
Phenethylamine (PEA) is an organic compound, natural monoamine alkaloid, and trace amine, which acts as a central nervous system stimulant in humans. In the brain, phenethylamine regulates monoamine neurotransmission by binding to trace amine-associated receptor 1 (TAAR1) and inhibiting vesicular monoamine transporter 2 (VMAT2) in monoamine neurons. To a lesser extent, it also acts as a neurotransmitter in the human central nervous system. In mammals, phenethylamine is produced from the amino acid L-phenylalanine by the enzyme aromatic L-amino acid decarboxylase via enzymatic decarboxylation. In addition to its presence in mammals, phenethylamine is found in many other organisms and foods, such as chocolate, especially after microbial fermentation.

2C-T-2 is a psychedelic and entactogenic phenethylamine of the 2C family. It was first synthesized in 1981 by Alexander Shulgin, and rated by him as one of the "magical half-dozen" most important psychedelic phenethylamine compounds. The drug has structural and pharmacodynamic properties similar to those of 2C-T-7.
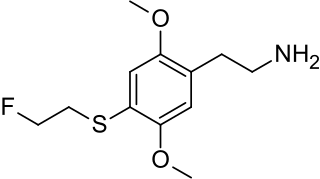
2C-T-21 is a psychedelic phenethylamine of the 2C family sometimes used as an entheogen. It was first synthesized by Alexander Shulgin.

2C-T-8 is a psychedelic phenethylamine of the 2C family. It was first synthesized by Alexander Shulgin, sometimes used as an entheogen.

Escaline (3,5-methoxy-4-ethoxyphenethylamine) is a psychedelic drug and entheogen of the phenethylamine class of compounds. Escaline was first synthesized and reported in the scientific literature by Benington, et al., in 1954, but was later re-examined in the laboratory of David E. Nichols, who prepared a series of mescaline analogues that included escaline, proscaline, and isoproscaline. The effects of this and related mescaline analogues in humans were first described by Alexander Shulgin. In his book PiHKAL , Shulgin lists the dosage range as 40 to 60 mg, consumed orally. The duration of action was stated to be 8–12 hours.

2C-G is a psychedelic phenethylamine of the 2C-series. First synthesized by Alexander Shulgin, it is sometimes used as an entheogen. It has structural and pharmacodynamic properties similar to 2C-D and Ganesha. Like many of the phenethylamines in PiHKAL, 2C-G and its homologs have only been taken by Shulgin and a small test group, making it difficult to ensure completeness when describing effects.

2C-TFM is a psychedelic phenethylamine of the 2C family. It was first synthesized in the laboratory of David E. Nichols. It has also been called 2C-CF3, a name derived from the Para-trifluoromethyl group it contains.

Macromerine is a phenethylamine derivative. It was first identified from the cactus Coryphantha macromeris. It can also be found in C. runyonii, C. elephantidens, and other related members of the family Cactaceae. The plants may have been used by Tarahumara shamans for their entheogenic effects.
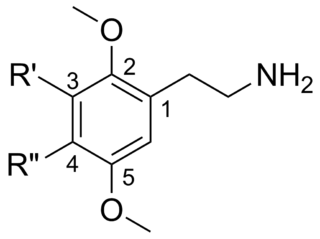
2C (2C-x) is a general name for the family of psychedelic phenethylamines containing methoxy groups on the 2 and 5 positions of a benzene ring. Most of these compounds also carry lipophilic substituents at the 4 position, usually resulting in more potent and more metabolically stable and longer acting compounds. Most of the currently known 2C compounds were first synthesized by Alexander Shulgin in the 1970s and 1980s and published in his book PiHKAL. Shulgin also coined the term 2C, being an acronym for the 2 carbon atoms between the benzene ring and the amino group.

2C-T-13 is a psychedelic phenethylamine of the 2C family. It was presumably first synthesized by Alexander Shulgin and reported in his book PiHKAL.

2C-T-15 or 2,5-dimethoxy-4-(β-cyclopropylthio)phenethylamine is a psychedelic phenethylamine of the 2C family. It was presumably first synthesized by Alexander Shulgin and reported in his book PiHKAL .
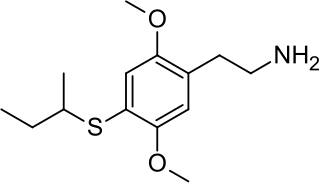
2C-T-17 or 2,5-dimethoxy-4-(β-secbutylthio)phenethylamine is a psychedelic phenethylamine of the 2C family. It was presumably first synthesized by Alexander Shulgin and reported in his book PiHKAL .

Substituted Methylenedioxyphenethylamines represent a diverse chemical class of compounds derived from phenethylamines. This category encompasses numerous psychoactive substances with entactogenic, psychedelic, and/or stimulant properties, in addition to entheogens. These compounds find application as research chemicals, designer drugs, and recreational substances.

2C-YN is an analog of phenethylamine that can be synthesized from 2C-I. Very little data exists about the pharmacological properties, metabolism, and toxicity of 2C-YN, although Daniel Trachsel lists it as having a dosage of around 50mg and a duration of around 2 hours, with relatively mild psychedelic effects.

Substituted phenethylamines are a chemical class of organic compounds that are based upon the phenethylamine structure; the class is composed of all the derivative compounds of phenethylamine which can be formed by replacing, or substituting, one or more hydrogen atoms in the phenethylamine core structure with substituents.

4-Substituted-2,5-dimethoxyamphetamines (DOx) is a chemical class of substituted amphetamine derivatives featuring methoxy groups at the 2- and 5- positions of the phenyl ring, and a substituent such as alkyl or halogen at the 4- position of the phenyl ring. Most compounds of this class are potent and long-lasting psychedelic drugs, and act as highly selective 5-HT2A, 5-HT2B, and 5-HT2C receptor partial agonists. A few bulkier derivatives such as DOAM have similarly high binding affinity for 5-HT2 receptors but instead act as antagonists, and so do not produce psychedelic effects though they retain amphetamine-like stimulant effects.
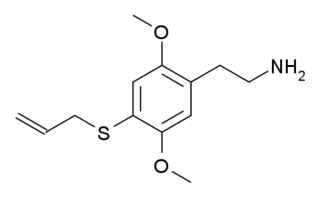
2C-T-16 is a lesser-known psychedelic drug. It was originally named by Alexander Shulgin as described in his book PiHKAL, however while Shulgin began synthesis of this compound he only got as far as the nitrostyrene intermediate, and did not complete the final synthetic step. Synthesis of 2C-T-16 was finally achieved by Daniel Trachsel some years later, and it was subsequently reported as showing similar psychedelic activity to related compounds, with a dose range of 10–25 mg and a duration of 4–6 hours, making it around the same potency as the better-known saturated analogue 2C-T-7, but with a significantly shorter duration of action. Binding studies in vitro showed 2C-T-16 to have a binding affinity of 44 nM at 5-HT2A and 15 nM at 5-HT2C. 2C-T-16 and related derivatives are potent partial agonists of the 5-HT1A, 5-HT2A, 5-HT2B and 5-HT2C receptors and induce a head-twitch response in mice.

2C-Se is a lesser-known psychedelic drug. It was originally named by Alexander Shulgin as described in his book PiHKAL. Shulgin considered 2C-Se to be around three times the potency of mescaline, but was too concerned about toxicity to test it extensively, though he considered it noteworthy as the only psychedelic drug to contain a selenium atom.

2C-CP (2C-cP) is a recreational designer drug from the substituted phenethylamine family, with psychedelic effects. It was first synthesised by Daniel Trachsel and colleagues in 2006. It has a binding affinity (Ki) of 95 nM at the serotonin receptor 5-HT2A and 41 nM at 5-HT2C and is active at a dosage of between 15 and 35 mg with a duration of 3 to 6 hours.

2C-V is a recreational designer drug from the substituted phenethylamine family, with psychedelic effects. It was first synthesised by Daniel Trachsel and colleagues in 2006. It is active at a dosage of 25 mg with a duration of around 5 hours.





















