
Non-steroidal anti-inflammatory drugs (NSAID) are members of a therapeutic drug class which reduces pain, decreases inflammation, decreases fever, and prevents blood clots. Side effects depend on the specific drug, its dose and duration of use, but largely include an increased risk of gastrointestinal ulcers and bleeds, heart attack, and kidney disease.

Dermatitis is a term used for different types of skin inflammation, typically characterized by itchiness, redness and a rash. In cases of short duration, there may be small blisters, while in long-term cases the skin may become thickened. The area of skin involved can vary from small to covering the entire body. Dermatitis is also called eczema but the same term is often used for the most common type of skin inflammation, atopic dermatitis.

Tacrolimus, sold under the brand name Prograf among others, is an immunosuppressive drug. After allogenic organ transplant, the risk of organ rejection is moderate. To lower the risk of organ rejection, tacrolimus is given. The drug can also be sold as a topical medication in the treatment of T cell-mediated diseases such as eczema and psoriasis. For example, it is prescribed for severe refractory uveitis after a bone marrow transplant, exacerbations of minimal change disease, Kimura's disease, and vitiligo. It can be used to treat dry eye syndrome in cats and dogs.

Diclofenac, sold under the brand name Voltaren among others, is a nonsteroidal anti-inflammatory drug (NSAID) used to treat pain and inflammatory diseases such as gout. It can be taken orally, inserted rectally as a suppository, injected intramuscularly, injected intravenously, applied to the skin topically, or through eye drops. Improvements in pain last up to eight hours. It is also available as the fixed-dose combination diclofenac/misoprostol (Arthrotec) to help protect the stomach.

Seborrhoeic dermatitis is a long-term skin disorder. Symptoms include flaky, scaly, greasy, and occasionally itchy and inflamed skin. Areas of the skin rich in oil-producing glands are often affected including the scalp, face, and chest. It can result in social or self-esteem problems. In babies, when the scalp is primarily involved, it is called cradle cap. Mild seborrhoeic dermatitis of the scalp may be described in lay terms as dandruff due to the dry, flaky character of the skin. However, as dandruff may refer to any dryness or scaling of the scalp, not all dandruff is seborrhoeic dermatitis. Seborrhoeic dermatitis is sometimes inaccurately referred to as seborrhoea.

Triamcinolone is a glucocorticoid used to treat certain skin diseases, allergies, and rheumatic disorders among others. It is also used to prevent worsening of asthma and chronic obstructive pulmonary disease (COPD). It can be taken in various ways including by mouth, injection into a muscle, and inhalation.

Lumiracoxib is a COX-2 selective inhibitor nonsteroidal anti-inflammatory drug.

Piroxicam is a nonsteroidal anti-inflammatory drug (NSAID) of the oxicam class used to relieve the symptoms of painful inflammatory conditions like arthritis. Piroxicam works by preventing the production of endogenous prostaglandins which are involved in the mediation of pain, stiffness, tenderness and swelling. The medicine is available as capsules, tablets and, in some countries, as a prescription-free gel 0.5%. It is also available in a betadex formulation, which allows a more rapid absorption of piroxicam from the digestive tract. Piroxicam is one of the few NSAIDs that can be given parenteral routes.

Nimesulide is a nonsteroidal anti-inflammatory drug (NSAID) with pain medication and fever reducing properties. Its approved indications are the treatment of acute pain, the symptomatic treatment of osteoarthritis, and primary dysmenorrhoea in adolescents and adults above 12 years old.

Desonide (INN) is a low-potency topical corticosteroid anti-inflammatory that has been available since the 1970s. It is primarily used to treat atopic dermatitis (eczema), seborrheic dermatitis, contact dermatitis and psoriasis in both adults and children. It has a fairly good safety profile and is available as a cream, ointment, lotion, and as a foam under the tradename Verdeso Foam. Other trade names for creams, lotions, and ointments include Tridesilon, DesOwen, Desonate. It is a group VI corticosteroid under US classification, the second least potent group.

Ichthammol or ammonium bituminosulfonate, also known as black ointment, is a medication derived from sulfur-rich oil shale. It is used as a treatment for different skin diseases, including eczema and psoriasis. It is applied on the skin as an ointments, most commonly containing 10% or 20% ichthammol.

Betamethasone dipropionate is a glucocorticoid steroid with anti-inflammatory and immunosuppressive properties. It is applied as a topical cream, ointment, lotion or gel (Diprolene) to treat itching and other skin conditions such as eczema. Minor side effects include dry skin and mild, temporary stinging when applied. Betamethasone dipropionate is a "super high potency" corticosteroid used to treat inflammatory skin conditions such as dermatitis, eczema and psoriasis. It is a synthetic analog of the adrenal corticosteroids. Although its exact mechanism of action is not known, it is effective when applied topically to cortico-responsive inflammatory dermatoses. It is available as a generic medication.

Moxifloxacin is an antibiotic, used to treat bacterial infections, including pneumonia, conjunctivitis, endocarditis, tuberculosis, and sinusitis. It can be given by mouth, by injection into a vein, and as an eye drop.

Mometasone, also known as mometasone furoate, is a steroid medication used to treat certain skin conditions, hay fever, and asthma. Specifically it is used to prevent rather than treat asthma attacks. It can be applied to the skin, inhaled, or used in the nose. Mometasone furoate, not mometasone, is used in medical products.

Tolperisone is a centrally acting skeletal muscle relaxant used for the treatment of increased muscle tone associated with neurological diseases. It has been used since the 1960s.
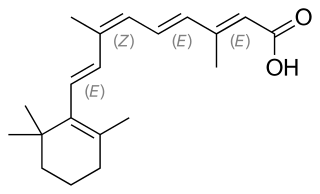
Alitretinoin, or 9-cis-retinoic acid, is a form of vitamin A. It is also used in medicine as an antineoplastic (anti-cancer) agent developed by Ligand Pharmaceuticals. It is a first generation retinoid. Ligand gained Food and Drug Administration (FDA) approval for alitretinoin in February 1999.

Bromfenac is a nonsteroidal anti-inflammatory drug (NSAID) marketed in the US as an ophthalmic solution by ISTA Pharmaceuticals for short-term, local use. Prolensa and Bromday are the once-daily formulation of bromfenac, while Xibrom was approved for twice-daily administration. In the European Union, the brand name is Yellox. Bromfenac is indicated for the treatment of ocular inflammation and pain after cataract surgery.

TopoTarget was a Copenhagen-based biotechnology company focused on the discovery and development of drugs and therapies to treat cancer. In 2014, it merged with BioAlliance Pharma and is now part of Onxeo.
Givinostat, sold under the brand name Duvyzat is a medication used for the treatment of Duchenne muscular dystrophy. It is a histone deacetylase inhibitor with potential anti-inflammatory, anti-angiogenic, and antineoplastic activities. It is a histone deacetylase (HDAC) inhibitor that works by targeting pathogenic processes to reduce inflammation and loss of muscle.

Delgocitinib, sold under the brand name Corectim among others, is a medication used for the treatment of autoimmune disorders and hypersensitivity, including inflammatory skin conditions. Delgocitinib was developed by Japan Tobacco and approved in Japan for the treatment of atopic dermatitis. In the United States, delgocitinib is in Phase III clinical trials and the Food and Drug Administration has granted delgocitinib fast track designation for topical treatment of adults with moderate to severe chronic hand eczema.



















