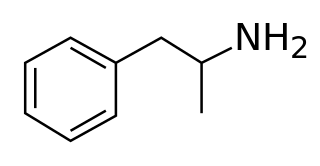
Amphetamine is a central nervous system (CNS) stimulant that is used in the treatment of attention deficit hyperactivity disorder (ADHD), narcolepsy, and obesity. Amphetamine was discovered as a chemical in 1887 by Lazăr Edeleanu, and then as a drug in the late 1920s. It exists as two enantiomers: levoamphetamine and dextroamphetamine. Amphetamine properly refers to a specific chemical, the racemic free base, which is equal parts of the two enantiomers in their pure amine forms. The term is frequently used informally to refer to any combination of the enantiomers, or to either of them alone. Historically, it has been used to treat nasal congestion and depression. Amphetamine is also used as an athletic performance enhancer and cognitive enhancer, and recreationally as an aphrodisiac and euphoriant. It is a prescription drug in many countries, and unauthorized possession and distribution of amphetamine are often tightly controlled due to the significant health risks associated with recreational use.

Phenethylamine (PEA) is an organic compound, natural monoamine alkaloid, and trace amine, which acts as a central nervous system stimulant in humans. In the brain, phenethylamine regulates monoamine neurotransmission by binding to trace amine-associated receptor 1 (TAAR1) and inhibiting vesicular monoamine transporter 2 (VMAT2) in monoamine neurons. To a lesser extent, it also acts as a neurotransmitter in the human central nervous system. In mammals, phenethylamine is produced from the amino acid L-phenylalanine by the enzyme aromatic L-amino acid decarboxylase via enzymatic decarboxylation. In addition to its presence in mammals, phenethylamine is found in many other organisms and foods, such as chocolate, especially after microbial fermentation.
Stimulant psychosis is a mental disorder characterized by psychotic symptoms. It involves and typically occurs following an overdose or several day binge on psychostimulants, though one study reported occurrences at regularly prescribed doses in approximately 0.1% of individuals within the first several weeks after starting amphetamine or methylphenidate therapy. Methamphetamine psychosis, or long-term effects of stimulant use in the brain, depend upon genetics and may persist for some time.

2,5-Dimethoxy-4-ethylamphetamine is a psychedelic drug of the phenethylamine and amphetamine chemical classes. It was first synthesized by Alexander Shulgin, and was described in his book PiHKAL.

β-Methylphenethylamine is an organic compound of the phenethylamine class, and a positional isomer of the drug amphetamine, with which it shares some properties. In particular, both amphetamine and β-methylphenethylamine are human TAAR1 agonists. In appearance, it is a colorless or yellowish liquid.

The substituted methylenedioxyphenethylamines represent a diverse chemical class of compounds derived from phenethylamines. This category encompasses numerous psychoactive substances with entactogenic, psychedelic, and/or stimulant properties, in addition to entheogens. These compounds find application as research chemicals, designer drugs, and recreational substances.
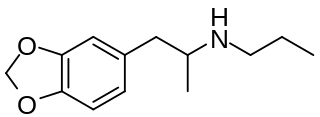
3,4-Methylenedioxy-N-propylamphetamine is a lesser-known psychedelic drug and a substituted amphetamine. MDPR was first synthesized by Alexander Shulgin. In his book PiHKAL, the minimum dosage is listed as 200 mg, and the duration unknown. MDPR is a promoter; by itself it has almost no effects on the mind, but it promotes the effects of hallucinogens, similarly to the closely related MDPH.

2,5-Dimethoxy-4-propylamphetamine (DOPR) is a psychedelic drug of the phenethylamine and amphetamine chemical classes. It was first synthesized by Alexander Shulgin, and was described in his book PiHKAL. Shulgin described DOPR as a "heavy duty psychedelic", complete with alterations of the thought process and visual distortion. Very little data exists about the pharmacological properties, metabolism, and toxicity of DOPR.

Etilamfetamine is a stimulant drug of the phenethylamine and amphetamine chemical classes. It was invented in the early 20th century and was subsequently used as an anorectic or appetite suppressant in the 1950s, but was not as commonly used as other amphetamines such as amphetamine, methamphetamine, and benzphetamine, and was largely discontinued once newer drugs such as phenmetrazine were introduced. It most likely acts primarily as a dopamine releasing agent. Its activity as a norepinephrine or serotonin releasing agent is not known.

1-Phenyl-2-propylaminopentane is an experimental drug related to selegiline which acts as a catecholaminergic activity enhancer (CAE).

Trace amine-associated receptor 1 (TAAR1) is a trace amine-associated receptor (TAAR) protein that in humans is encoded by the TAAR1 gene. TAAR1 is an intracellular amine-activated Gs-coupled and Gq-coupled G protein-coupled receptor (GPCR) that is primarily expressed in several peripheral organs and cells, astrocytes, and in the intracellular milieu within the presynaptic plasma membrane of monoamine neurons in the central nervous system (CNS). TAAR1 was discovered in 2001 by two independent groups of investigators, Borowski et al. and Bunzow et al. TAAR1 is one of six functional human trace amine-associated receptors, which are so named for their ability to bind endogenous amines that occur in tissues at trace concentrations. TAAR1 plays a significant role in regulating neurotransmission in dopamine, norepinephrine, and serotonin neurons in the CNS; it also affects immune system and neuroimmune system function through different mechanisms.

Substituted phenethylamines are a chemical class of organic compounds that are based upon the phenethylamine structure; the class is composed of all the derivative compounds of phenethylamine which can be formed by replacing, or substituting, one or more hydrogen atoms in the phenethylamine core structure with substituents.
Substituted amphetamines are a class of compounds based upon the amphetamine structure; it includes all derivative compounds which are formed by replacing, or substituting, one or more hydrogen atoms in the amphetamine core structure with substituents. The compounds in this class span a variety of pharmacological subclasses, including stimulants, empathogens, and hallucinogens, among others. Examples of substituted amphetamines are amphetamine (itself), methamphetamine, ephedrine, cathinone, phentermine, mephentermine, tranylcypromine, bupropion, methoxyphenamine, selegiline, amfepramone (diethylpropion), pyrovalerone, MDMA (ecstasy), and DOM (STP).
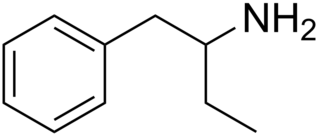
Phenylisobutylamine, also known as α-ethylphenethylamine, Butanphenamine, B or AEPEA, is a stimulant drug of the phenethylamine class. It is a higher homologue of amphetamine, differing from amphetamine's molecular structure only by the substitution of the methyl group at the alpha position of the side chain with an ethyl group. Compared to amphetamine, phenylisobutylamine has strongly reduced dopaminergic effects, and instead acts as a selective norepinephrine releasing agent. The dextroisomer of phenylisobutylamine partially substitutes for dextroamphetamine in rats.

4-Substituted-2,5-dimethoxyamphetamines (DOx) is a chemical class of substituted amphetamine derivatives featuring methoxy groups at the 2- and 5- positions of the phenyl ring, and a substituent such as alkyl or halogen at the 4- position of the phenyl ring. Most compounds of this class are potent and long-lasting psychedelic drugs, and act as highly selective 5-HT2A, 5-HT2B, and 5-HT2C receptor partial agonists. A few bulkier derivatives such as DOAM have similarly high binding affinity for 5-HT2 receptors but instead act as antagonists, and so do not produce psychedelic effects though they retain amphetamine-like stimulant effects.
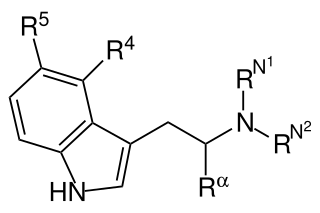
Substituted tryptamines, or serotonin analogues, are organic compounds which may be thought of as being derived from tryptamine itself. The molecular structures of all tryptamines contain an indole ring, joined to an amino (NH2) group via an ethyl (−CH2–CH2−) sidechain. In substituted tryptamines, the indole ring, sidechain, and/or amino group are modified by substituting another group for one of the hydrogen (H) atoms.
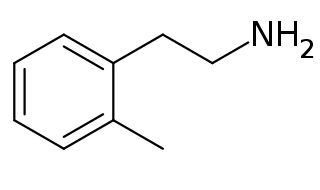
2-Methylphenethylamine (2MPEA) is an organic compound with the chemical formula of C9H13N. 2MPEA is a human trace amine associated receptor 1 (TAAR1) agonist, a property which it shares with its monomethylated phenethylamine isomers, such as amphetamine (α-methylphenethylamine), β-methylphenethylamine, and N-methylphenethylamine.

3-Methylphenethylamine (3MPEA) is an organic compound with the chemical formula of C9H13N. 3MPEA is a human trace amine associated receptor 1 (TAAR1) agonist, a property which it shares with its monomethylated phenethylamine isomers, such as amphetamine (α-methylphenethylamine), β-methylphenethylamine, and N-methylphenethylamine.

4-Methylphenethylamine (4MPEA), also known as para-methylphenethylamine, is an organic compound with the chemical formula of C9H13N. 4MPEA is a human trace amine associated receptor 1 (TAAR1) agonist, a property which it shares with its monomethylated phenethylamine isomers, such as amphetamine (α-methylphenethylamine), β-methylphenethylamine, and N-methylphenethylamine. 4MPEA also appears to inhibit the human cytochrome P450 enzymes CYP1A2 and CYP2A6, based upon the published literature.
Butylamphetamine (code name PAL-90), also known as N-butylamphetamine (NBA), is a psychostimulant of the substituted amphetamine family which was never marketed. It is the N-butyl analogue of amphetamine and is approximately 6-fold less potent than amphetamine in rats. The drug has been found to be inactive as a dopamine reuptake inhibitor or releasing agent (IC50Tooltip half-maximal inhibitory concentration and EC50Tooltip half-maximal effective concentration > 10,000 nM, respectively). With regard to structure–activity relationships, the potency of N-substituted amphetamine derivatives decreases with increasing chain length in terms of both in vitro and in vivo activity. The pharmacokinetics of butylamphetamine have been studied in humans. It can be metabolized by CYP2D6 via ring hydroxylation similarly to amphetamine.


















