, alpha-chymar ophth, avazyme, chymar, chymotest, enzeon, quimar, quimotrase, alpha-chymar, alpha-chymotrypsin A, alpha-chymotrypsin) is a digestive enzyme component of pancreatic juice acting in the duodenum, where it performs proteolysis, the breakdown of proteins and polypeptides. Chymotrypsin preferentially cleaves peptide amide bonds where the side chain of the amino acid N-terminal to the scissile amide bond (the P1 position) is a large hydrophobic amino acid (tyrosine, tryptophan, and phenylalanine). These amino acids contain an aromatic ring in their side chain that fits into a hydrophobic pocket (the S1 position) of the enzyme. It is activated in the presence of trypsin. The hydrophobic and shape complementarity between the peptide substrate P1 side chain and the enzyme S1 binding cavity accounts for the substrate specificity of this enzyme. Chymotrypsin also hydrolyzes other amide bonds in peptides at slower rates, particularly those containing leucine at the P1 position.

In organic chemistry, a peptide bond is an amide type of covalent chemical bond linking two consecutive alpha-amino acids from C1 of one alpha-amino acid and N2 of another, along a peptide or protein chain.
A polyamide is a polymer with repeating units linked by amide bonds.
The Hofmann rearrangement is the organic reaction of a primary amide to a primary amine with one less carbon atom. The reaction involves oxidation of the nitrogen followed by rearrangement of the carbonyl and nitrogen to give an isocyanate intermediate. The reaction can form a wide range of products, including alkyl and aryl amines.
The Dakin–West reaction is a chemical reaction that transforms an amino-acid into a keto-amide using an acid anhydride and a base, typically pyridine. It is named for Henry Drysdale Dakin and Randolph West. In 2016 Schreiner and coworkers reported the first asymmetric variant of this reaction employing short oligopeptides as catalysts.

ᴅ-Lysergic acid α-hydroxyethylamide, also known as ᴅ-lysergic acid methyl carbinolamide, is an ergoamide and an ergoline. It is perhaps the main constituent of the parasitic fungus, Claviceps paspali; and found in trace amounts in Claviceps Purpurea. C. paspali and C. purpurea are ergot-spreading fungi. Periglandula, Clavicipitacepus fungi, are permanently symbiotically connected to an estimated 450 species of Convolvulaceae and thus generate LAH in some of them. The most well-known ones are Ipomoea tricolor, Turbina corymbosa (coaxihuitl), and Argyreia nervosa.

EX-597 is a fatty acid amide hydrolase inhibitor which is under development for the treatment of social anxiety disorder and post-traumatic stress disorder (PTSD).
Bis(trimethylsilyl)amine (also known as hexamethyldisilazane and HMDS) is an organosilicon compound with the molecular formula [(CH3)3Si]2NH. The molecule is a derivative of ammonia with trimethylsilyl groups in place of two hydrogen atoms. An electron diffraction study shows that silicon-nitrogen bond length (173.5 pm) and Si-N-Si bond angle (125.5°) to be similar to disilazane (in which methyl groups are replaced by hydrogen atoms) suggesting that steric factors are not a factor in regulating angles in this case. This colorless liquid is a reagent and a precursor to bases that are popular in organic synthesis and organometallic chemistry. Additionally, HMDS is also increasingly used as molecular precursor in chemical vapor deposition techniques to deposit silicon carbonitride thin films or coatings.

Fatty-acid amide hydrolase 1 (FAAH) is a member of the serine hydrolase family of enzymes. It was first shown to break down anandamide (AEA), an N-acylethanolamine (NAE) in 1993. In humans, it is encoded by the gene FAAH.
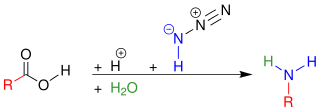
In organic chemistry, the Schmidt reaction is an organic reaction in which an azide reacts with a carbonyl derivative, usually an aldehyde, ketone, or carboxylic acid, under acidic conditions to give an amine or amide, with expulsion of nitrogen. It is named after Karl Friedrich Schmidt (1887–1971), who first reported it in 1924 by successfully converting benzophenone and hydrazoic acid to benzanilide. The intramolecular reaction was not reported until 1991 but has become important in the synthesis of natural products. The reaction is effective with carboxylic acids to give amines (above), and with ketones to give amides (below).
The Bergmann degradation is a series of chemical reactions designed to remove a single amino acid from the carboxylic acid (C-terminal) end of a peptide. First demonstrated by Max Bergmann in 1934, it is a rarely used method for sequencing peptides. The later developed Edman degradation is an improvement upon the Bergmann degradation, instead cleaving the N-terminal amino acid of peptides to produce a hydantoin containing the desired amino acid.

AM404, also known as N-arachidonoylphenolamine, is an active metabolite of paracetamol (acetaminophen), responsible for all or part of its analgesic action and anticonvulsant effects. Chemically, it is the amide formed from 4-aminophenol and arachidonic acid. AM404 is one of the AM cannabinoids discovered by Alexandros Makriyannis and his team.
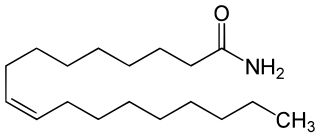
Oleamide is an organic compound with the formula CH3(CH2)7CH=CH(CH2)7CONH2. It is the amide derived from the fatty acid oleic acid. It is a colorless waxy solid and occurs in nature. Sometimes labeled as a fatty acid primary amide (FAPA), it is biosynthesized from N-oleoylglycine.

Methylisopropyllysergamide is an analogue of LSD that was originally discovered by Albert Hofmann at Sandoz during the original structure-activity research into LSD. It has subsequently been investigated in more detail by the team led by David E. Nichols at Purdue University. Methylisopropyllysergamide is a structural isomer of LSD, with the alkyl groups on the amide nitrogen having been subjected to a methylene shuffle. MIPLA and its ethylisopropyl homologue EIPLA are the only simple N,N-dialkyl lysergamides that approach the potency of LSD itself, being around 1/3-1/2 the potency of LSD, while all other dialkyl analogues tested are only around 1/10 as potent as LSD, although some N-monoalkyl lysergamides such as the sec-butyl and t-butyl derivatives were also found to show an activity profile and potency comparable to LSD, and the mono-isopropyl derivative is only slightly weaker than MIPLA. Apart from its lower potency, the hallucinogenic effects of methylisopropyllysergamide are similar to those of LSD itself, and the main use for this drug has been in studies of the binding site at the 5-HT2A receptor through which LSD exerts most of its pharmacological effects.

Lysergic acid 2-butyl amide (2-Butyllysergamide, LSB) is an analogue of LSD originally developed by Richard Pioch at Eli Lilly in the 1950s, but mostly publicised through research conducted by the team led by David E. Nichols at Purdue University. It is a structural isomer of LSD, with the two ethyl groups on the amide nitrogen having been replaced by a single sec-butyl group, joined at the 2-position. It is one of the few lysergamide derivatives to exceed the potency of LSD in animal drug discrimination assays, with the (R) isomer having an ED50 of 33nmol/kg for producing drug-appropriate responding, vs 48nmol/kg for LSD itself. The corresponding (R)-2-pentyl analogue has higher binding affinity for the 5-HT1A and 5-HT2A receptors, but is less potent in producing drug-appropriate responding, suggesting that the butyl compound has a higher efficacy at the receptor target. The drug discrimination assay for LSD in rats involves both 5-HT1A and 5-HT2A mediated components, and while lysergic acid 2-butyl amide is more potent than LSD as a 5-HT1A agonist, it is slightly less potent as a 5-HT2A agonist, and so would probably be slightly less potent than LSD as a hallucinogen in humans. The main use for this drug has been in studies of the binding site at the 5-HT2A receptor through which LSD exerts most of its pharmacological effects, with the stereoselective activity of these unsymmetric monoalkyl lysergamides foreshadowing the subsequent development of compounds such as lysergic acid 2,4-dimethylazetidide (LSZ).

Hopantenic acid, also known as N-pantoyl-GABA, is a central nervous system depressant. Formulated as the calcium salt, it is used as a pharmaceutical drug in the Russian Federation for a variety of neurological, psychological and psychiatric conditions and sold as Pantogam.

Biochanin A is an O-methylated isoflavone. It is a natural organic compound in the class of phytochemicals known as flavonoids. Biochanin A can be found in red clover in soy, in alfalfa sprouts, in peanuts, in chickpea and in other legumes.

Arachidonoyl serotonin is an endogenous lipid signaling molecule. It was first described in 1998 as being an inhibitor of fatty acid amide hydrolase (FAAH). In 2007, it was shown to have analgesic properties and to act as an antagonist of the TRPV1 receptor. In 2011, it was shown to be present in the ileum and jejunum of the gastrointestinal tract and modulate glucagon-like peptide-1 (GLP-1) secretion. In addition to this, in 2016, AA-5-HT was also found to affect the signaling mechanisms responsible for anxiety, by inhibiting dopamine release from the Basolateral amygdala following fear behavior. In 2017, AA-5-HT was tested in its effects on the sleep wake cycle, where it was found to affect the sleep homeostasis when used in conjunction with molecules and chemicals that affect wake-related neurotransmitters.
4-Nonylphenylboronic acid is a potent and selective inhibitor of the enzyme fatty acid amide hydrolase (FAAH), with an IC50 of 9.1nM, and 870x selectivity for FAAH over the related enzyme MAGL, which it inhibits with an IC50 of 7900nM. It is also a weaker inhibitor of the enzymes endothelial lipase and lipoprotein lipase, with IC50 values of 100 nM and 1400 nM respectively.