
Positron emission tomography (PET) is a functional imaging technique that uses radioactive substances known as radiotracers to visualize and measure changes in metabolic processes, and in other physiological activities including blood flow, regional chemical composition, and absorption. Different tracers are used for various imaging purposes, depending on the target process within the body.

Single-photon emission computed tomography is a nuclear medicine tomographic imaging technique using gamma rays. It is very similar to conventional nuclear medicine planar imaging using a gamma camera, but is able to provide true 3D information. This information is typically presented as cross-sectional slices through the patient, but can be freely reformatted or manipulated as required.

Nuclear medicine, is a medical specialty involving the application of radioactive substances in the diagnosis and treatment of disease. Nuclear imaging is, in a sense, radiology done inside out, because it records radiation emitted from within the body rather than radiation that is transmitted through the body from external sources like X-ray generators. In addition, nuclear medicine scans differ from radiology, as the emphasis is not on imaging anatomy, but on the function. For such reason, it is called a physiological imaging modality. Single photon emission computed tomography (SPECT) and positron emission tomography (PET) scans are the two most common imaging modalities in nuclear medicine.
A radioactive tracer, radiotracer, or radioactive label is a synthetic derivative of a natural compound in which one or more atoms have been replaced by a radionuclide. By virtue of its radioactive decay, it can be used to explore the mechanism of chemical reactions by tracing the path that the radioisotope follows from reactants to products. Radiolabeling or radiotracing is thus the radioactive form of isotopic labeling. In biological contexts, experiments that use radioisotope tracers are sometimes called radioisotope feeding experiments.

Iodobenzamide is a pharmaceutical drug used for diagnostic purposes. It is a dopamine antagonist and it can be used by nuclear medicine physicians as a radioactive tracer for SPECT where the radioactive isotope is iodine-123 or iodine-125. The main purpose of a brain study with IBZM is the differentiation of Parkinson's disease from other neurodegenerative diseases such as Lewy Body dementia and multiple system atrophy.

Scintigraphy, also known as a gamma scan, is a diagnostic test in nuclear medicine, where radioisotopes attached to drugs that travel to a specific organ or tissue (radiopharmaceuticals) are taken internally and the emitted gamma radiation is captured by gamma cameras, which are external detectors that form two-dimensional images in a process similar to the capture of x-ray images. In contrast, SPECT and positron emission tomography (PET) form 3-dimensional images and are therefore classified as separate techniques from scintigraphy, although they also use gamma cameras to detect internal radiation. Scintigraphy is unlike a diagnostic X-ray where external radiation is passed through the body to form an image.

Perfusion is the passage of fluid through the circulatory system or lymphatic system to an organ or a tissue, usually referring to the delivery of blood to a capillary bed in tissue. Perfusion may also refer to fixation via perfusion, used in histological studies. Perfusion is measured as the rate at which blood is delivered to tissue, or volume of blood per unit time per unit tissue mass. The SI unit is m3/(s·kg), although for human organs perfusion is typically reported in ml/min/g. The word is derived from the French verb perfuser, meaning to "pour over or through". All animal tissues require an adequate blood supply for health and life. Poor perfusion (malperfusion), that is, ischemia, causes health problems, as seen in cardiovascular disease, including coronary artery disease, cerebrovascular disease, peripheral artery disease, and many other conditions.

There are 40 known isotopes of iodine (53I) from 108I to 147I; all undergo radioactive decay except 127I, which is stable. Iodine is thus a monoisotopic element.
Iodine-123 (123I) is a radioactive isotope of iodine used in nuclear medicine imaging, including single-photon emission computed tomography (SPECT) or SPECT/CT exams. The isotope's half-life is 13.2232 hours; the decay by electron capture to tellurium-123 emits gamma radiation with a predominant energy of 159 keV. In medical applications, the radiation is detected by a gamma camera. The isotope is typically applied as iodide-123, the anionic form.
Copper-64 (64Cu) is a positron and beta emitting isotope of copper, with applications for molecular radiotherapy and positron emission tomography. Its unusually long half-life (12.7-hours) for a positron-emitting isotope makes it increasingly useful when attached to various ligands, for PET and PET-CT scanning.

RTI(-4229)-55, also called RTI-55 or iometopane, is a phenyltropane-based psychostimulant used in scientific research and in some medical applications. This drug was first cited in 1991. RTI-55 is a non-selective dopamine reuptake inhibitor derived from methylecgonidine. However, more selective analogs are derived by conversion to "pyrrolidinoamido" RTI-229, for instance. Due to the large bulbous nature of the weakly electron withdrawing iodo halogen atom, RTI-55 is the most strongly serotonergic of the simple para-substituted troparil based analogs. In rodents RTI-55 actually caused death at a dosage of 100 mg/kg, whereas RTI-51 and RTI-31 did not. Another notable observation is the strong propensity of RTI-55 to cause locomotor activity enhancements, although in an earlier study, RTI-51 was actually even stronger than RTI-55 in shifting baseline LMA. This observation serves to highlight the disparities that can arise between studies.

Ioflupane (123I) is the international nonproprietary name (INN) of a cocaine analogue which is a neuro-imaging radiopharmaceutical drug, used in nuclear medicine for the diagnosis of Parkinson's disease and the differential diagnosis of Parkinson's disease over other disorders presenting similar symptoms. During the DaT scan procedure it is injected into a patient and viewed with a gamma camera in order to acquire SPECT images of the brain with particular respect to the striatum, a subcortical region of the basal ganglia. The drug is sold under the brand name Datscan and is manufactured by GE Healthcare, formerly Amersham plc.
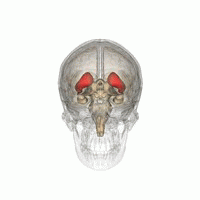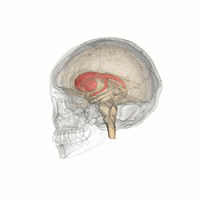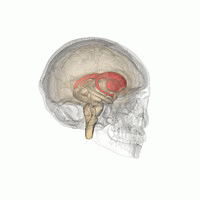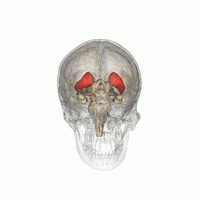
DaT Scan commonly refers to a diagnostic method, based on SPECT imaging, to investigate if there is a loss of dopaminergic neurons in striatum. The term may also refer to a brand name of Ioflupane (123I) tracer used for the study. The scan principle is based on use of the radiopharmaceutical Ioflupane (123I) which binds to dopamine transporters (DaT). The signal from them is then detected by the use of single-photon emission computed tomography (SPECT) which uses special gamma-cameras to create a pictographic representation of the distribution of dopamine transporters in the brain.
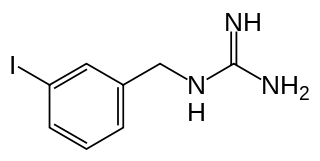
Iobenguane, or MIBG, is an aralkylguanidine analog of the adrenergic neurotransmitter norepinephrine (noradrenaline), typically used as a radiopharmaceutical. It acts as a blocking agent for adrenergic neurons. When radiolabeled, it can be used in nuclear medicinal diagnostic and therapy techniques as well as in neuroendocrine chemotherapy treatments.

Altanserin is a compound that binds to the 5-HT2A receptor. Labeled with the isotope fluorine-18 it is used as a radioligand in positron emission tomography (PET) studies of the brain, i.e., studies of the 5-HT2A neuroreceptors. Besides human neuroimaging studies altanserin has also been used in the study of rats.

DASB, also known as 3-amino-4-(2-dimethylaminomethylphenylsulfanyl)-benzonitrile, is a compound that binds to the serotonin transporter. Labeled with carbon-11 — a radioactive isotope — it has been used as a radioligand in neuroimaging with positron emission tomography (PET) since around year 2000. In this context it is regarded as one of the superior radioligands for PET study of the serotonin transporter in the brain, since it has high selectivity for the serotonin transporter.
Perfusion is the passage of fluid through the lymphatic system or blood vessels to an organ or a tissue. The practice of perfusion scanning is the process by which this perfusion can be observed, recorded and quantified. The term perfusion scanning encompasses a wide range of medical imaging modalities.

5-I-R91150 is a compound that acts as a potent and selective antagonist of 5-HT2A receptors. Its main application is as its iodine-123 radiolabeled form, in which it can be used in SPECT scanning in human neuroimaging studies, to examine the distribution of the 5-HT2A receptor subtype in the brain, e.g. with respect to sex and age and in adults with Asperger syndrome or Alzheimer's disease.

Mefway is a serotonin 5-HT1A receptor antagonist used in medical research, usually in the form of mefway (18F) as a positron emission tomography (PET) radiotracer.

Iomazenil is an antagonist and partial inverse agonist of benzodiazepine and a potential treatment for alcohol use disorder. The compound was introduced in 1989 by pharmaceutical company Hoffmann-La Roche as an Iodine-123-labelled SPECT tracer for imaging benzodiazepine receptors in the brain. Iomazenil is an analogue of flumazenil (Ro15-1788).
















