In chemistry, an enantiomer – also called optical isomer, antipode, or optical antipode – is one of two stereoisomers that are nonsuperposable onto their own mirror image. Enantiomers of each other are much like one's right and left hands; without mirroring one of them, hands cannot be superposed onto each other. It is solely a relationship of chirality and the permanent three-dimensional relationships among molecules or other chemical structures: no amount of re-orientiation of a molecule as a whole or conformational change converts one chemical into its enantiomer. Chemical structures with chirality rotate plane-polarized light. A mixture of equal amounts of each enantiomer, a racemic mixture or a racemate, does not rotate light.
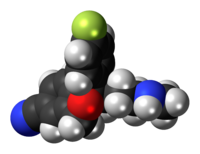
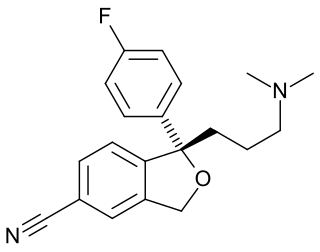
Escitalopram, sold under the brand names Lexapro and Cipralex, among others, is an antidepressant of the selective serotonin reuptake inhibitor (SSRI) class. Escitalopram is mainly used to treat major depressive disorder and generalized anxiety disorder. It is taken by mouth, available commercially as an oxalate salt exclusively.

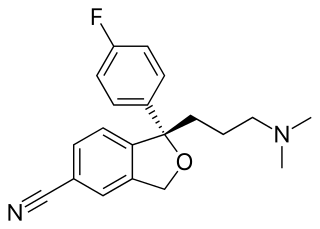
Citalopram, sold under the brand name Celexa among others, is an antidepressant of the selective serotonin reuptake inhibitor (SSRI) class. It is used to treat major depressive disorder, obsessive compulsive disorder, panic disorder, and social phobia. The antidepressant effects may take one to four weeks to occur. It is typically taken orally. In some European countries, it is sometimes given intravenously to initiate treatment, before switching to the oral route of administration for continuation of treatment. It has also been used intravenously in other parts of the world in some other circumstances.
In chemistry, racemization is a conversion, by heat or by chemical reaction, of an optically active compound into a racemic form. This creates a 1:1 molar ratio of enantiomers and is referred to as a racemic mixture. Plus and minus forms are called Dextrorotation and levorotation. The D and L enantiomers are present in equal quantities, the resulting sample is described as a racemic mixture or a racemate. Racemization can proceed through a number of different mechanisms, and it has particular significance in pharmacology as different enantiomers may have different pharmaceutical effects.
ATC code N06Psychoanaleptics is a therapeutic subgroup of the Anatomical Therapeutic Chemical Classification System, a system of alphanumeric codes developed by the World Health Organization (WHO) for the classification of drugs and other medical products. Subgroup N06 is part of the anatomical group N Nervous system.
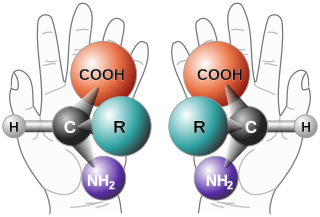
In chemistry, a molecule or ion is called chiral if it cannot be superposed on its mirror image by any combination of rotations, translations, and some conformational changes. This geometric property is called chirality. The terms are derived from Ancient Greek χείρ (cheir) 'hand'; which is the canonical example of an object with this property.

The American Journal of Psychiatry is a monthly peer-reviewed medical journal covering all aspects of psychiatry, and is the official journal of the American Psychiatric Association. The first volume was issued in 1844, at which time it was known as the American Journal of Insanity. The title changed to the current form with the July issue of 1921.
Forest Laboratories was a company in the pharmaceutical industry incorporated in Delaware, with its principal office in New York City. It was known for licensing European pharmaceuticals for sale in the United States. On July 1, 2014, the company was acquired by Actavis.
Allosteric serotonin reuptake inhibitor is a type of selective serotonin reuptake inhibitor (SSRI).
A spontaneous orgasm, or spontaneous ejaculation when it occurs in males, is an orgasm which occurs spontaneously and involuntarily without sexual stimulation. Nocturnal emissions may be considered a normal/physiological form of spontaneous orgasm. Pathological spontaneous orgasms can be experienced as pleasurable, non-pleasurable, or unpleasant, and can be distressing. Causes of pathological spontaneous orgasms include spinal cord lesions, psychological causes, rabies, and medications. Some cases may have no identifiable cause. Spontaneous orgasms may have no trigger or may be triggered by various non-sexual circumstances. They may occur in both males and females. Treatment of spontaneous orgasms include psychotherapy, selective serotonin reuptake inhibitors (SSRIs), the alpha-1 blocker silodosin, and anxiolytics.

A serotonin reuptake inhibitor (SRI) is a type of drug which acts as a reuptake inhibitor of the neurotransmitter serotonin by blocking the action of the serotonin transporter (SERT). This in turn leads to increased extracellular concentrations of serotonin and, therefore, an increase in serotonergic neurotransmission. It is a type of monoamine reuptake inhibitor (MRI); other types of MRIs include dopamine reuptake inhibitors and norepinephrine reuptake inhibitors.
The number of new psychiatric drugs, and especially antidepressants on the market in Japan, is significantly less than Western countries.
Reuptake inhibitors (RIs) are a type of reuptake modulators. It is a drug that inhibits the plasmalemmal transporter-mediated reuptake of a neurotransmitter from the synapse into the pre-synaptic neuron. This leads to an increase in extracellular concentrations of the neurotransmitter and an increase in neurotransmission. Various drugs exert their psychological and physiological effects through reuptake inhibition, including many antidepressants and psychostimulants.

Talopram, also known as phthalapromine, is a selective norepinephrine reuptake inhibitor (NRI) which was researched for the management of depression in the 1960s and 1970s but was never commercialized. Along with talsupram, talopram is structurally related to the selective serotonin reuptake inhibitor (SSRI) citalopram, as well as to melitracen:
In 1971, the company hired Klaus Bøgesø as a medicinal chemist. Over the years Bøgesø turned out to have a Midas touch at the game of drug hunting, creating more molecules that made it to the market than almost any other medicinal chemist in the field. The challenge facing him in 1971 following his recruitment was to produce a selective norepinephrine reuptake inhibitor. Like other companies at the time, Lundbeck had little interest in an SSRI. Bøgesø began from an accident in the laboratory. Trying to create a derivative of their norepinephrine reuptake inhibiting antidepressant melitracen, Lundbeck chemists accidentally produced a new chemical — a phenylphthalene. Against all the odds, just like melitracen, this was also a selective norepinephrine reuptake inhibitor. Two potential antidepressants came out of this — talopram and tasulopram, which were pressed into clinical trials. Both however turned out to be energizing, and in a number of cases there were suicide attempts. The fact that there were suicide attempts appeared to confirm another proposal of Paul Kielholz, that activating antidepressants might lead to suicide. Lundbeck's experience suggested that norepinephrine reuptake inhibitors were likely to lead to just this problem. Lundbeck retreated, scared. If norepinephrine reuptake inhibitors were likely to trigger suicide, the greatest hazard of an antidepressant, then Kielholz's view suggested that an SSRI would be less likely to lead to suicide. Bøgesø's job was to see whether the new series of drugs could be converted into a series of SSRIs. Following a lead from Carlsson on how to do this, he converted talopram into citalopram, the most selective serotonin reuptake inhibitor to come to the market.

Didesmethylcitalopram is an active metabolite of the antidepressant drug citalopram (racemic). Didesmethylescitalopram is an active metabolite of the antidepressant escitalopram, the S-enantiomer of citalopram. Like citalopram and escitalopram, didesmethyl(es)citalopram functions as a selective serotonin reuptake inhibitor (SSRI), and is responsible for some of its parents' therapeutic benefits.

Selective serotonin reuptake inhibitors (SSRIs) are a class of drugs that are typically used as antidepressants in the treatment of major depressive disorder, anxiety disorders, and other psychological conditions.
The molecular formula C20H21FN2O (molar mass: 324.39 g/mol, exact mass: 324.1638 u) may refer to:
Selective serotonin reuptake inhibitors, or serotonin-specific re-uptake inhibitor (SSRIs), are a class of chemical compounds that have application as antidepressants and in the treatment of depression and other psychiatric disorders. SSRIs are therapeutically useful in the treatment of panic disorder (PD), posttraumatic stress disorder (PTSD), social anxiety disorder, obsessive-compulsive disorder (OCD), premenstrual dysphoric disorder (PMDD), and anorexia. There is also clinical evidence of the value of SSRIs in the treatment of the symptoms of schizophrenia and their ability to prevent cardiovascular diseases.
The term "me-too drug" or "follow-on drug" refers to a medication that is similar to a pre-existing drug, usually by making minor modifications to the prototype, reflected in slight changes in the profiles of side effects or activity, and used to treat conditions for which drugs already exist. While pharmaceutical companies have justified the development of me-toos as offering incremental improvements in efficacy, side-effects, compliance and cost, critics have questioned the increasing marketing of me-toos, their absorption of research and development resources and their impact on the innovation of new treatments.